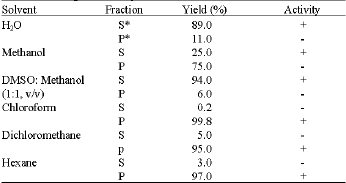
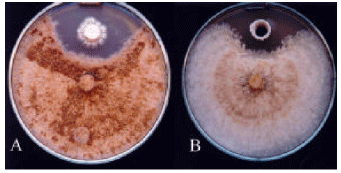
Research Article
Biological Control of Potato Isolate of Rhizoctonia solani by Streptomyces olivaceus Strain 115
College of Agriculture Science, Bahonar University of Kerman, Iran
G.H. Shahidi Bonjar
College of Agriculture Science, Bahonar University of Kerman, Iran
I. Saadoun
Jordan University of Science And Technology, Irbid, Jordan