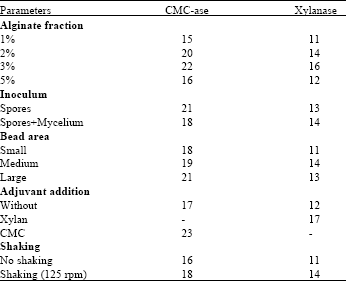
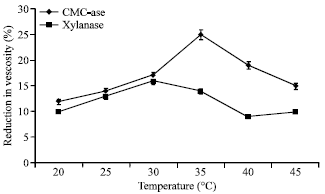
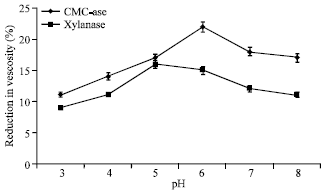
Research Article
Improvement of Carboxymethyl Cellulase and Xylanase Production by Alginate Immobilized Trichoderma harzianum
Omar Al-Mukhtar University, Faculty of Science, Department of Botany, Box 919, Al-Bayda, Libya
Baharuddin Salleh
Universiti Sains Malaysia, School of Biological Sciences, 11800, Pulau Pinang, Malaysia