ABSTRACT
This study is aimed at identifying differences in terms of the practical implementation of practical Chemistry teaching on the basis of teachers’ perspectives based on gender and location. Implementation of practical Chemistry in the current study focuses on four major aspects such as the design, preparation, implementation and evaluation. Furthermore, the study also explored the relationship among the aspects of design, preparation, implementation and evaluation. The study used a survey approach by using questionnaire. The study sample comprised of 236 Chemistry teachers, randomly selected in Pekan Baru, Riau in Indonesia. Pilot test involved 40 teachers and the findings of the pilot test revealed that the Cornbach Alpha value of the instrument constructs was between 0.81 and 0.91, hightening that the instrument has high level of reliability. The data was analyzed using inferential statistics such as independent t-test and Pearson correlation employing SPSS 19. The results revealed that there was no significant difference in practical implementation of practical Chemistry science among the teachers regarding the aspects of the design, preparation, implementation and evaluation based on gender and location. However, there is a significant different in relation to the aspect of preparation based on location. Meanwhile, Pearson correlation analysis indicated that there are significant correlations between and among all aspects associated with implementation of practical Chemistry. It is suggested that training and workshops for chemical science teachers can be implemented by the related organizations including the Ministry of Education, the Board of Education Quality Assurance as well as Center for Development and Empowerment of Educators for science. Besides, the collaboration is also highlighted for experienced teachers to be able to train and teach practical skills to novice teachers, especially in preparing tools and materials needed for practical activities.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajsr.2015.22.40
URL: https://scialert.net/abstract/?doi=ajsr.2015.22.40
INTRODUCTION
One of the most significant aspects of teaching and learning is design. The rationale behind this fact is that learning is a system that consists of several components that cumulatively lead to the goal. Therefore, learning must be planned appropriately. Kunandar (2007) defines Learning Implementation Plan (LIP) as a plan that describes the procedures and organizational learning to achieve a basic competence specified in the learning standard and elaborated in the syllabus contents. Rohani (2004) suggested that for building teaching and learning design, teachers need to have knowledge and skills in organizing learning design. A learning design is a tool that can assist teachers in implementing effective and efficient learning.
Practical chemical science preparation is the process of providing equipment, location or materials used before the implementation of the experiment. Preparation is also an activity to create a pre condition to learn, study the situation which arises interests and learning profit (Mulyati, 1995). According to Leonard et al. (2013), preparation consists of the use and handling of equipment and materials science correctly, storing equipment and materials science properly and safely, cleaning the scientific equipment in the right way, handling specimen correctly and carefully and drawing specimens, science equipments and materials accurately. Preparation for success and security related to practical work and preparation is one of the important processes to get the findings of the experiment.
Practical chemical science implementation is a practical teaching activity which is implemented in classes and labs. Directing activities of teaching is the teacher of the student in conducting experiments, doing observation on students in experimentation and analysis of laboratory findings and outcome-evaluation practice. Good research consists of processes and products with scientific method (including concepts, principles, theories and laws), both of which must be a component of science learned in school (Van Heuvelen, 2001). Evaluation in education and teaching including practical teaching Chemistry that can use several approaches. An approach commonly used in the evaluation of cognitive, constructivist (Dunham et al., 2002) and behaviorism theory can be applied in practical Chemistry evaluation essentially intended to measure the real physical behavior in the performance of the chemical laboratory experiment activity which can be observed directly. The prominent characteristic of this study is to acquire more preferred strategy compared to how much knowledge students acquire and recall. This theory is very well applied in practical teaching Chemistry because these assessment characteristics match to the characteristics and purposes of practical Chemistry lessons (Brooks, 2011).
Building a practical design is a process of chemical science internship program restructuring the related components of other practical material components, learning completeness, organizing practical preparatory measures, the implementing practical, practical evaluation, determining the chemical science practical purposes, allocating the time, the organization of internships and other environments (Marks and Eilks, 2009). Practical chemical science preparation is the process of providing equipment, place or materials used before the implementation of the experiment. Practical evaluation of chemical science includes practical observations of the overall process in the achievement of the purpose of learning as well as other practical assessments in experimentation, organization of systematic, practical work and practical results.
Figure 1 illustrates the conceptual framework of the study. As it can be seen, the four aspects of design, preparation, evaluation and implementation are interrelated. This study aims to identify the teachers’ perceptions of the aspects of design, preparation, evaluation and implementation based teachers’s background such as gender and location. The study also determines the relationship between and among these aspects.
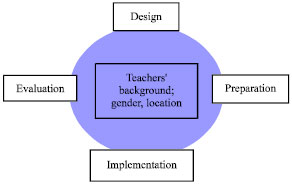 | |
| Fig. 1: | Conceptual framwork of the study |
STATEMENT OF THE PROBLEM
Chemical science teaching and learning is not only done in the classroom but also in the laboratory (Byers, 2002). This has long been a belief in science education laboratory that has the potential to become a place in which the theory is tested in a practical truth. The bulk of the teaching and learning takes place in a science laboratory (Mokhtar, 2007). In schools, science laboratory is the most appropriate place for students to learn how to research, organize, clarify and measure all the sciences. Most researchers agree that practical work is an important activity in school science but there are variations in the importance of the role and purpose of practical work done in the classroom. A number of teachers highlight the importance of conducting practical Chemistry science in schools in line with students’ achievement in the field of Chemistry science (Edelson, 2001; Kapenda et al., 2002).
Effective science teaching requires that teachers have the knowledge, skills, attitudes and ability to apply science in the lab in better way. This view supports that teachers should have the scientific competence (cognitive) and manipulative skills associated with psychomotor (Aktamis and Acar, 2010). Effective teaching happens if teachers have the knowledge and skill because the concept of proof in the form of laboratory Chemistry science is done through observation and performance analysis. Chemistry science teachers competencies are seen as scientific experimentation in the laboratory. To improve the quality and quantity of the adoption of practical Chemistry learning, Chemistry teachers are required to master in the skills of competency IPA process, the skills to use the equipment in the laboratory and laboratory management skills and the spirit of strong will and motivation to apply practical methods in learning Chemistry.
The teachers have not been able to implement practical evaluation in an objective Chemistry science to the provision of the information which is necessary to make alternative decisions, not fully established (Mehrens and Lehmann, 1999). In addition, lack of availability of teachers who master the teaching material, lack of teachers’ ability in using evaluation instrument and performing assessments are collecting reports from the implementation of practical evaluation of Chemistry science (Worthen and Sanders, 2000).
In the process of implementation of practical evaluation, science teachers often experience different problems and constraints that need to be solved. Teaching issues, among others, form slight efficiency in performance and reporting the results of the practical evaluation of Chemistry science. This problem needs a serious solution so that teachers’ proficiency can be improved (Grounlund, 2000). The problem of practical implementation of a practical Chemistry science as part of the science of Chemistry is prevailing. Practical processes ranging from how science teachers make practical design of science, how teacher preparation is done before the practical work is carried out and how the practical work is carried out under the guidance of teachers (implementation) and accordingly evaluation of their performances and students’ achievements are vital for teachers. Science education should be regarded as “education through science” rather than “science through education” (Holbrook and Rannikmae, 2007). This shows the importance of practical implementation of practical Chemistry which is the focus of the current study. Literature reports that very few studies on the practical implementation of practical Chemistry have been conducted in Indonesia (Copriady, 2013). Therefore, the current study is undertaken to address the problem and the research gap.
In conducting the litrature review, the aspects of design, preparation, implementation and evaluation were taken into consideration. In addition, the effects of teachers’ location and gender, in past studies, on practical Chemistry teaching will be discusssed.
PROBLEM-BASED LEARNING (PBL)
Given the importance of practical teaching of Chemistry, problem-based learning is discussed. Problem-Based Learning (PBL) refers to a teaching and learning method rooted in the medical sciences, first introduced in 1969, is increasingly popularizing in other academic disciplines including education, psychology as well as business (Coombs and Elden, 2004). Also, this method is becoming increasingly popular in science including Chemistry (Belt et al., 2002). Here, the development of PBL approach concerning a traditional Chemistry laboratory module through examining the traditional laboratory format to explore the rationale for a change. Accordingly, PBL will, then, be elaborated on to see how this method can solve this problem.
Traditional labs: Laboratory classes normally involve students carrying out teacher-structured laboratory exercises or/and experiments, where each step of a procedure is vigilantly prescribed and students are supposed to follow and keep on the procedures precisely. Generally, the students cannot be thinking and creative. This kind of laboratory activity is frequently known as a ‘recipe lab’ (Domin, 1999), in which little student involvement with the content is required. Likewise, Johnstone et al. (1994) expounded, “students can be successful in their laboratory class even with little understanding of what they are actually doing”. Nevertheless, the student may have little option but to accept this passive approach whilst, they deal with new techniques and/or equipment, particularly when the lab preparation involves no more than reading and understanding the laboratory manual. In this vein, Johnstone (1991) commented that the laboratory is regarded as an information overload place, resulting in students with little ‘brain space’ in order to process information and consequently, they blindly and thoughtlessly follow the instructions. In addition, they seldom interpret their observations or/and the results obtained during the experiment.
Hence, it is significant to emphasize the expense of running and administering laboratory sessions in school and university. First, it is costly to build, equip, maintain and uphold specialized laboratory space. Second, technical and academic staffing plus postgraduate demonstrators are required. Furthermore, laboratory work is time-consuming and lastly, there is ongoing and constant expense of consumable apparatus and chemicals (Bennett and O’Neale, 1998). However, the advantage of such laboratory sessions for students is questionable. Also, it is not clear if the usual and typical recipe lab format is justifiable concerning such expense.
The design of recipe labs activities is prearranged, as technicians, demonstrators and staff, all obviously knowing what is the activity and resulting outcome. Therefore, the teaching staff can clearly identify and rectify the errors for the students before continuing with the laboratory work and consequently the students get little problem-solving experience in the laboratory. Furthermore, all the students are usually performing the same experiment which can cause students to be only concerned with obtaining the same findings as their laboratory neighbor. Nevertheless, recipe labs enjoy the great merits that let the inexperienced student to have the same attitude towards laboratory work like professional scientist. Also, the recipe lets the student to dedicate all his/her attention to the method and not to be concerned at all about theory (Garratt, 1997). They take advantage of opportunities to directly improve manipulative as well as technical skills which maximize students’ practical experience as well as maximizes the validity of the results the students can potentially obtain. Contrariwise, the students do not care about matching their laboratory learning to previous experience. On the same note, Johnston (1991) declare “Consolidating their learning by asking themselves what is going on in their own heads”, despite the professional scientists and researchers conducting the laboratory work for a specific purpose which is meaningful for them. Another problem associated with recipe type labs is that the real practical aspect of any experiment shows only a small portion of the entire process of experimental science (Garratt, 1997), whereas in recipe labs only the practical aspect is covered.
Hunter et al. (2000) argues that the recipe lab deletes the planning and design stages” and it focuses on ‘data processing’ instead of ‘data interpretation’. Garratt (2002) suggests that there are various steps that can take a research scientist prior to dealing with the practical phase of the experiment:
| • | What questions are we trying to answer? |
| • | What observations would provide an answer to the questions? |
| • | How can we best create conditions for making the desired observations? |
| • | How will we process and evaluate the observations? |
| • | What will we do next? |
These issues comprise practical problem aspects that students have no connection with, as the laboratory technician and instructor decide on these issues long before the students embark on the experiment. Obviously, the recipe labs have their own merits and with some modifications, can be much more efficient and effective in the process of teaching and learning science. Integrating student ownership, connecting experiments with previous experiences, as well as helping students using higher order cognitive skills, can provide authentic and genuine investigative processes (Johnstone and Al-Shuaili, 2001). Laboratory sessions should provide the proper opportunity for students to hypothesize, criticize, analyze, explain and evaluate arguments and evidence. Likewise, Bailey (2001) emphasized the significance of transferable or broad-spectrum skill development in a UK context, recommending that a stress on transferable skills at the third level is greatly desirable.
Problem-based learning (a learner-centered approach): PBL perceives a shift in educational and learning focus from a teacher-dominated approach to teaching/learning to a student-dominated one, in which students create meaning for themselves through connecting new concepts and ideas with previous knowledge, an alternative approach to teaching/learning, encouraging dynamic and active engagement of the learner (Tan, 2004). As a learner-centered approach, it demands the learner to take a gradually increasing responsibility for his/her own learning. Therefore, PBL is in line with the constructivist theory (Coombs and Elden, 2004). Moreover, it also concentrates on anther aspect of constructivism that is associated with learning via social interaction, recognizing the influence of others’ views on the way of learners appreciate things (Harlen, 2005).
The purpose of PBL is developing reflective, self-directed and lifelong learners who are able to integrate knowledge, work collaboratively with other students and think critically (MacKinnon, 1999), hence enhancing the chances of students emerging with some of the skills, highly desirable and useful in the work place. Besides, by employing unstructured real-life problems instead of the content as the hub of attention, students are provided with opportunities to truly learn how to learn (Tan, 2004).
In this vein, White (2002) claimed that PBL could be perceived as an alternative to traditional method of education. Practical PBL, in which the problem is the focus of the learning, provokes lengthy collaboration among groups which leads to conceptual learning. Students automatically need to activate their prior knowledge to contemplate and start thinking, concerning the problem facing them and accordingly build new knowledge that is the main premise of constructivism. This has been demonstrated to augment learning which is in sharp contrast with traditional labs which make use of tasks with obvious procedures and true answers, related to limited exchange of information among students which leads to simple explanations plus routine learning (Wilkerson, 1996). Correspondingly, Belt et al. (2002) claim that problems are considered as the context as well as the driving force for learning. Thus, the achievement of new knowledge is carried out through these contexts. Practical PBL is different from the familiar case-based and/or problem-solving methods, as in PBL, the problems are faced before all the pertinent knowledge has been obtained and acquired (Albanese and Mitchell, 1993). Nevertheless, there are many implementation issues that need to be addressed. Similarly, Woods (1997) has listed the key issues with PBL implementation approach in relation to a traditional chemical lecture course as follows:
| • | Providing the students with the prerequisite skills |
| • | Students must be enthusiastic to take the responsibility of their own learning and to deal positively with the attitudinal changes that happen when they undergo change |
| • | Enabling the students to act as their own facilitators |
| • | Selecting and training teachers to handle PBL courses (known as the facilitators) |
| • | Encouraging student’s attendance and participation |
| • | Selecting and formulating the problem, as well as preparing resources and placing the student in groups |
Moreover, facilitators need to be eager to take risks as they take the responsibility of their own learning (Woods, 1997). The important factors useful for practical problem based learning include:
| • | Problems were developed according to existing experiments |
| • | Problems were designed to guarantee that students dealt with a pre-defined area of knowledge and also to assist students in learning a set of vital concepts, skills, ideas and techniques |
| • | Form that the problems typically take are descriptive statements |
| • | Students work either individually or in group |
| • | Students take part in a pre-lab, in which the students’ original answers to the problems are discussed with the instructor of lab who provide supplementary context and/or Chemistry support and also who highlight potential drawbacks prior to embarking on the lab work |
Theoretical background: The core aims of practical PBL comprise of the development of skills (general and technical), knowledge and understanding and chemical concepts. An alternative teaching and learning environment which combines pre-labs, group work, discussion, practical as well as alternative assessment, can help to achieve theses aims.
Skills: In the development of the practical PB laboratory, the first stage is to reflect on the favorite learning outcomes in relation to skills and scientific method developed from laboratory work. Garratt (1997) and Bennett and O’Neale (1998) depict the range of skills that should be developed via laboratory work as illustrated in Table 1. Laboratory work must also provide the students with the experience of designing an experiment, the process of science and combining subject knowledge with practical experience (Garratt, 1997). There are clear similarities between the skills associated with practical PBL proposed by these authors.
| Table 1: | Skills to be developed through practical work |
 | |
Skills including technical and observation skills, data collecting and confidence in practical work, are fundamental parts of most laboratory sessions. Nevertheless, as a result of reviewing the traditional module, it is revealed that skills like data interpretation, teamwork, problem solving and communication of findings are missing. Thus, it is crucial for the students to appreciate the opportunity to develop and use all these skills.
Teaching and learning environment: After determining the skills, techniques and concepts, the next phase involves designing the teaching/learning environment to cover pre-lab work, group work, discussion as well as alternative assessment.
Pre-lab: The use of pre-lab session prior to the laboratory to brain storm the learners is not a new issue. Johnston (1991) defines the elements of an efficient and effective pre-lab exercise as follows:
| • | Revision of theory |
| • | Re-acquaintance with skills |
| • | Planning the experiment to some degree |
| • | Discussion with peers |
When the abovementioned elements are combined with elements of ownership together with relevance for the students, consequently the pre-lab could be very effective for preparing the learner mind (Johnston, 1991). In addition, if students associate the laboratory experience with real life examples, they will be motivated to actually do the experiment.
Johnstone et al. (1998) expounded on the application of pre-labs to physics: “The aim of the pre-labs was to prepare students to take an intelligent interest in the experiment by knowing where they were going, why they were going there and how they were going to get there”. Furthermore, Sirhan et al. (1999) commented on pre-lectures in Chemistry being “a useful tool in enabling students to make more sense of lectures, the effort being particularly important for students whose background in Chemistry is less than adequate”. Likewise, Allen et al. (1996) reported how problems in practical PBL can be put forth with mini lectures.
Group work: Being involved in small-group co-operative learning, students can pursue their own learning within the context of the group. Also, they can refer to others for the purpose of support, validation and feedback. Interaction can become champion learning between group members, if it is suitably structured to permit discussion and consideration of different views (McManus and Gettinger, 1996). Group work is an integral part of practical PBL in and out of lab.
Discussion: Another important element in practical PBL is discussion. Discussion is referred to, broadly speaking, as a wide spectrum of informal situations in which talk between people occurs. Specifically, it is associated with a special arrangement of a group interaction in which members work collaboratively to address a question of common interest, while exchanging different viewpoints to build a better understanding of the issue in question (Bahar, 2003). Likewise, discussion before the lab class as well as during the pre-lab session is recommended in practical PBL. Nicol and Boyle (2003) demonstrated that students who have discussion in small groups not only their conceptual understanding is developed but also a strong motivating force is created. It was also stated that students indicated a preference for contemplating about the problem before the discussion. Accordingly, students would be more likely to involve in dialogue and to provide reasons for identifying gaps in their thinking. The purpose is to engage students to think creatively and critically and questions need to be posed to support these demands (Bahar, 2003).
Assessment: According to Savin-Baden (2004), currently assessment appears to be one of the most challenging and controversial issues in practical PBL. She declares “many of the concerns about assessment in higher education seems to relate to the unintended side-effects that undermine staff intentions to encourage students to learn effectively”. Her study revealed that students have following main challenging issues with assessment in practical PBL:
| • | Unrewarded learning |
| • | Disabling assessment mechanisms |
The effect of assessment on group work through PBL allows for more alternative and diverse assessment tools, such as written reports, oral and poster presentations and peer assessment, etc. The issue of undervalued and underrated learning in groups is a challenging one to address. Correspondingly, Overton (2001) expounded on various assessment tools employed in case studies:
“Assessment tools which have been successfully used include oral presentations to other scientists, oral presentations to a lay audience, written reports, summaries of data collected, peer assessment of group participation and individual reflection on skills development.”
Many supporters of practical PBL encourage the implementation of oral presentations in different disciplines (Allen and Tanner, 2003; McGarvey, 2004). Likewise, McGarvey (2004) has reported the application of poster presentations for Chemistry subject. He heightened the advantage of posters as he claims that, it triggers creativity and accordingly offers another platform for assessment, taking into account the students who might have been overlooked and unnoticed in the traditional assessment designs. In addition, due to their limited size, posters emphasize the significance of precise and concise information. Besides, they can promote collaborative work which cannot be supported by written lab reports. Lastly, he reported a positive attitude toward posters both by students and instructors.
Moreover, the written report is still advocated and has its benefits that it combines the method with results and allows students to report, analyze and draw conclusions concisely and informatively. Consequently, written reports constitute a fundamental part of the practical PBL, however, the stress is on the outcomes and conclusions drawn by the students as well as associating the findings with the original problem in hand.
Higher Order Thinking (HOT) legacy in international science curricula and assessment: Higher Order Thinking (HOT) has been used consistently in science education in terms of reform agendas internationally (Osborne and Dillon, 2008). Suggestions for promoting science education all over the European Union (EU) encompasses the following. Science courses that engage students in higher-order thinking includes constructing arguments, asking questions, making comparisons, establishing causal relationships, identifying hidden assumptions, evaluating and interpreting data, formulating hypotheses and identifying and controlling variables (Osborne and Dillon, 2008).
Higher order thinking in Chemistry: Osborne and Collins (2001) claim that assessment has remained part of the “forgotten landscape” or unaddressed asspect of science education research due to the capacity of assessment to champion or block a curriculum’s intent for Higher Order Thinking (HOT) by specifying the pedagogies used in school science classrooms (Fensham, 2006; Liang and Yuan, 2008).
Comparing the curriculum guidelines of 12th grade Physics with the test content of the exit examinations in China, shows the stress on low levels of knowledge remembering and application (Liang and Yuan, 2008). In fact, it was explored that the curriculum as well as examinations did not propmote or demand creativity, critical thinking or the capability to carry out scientific enquiry which are the skills generally associated with HOT. In China, there is a system called ‘the merit pay system’ which potentially reinforces a concentration on such low levels of cognitive demand in pedagogy along with learning since, it rewards teachers who train high achiever students in terms of examinations. These findings are consistent with research studies that have been done recently across two countries, namely New Zealand and Northern Ireland which explored that high-stakes summative assessment plus accountability measures blocked the students to develop thinking skill in terms of curriculum intent (Gallagher et al., 2012; Liang and Yuan, 2008) studies reported a comprehensive model of measurement-driven instruction designed precisely to employ assessment to modify and improve teaching practices. If standardized instruments focus on lower order thinking, then teachers are most likely going to focus their pedagogy on these goals. On the contrary, it can be anticipated that if assessment is designed to promote HOT, then this should influence teaching practices due to the “teaching-to-the-test” effect in standardized high stakes assessments.
Fensham and Bellocchi (2013) drew on a number of international examples in which teacher-generated assessment instruments provided the scope for attaining various HOT aspects of science curriculum intent that included connecting science and technology, involving students in decision making and developing knowledge about the nature of science. Zoller (1993) has also reported success in examination questions requiring Higher Order Critical Thinking Skills (HOCS) through the use of appropriate teaching strategies in undergraduate Chemistry classes. From these two sets of examples, it is evident that assessment regimes have the capacity to support or hinder any reform agenda for HOT or curriculum intentions for HOT. Furthermore, they suggest that the inflexibility of externally designed assessment instruments is more likely to hinder HOT, whereas the flexibility that is possible with internal instruments can support HOT.
In the context of this study, internal assessment refers to assessment instruments developed by school teachers and external assessment refers to assessment instruments (typically examinations) that are developed by assessment and curriculum organizations and administered to all students within a state or territory. The instrument inflexibility in an external test enables achievement scores to be compared norm referenced measurement. The flexibility, that internal instruments can have, makes such numerical comparisons difficult but the students’ performances can be assessed and compared against a commonly described set of criteria on standards-based measurement.
A degree of internal assessment is a key feature in the study, therefore, it is useful to indicate that we mean that the Chemistry teachers in a school take responsibility for the design and grading of their students’ work on a set of tasks that are carried out during the course of study. In the high stakes assessments , the nature of these tasks is externally prescribed in broad terms, as are the criteria for the award of the different grades. This type of teacher responsibility extends the assessment practices that science teachers regularly undertake in their classrooms to check the various learning tasks they set their students. A long-standing example of one of these tasks in science is practical investigative skills. These can only be assessed in a balanced way by the classroom teacher as argued by Black (2004) and Yung (2006), who has described how biology teachers in Hong Kong carried out this role in high stakes testing with reference to the issue of fairness and comparability. The intended learning outcomes that are now expected in contemporary science curricula extends more of this assessment responsibility to the classroom teacher (Fensham and Bellocchi, 2013).
A shift from the curriculum to the student: Prior to about 1975, the Chemistry education community was concerned essentially with the subject matter in terms of “What should we teach?” The implication was that the ‘answer’ for chemical education lay in the selection or design of the ‘right’ content for the curriculum. It was taken for granted that those who taught Chemistry knew the subject matter well.
Since then, there has been a surge in research into the question “What is learned?”. The focus has shifted from the curriculum to the student and reflection has given way to experimental investigation. Probing students’ understandings (‘misconceptions’ research) became an industry. The findings support the view that formal learning often constitutes little more than an ability to reproduce symbols and words and to apply algorithms.
Now we have encyclopedic collections of student misconceptions but usually no more than bland, general statements about preventative or curative actions. We have an enhanced knowledge of the conditions for effective learning, based upon which a range of student-centered teaching methodologies, such as cooperative learning, have become fashionable but little guidance as to how teachers might apply these to the teaching of particular Chemistry topics such as reaction kinetics or stereochemistry. Educational research has had little impact on science teaching.
Perhaps, this is partly because much of chemical education research has used Chemistry subject matter simply as a vehicle to develop ideas and theories of pedagogy, such as constructivist approaches to learning, co-operative learning, the purposes of laboratory work, metacognition, questioning, styles of learning and online learning, all of which can be considered independently of particular subject matter. I suspect that in many institutions, the emphasis of the teacher education programs are on the pedagogical issues that are the central objects of such research.
Commenting on pedagogy-based criteria commonly used for evaluation of teaching, Shulman (1986) asked “Where did the subject matter go? And what happened to the content?”. Perhaps, what is a productive path for us to travel (Shulman, 1987) has labelled Pedagogical Content Knowledge (PCK). While, content knowledge refers to one’s understanding of the subject matter and pedagogical knowledge refers to one’s understanding of teaching and learning processes independent of subject matter, pedagogical content knowledge refers to knowledge about the teaching and learning of particular subject matter, taking into account the particular learning demands inherent in the subject matter.
The rationale for doing this is aptly put by Geddis (1993).
The outstanding teacher is not simply a ‘teacher’ but rather a ‘history teacher’, a ‘Chemistry teacher’ or an ‘English teacher’. While in some sense, there are generic teaching skills, many of the pedagogical skills of the outstanding teacher are content-specific. Beginning teachers need to learn not just ‘how to teach’ but rather ‘how to teach electricity’, how to teach world history’ or ‘how to teach fractions’.Hence, in order to be able to transform subject matter content knowledge into a form accessible to students, teachers need to know a multitude of particular things about the content that are relevant to its teachability. There is a vast difference between knowing about a topic (content knowledge) and knowing about the teaching and learning of that topic (pedagogical content knowledge). Some knowledge about teaching and learning Chemistry is specific to the particular subject matter; the skills of teaching stereochemistry, for example, are different from those of teaching thermodynamics. In this study, selected examples from the topics of chemical equilibrium, thermodynamics and reaction mechanisms in organic Chemistry are used to illustrate the critical importance of PCK.
The profession of science teaching is afflicted with amnesia in the sense that the understandings that drive the strategies of competent teachers are seldom recorded, so new teachers need to develop their abilities ‘from scratch’ through experience, rather than stand on the shoulders of those who have gone before them. The chemical education enterprise is crying out for investigations that probe and report the topic-specific PCK of competent teachers, thus creating records from which new teachers can gain insights into their complex task. This can be regarded as applied research.
Part of PCK is an understanding of the various levels of operation engaged in which practising engage chemists. A more refined model than the famous macroscopic-submicroscopic-symbolic triangle of Johnstone (1982) is developed and presented. This includes the view that people engaged in thinking, imagining and musing (i.e., modelling) are at the heart of the Chemistry enterprise and that the courses in the subject ought to reflect this. Samuelowicz and Bain (2001) conducted two case studies on the academics’ beliefs about teaching and learning. Table 2 illustrated the results of two case studies.
They provided two stories to illustrate the nexus between belief and practice and to demonstrate that the differences between teaching-centred and learning-centred orientations are substantial.
The first of these is teaching-centered, whereas, the second is learning-centered, as the stories presented earlier clearly illustrate. While in both cases, the academics want their students to gain a thorough understanding of the subject matter, their beliefs about the nature of understanding and learning and about their roles in knowledge organization and teacher-student interaction vary substantially. In the first case, the academic provides ready made understandings and methods for students, shows them how to apply the knowledge and interacts with them to ensure that the understanding has taken place. In the second case, the teacher assists the students, through extensive interaction, to personalize their understanding of the material and to use their new understanding to interpret the world in an altered way. These two orientations share only two of nine beliefs.
| Table 2: | Comparison of the two illustrative cases organized by belief dimensions |
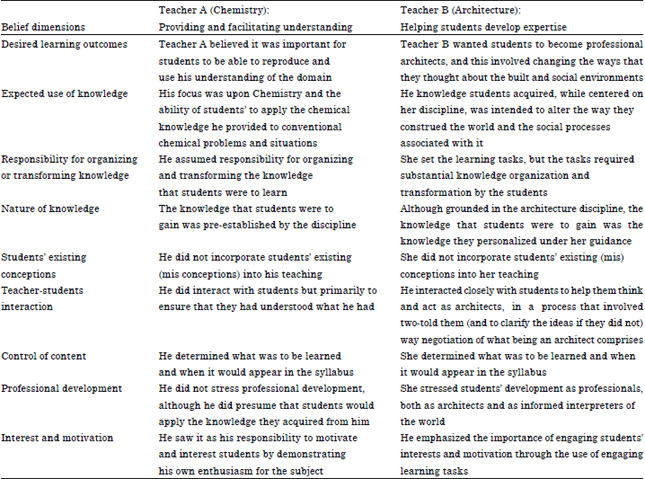 | |
| Samuelowicz and Bain (2001) | |
Teacher’s gender: Abuseji (2007) reported that teacher’s gender has direct influence on students’ achievement in Chemistry. The direct effect was responsible for 0.97% of the total effect of all the seven independent variables on student’s achievement in Chemistry while, its indirect effect accounted for 3.37% of the total effect. In total, teacher’s gender accounted for 2.40% of the total effect of the seven independent variables on students’ achievement in secondary school Chemistry. This finding is consistent with the studies of Orosan et al. (1992), Reap and Cavallo (1992) and Smith (1992), who claimed that gender could predict academic achievement. However, this finding contradicts the studies of Onocha (1985) and Miller (1984), who found that gender alone has no effect on academic achievement but could act in conjunction with other variables to affect learning outcomes.
METHODOLOGY
The current study employed a survey approach. The study involved a total of 234 participants who were randomly selected among the teachers in secondary schools in Pekan Baru, Riau in Indonesia. The total of the sample is appropriate based on list of percentage suggested by Krejcie and Morgan (1970). The study used a questionnaire regarding the practical implementation of practical Chemistry in the secondary school, consisting of four constructs such as practical design comprising 24 items, practical preparation involving 22 items, practical implementation involving 27 items and evaluation involving 29 items. The questionnaire was adapted from Sampson (2004), Muijs and Reynolds (2001) and Meehan et al. (2004). A pilot test was conducted on 40 high school Chemistry teachers in Pekan Baru, Riau, in Indonesia, who were not involved in the main study. The pilot test results indicate that the study constructs have high reliability with Cronbach Alpha values between 0.81-0.91.
Statistical analysis: To analyze the obtained data, inferential statistics including independent t-test and Pearson correlation were employed using SPSS 19.
RESULTS AND DISCUSSION
Differences regarding practical design, preparation, implementation and evaluation based on gender: Independent t-test analysis was conducted to identify the differences of design, preparation, implementation and evaluation based on gender. Independent t-test analysis results are shown in Table 3.
Table 3 shows that there is no significant difference in practical designs (t = 1.480 and sig. = 0.140, p>0.05), practical teacher preparation (t = 0.254 and sig. = 0.254, p>0.05), practical implementation (t = 0726 and sig. = 0.469, p = 0.469) and practical assessment (t = 0.214 and sig. = 0831, p>0.05) based on gender. The results show that male students have a higher mean compared to female students for every aspect of the study. However, there is no significant difference in the design, preparation, implementation and evaluation based on gender. This is because teachers have the same advantage in the teaching and learning process. Chemical science instructional design guidelines have brought teachers together in the Assembly Subject Teachers (MGMP). Practical Chemistry science tools and materials are appropriate to the students’ and teachers’ needs to be performed by male and female teachers. On the practical level, male and female teachers have equal opportunity in practice as well as the assessment. The findings are in line with Achor et al. (2012) that there was no significant difference in the level of creativity in terms of gender. This finding is also supported by research study conducted by Bayraktar (2011) that there was no significant difference in teaching methods and gender factors.
Differences regarding practical design, preparation, implementation and evaluation based on location: Independent t-test analysis was conducted to identify the differences of design, preparation, implementation and evaluation based on location. Independent t-test analysis results are shown in Table 4.
| Table 3: | Result of independent t-test differences regarding design, preparation, implementation and evaluation based on gender |
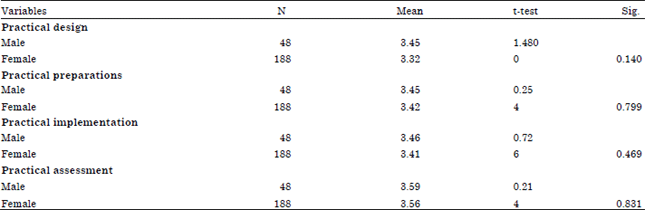 | |
| Table 4: | Differences regarding design, preparation, implementation and evaluation based on location |
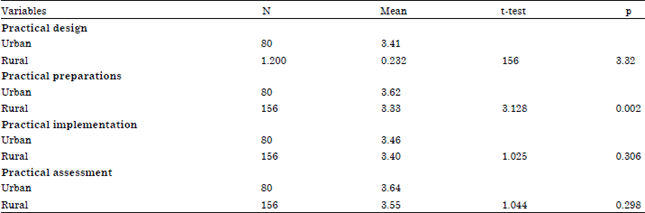 | |
| Table 5: | Pearson correlation significant relationship between design, preparation, implementation, and evaluation of practical Chemistry science |
 | |
| **Significant at p<0.05 | |
Table 4 shows that there are differences based on the practical preparation with the value of t = 3.128 and sig. = 0.002, p<0.05, while there are no significant differences in the practical design with value t = 1.200 and sig. = 0.232, p>0.05), practical implementation (t = 1.025, sig. = 0.306, p>0.05) and practical assessment (t = 1.044 and sig. = 0298) based on location. It means that there is no significant difference in SMA teacher competency in Riau on the urban and rural areas. Regarding practical preparation, this indicates that the chemical science teachers who have taught more in urban area can prepare tools and materials which are better than those of rural teachers (Copriady, 2013; Cheung, 2009). The result is in contrast with Rosinah (2005) research findings that teachers of urban and rural schools do not adopt a different inquiry-discovery. Data also reveals that teachers in urban areas have better facilities, equipments and materials compared to their counterparts in the rural areas.
Significant relationship between design, preparation, implementation and evaluation: Pearson correlation analysis was conducted to identify the relationship between the design, preparation, implementation and evaluation. Pearson correlation analysis results are shown in Table 5.
Correlation analysis shows a significant relationship between and among instructional designs and teaching preparation, implementation and evaluation as the strength of the relationship is moderate. This shows that the better the Chemistry teaching design, the better the preparation, implementation and evaluation activities. Furthermore, there is a significant relationship between the preparations and the implementation and the evaluation and the strength of the relationship is moderate. Moreover, there is a significant correlation between the implementation of the evaluation. Practical design has a major function in the practical implementation of scientific and practical design which reflects all the processes. Correlation analysis shows a significant relationship between and among instructional designs and teaching preparation, implementation and evaluation as the strength of the relationship is moderate. This shows that the better the Chemistry teaching design, the better the preparation, implementation and evaluation activities. Furthermore, there is a significant relationship between the preparations and the implementation and the evaluation and the strength of the relationship is moderate. Moreover, there is a significant correlation between the implementation of the evaluation. Practical design has a major function in the practical implementation of scientific and practical design which reflects all the processes performed in the practical chemical science lessons. Teachers need to make a comprehensive design, appropriate material, determine tools and materials as well as determine practical teaching procedures and practical evaluation methods (Chang et al., 2009). The importance of teaching in the laboratory is generally defined as inquiry which involves a range of activities in making observations, asking questions, examining books and resources that are already in the know, planning investigations, observing what has been demonstrated in experiments which clearly shows the evidence, filing an answer, explaining, predicting and communicating the results (Marks and Eilks, 2009).
Likewise, there is a strong need to develop the life-cycle assessment education in Chemistry and also the overall sustainability education must become more extensive in science (Bybee and McCrae, 2011).
IMPLICATIONS AND RECOMMENDATIONS
However, some studies have highlighted the importance of gender and school location but in the current study, gender and location are not important. It is knowledge that is important. Practical implementation among teachers in design, preparation, implementation and evaluation based on gender and location is almost the same. This shows that the design of practical Chemistry science is a significant concern for chemical science teachers with a variety of demographic backgrounds.
It is recommended that training and workshops for chemical science teachers be implemented by different organizations including the Ministry of Education, the Board of Education Quality Assurance as well as Center for Development and Empowerment of Educators for science teachers in making instructional design. In addition, the collaboration is also emphasized for experienced teachers to be able to train and teach practical skills to novice teachers, especially in preparing tools and materials needed for practical activities (Catalano, 2010; Copriady, 2013). In addition, teachers are also required to provide the student teachers with the practical results which are implemented in the theoretical knowledge of chemical science to avoid failure in achieving the learning objectives.
CONCLUSION
This study was conducted to identify differences in the practical implementation of practical Chemistry teaching based on teachers’ gender and location. Practical implementation encompassed the aspects of the design, preparation, implementation and evaluation. The study used a survey approach using questionnaire. Moreover, the study determined the relationship between teachers’ competency concerning design, preparation, implementation and evaluation. The results showed that there was no significant difference in learning practical implementation of practical Chemistry science regarding the aspects of the design, preparation, implementation and evaluation based on gender and location. Furthermore, Pearson correlation analysis showed that there were significant correlations between all abovementioned aspects.
Based on study findings and discussion, it is recommended that that there should be further study on the curriculum and learning standards for practical laboratory Chemistry science. In addition, in-depth study of teacher competence in the teaching of practical Chemistry based science, teacher profile analysis and competency is suggested.
REFERENCES
- Albanese, M.A. and S. Mitchell, 1993. Problem-based learning: A review of literature on its outcomes and implementation issues. Acad. Med., 68: 52-81.
PubMed - Allen, D. and K. Tanner, 2003. Approaches to cell biology teaching: Learning content in context-problem-based learning. Cell Biol. Educ., 2: 73-81.
CrossRef - Bahar, M., 2003. The effects of motivational styles on group work and discussion-based seminars. Scand. J. Educ. Res., 47: 461-473.
CrossRefDirect Link - Bailey, P.D., 2001. Teaching chemists to communicate? Not my job. Univ. Chem. Educ., 5: 80-86.
Direct Link - Belt, S.T., E.E. Hywel, T. McCreedy, T.L. Overton and S. Summerfield, 2002. A problem based learning approach to analytical and applied chemistry. Univ. Chem. Educ., 6: 65-72.
Direct Link - Bennett, S.W. and K. O'Neale, 1998. Skills development and practical work in chemistry. Univ. Chem. Educ., 2: 58-62.
Direct Link - Chang, H.Y., C. Quintana and J.S. Krajcik, 2009. The impact of designing and evaluating molecular animations on how well middle school students understand the particulate nature of matter. Sci. Educ., 94: 73-94.
CrossRef - Cheung, D., 2009. Students' attitudes toward chemistry lessons: The interaction effect between grade level and gender. Res. Sci. Educ., 39: 75-91.
CrossRefDirect Link - Coombs, G. and M. Elden, 2004. Introduction to the special issue: Problem-based learning as social inquiry-PBL and management education. J. Manage. Educ., 28: 523-535.
CrossRefDirect Link - Copriady, J., 2013. The implementation of lesson study programme for developing professionalism in teaching profession. Asian Soc. Sci., 9: 176-186.
Direct Link - Domin, D.S., 1999. A review of laboratory instruction styles. J. Chem. Educ., 76: 543-547.
CrossRefDirect Link - Dunham, T., J.G. Wells and K. White, 2002. Biotechnology education: A multiple instructional strategies approach. J. Technol. Educ., 14: 65-82.
Direct Link - Edelson, D.C., 2001. Learning-for-use: A framework for the design of technology-supported inquiry activities. J. Res. Sci. Teach., 38: 355-385.
Direct Link - Fensham, P.J. and A. Bellocchi, 2013. Higher order thinking in chemistry curriculum and its assessment. Skills Creativity, 10: 250-264.
CrossRefDirect Link - Abuseji, F.A., 2007. Student and teacher related variables as determinants of secondary school students academic achievement in chemistry. J. Pendidikan, 32: 3-18.
Direct Link - Gallagher, C., R. Hipkins and A. Zohar, 2012. Positioning thinking within national curriculum and assessment systems: Perspectives from Israel, New Zealand and Northern Ireland. Thinking Skills Creativity, 7: 134-143.
CrossRefDirect Link - Garratt, J., 1997. Virtual investigations: Ways to accelerate experience. Univ. Chem. Educ., 1: 19-27.
Direct Link - Geddis, A.N., 1993. Transforming subject-matter knowledge: The role of pedagogical content knowledge in learning to reflect on teaching. Int. J. Sci. Educ., 15: 673-683.
CrossRefDirect Link - Van Heuvelen, A., 2001. Millikan lecture 1999: The workplace, student minds and physics learning systems. Am. J. Phys., 69: 1139-1146.
CrossRef - Aktamis, H. and E. Acar, 2010. The effect of laboratory practices in science teaching course on development of prospective science teachers self-regulation skills. Procedia Soc. Behav. Sci., 2: 5549-5553.
CrossRefDirect Link - Holbrook, J. and M. Rannikmae, 2007. The nature of science education for enhancing scientific literacy. Int. J. Sci. Educ., 29: 1347-1362.
CrossRefDirect Link - Hunter, C., S. Wardell and H. Wilkins, 2000. Introducing first-year students to some skills of investigatory laboratory work. Univ. Chem. Educ., 4: 14-17.
Direct Link - Johnston, K., 1991. High school science teachers conceptualisations of teaching and learning: Theory and practice. Eur. J. Teach. Educ., 14: 65-78.
CrossRefDirect Link - Johnstone, A.H. and A. Al-Shuaili, 2001. Learning in the laboratory: Some thoughts from the literature. Univ. Chem. Educ., 5: 42-51.
Direct Link - Kapenda, H.M., H.U. Kandjeo-Marenga, C.D. Kasandra and F. Lubben, 2002. Characteristics of practical work in science classrooms in namibia. Res. Sci. Technol. Educ., 20: 53-65.
CrossRefDirect Link - Krejcie, R.V. and D.W. Morgan, 1970. Determining sample size for research activities. Educ. Psychol. Meas., 30: 607-610.
CrossRefDirect Link - Liang, L.L. and H. Yuan, 2008. Examining the alignment of Chinese national physics curriculum guidelines and 12th‐grade exit examinations: A case study. Int. J. Sci. Educ., 30: 1823-1835.
CrossRefDirect Link - MacKinnon, M.M., 1999. CORE elements of student motivation in problem-based learning. New Directions Teach. Learn., 1999: 49-58.
CrossRefDirect Link - McGarvey, D.J., 2004. Experimenting with undergraduate practicals. Univ. Chem. Educ., 8: 58-65.
Direct Link - Meehan, M.L., K.S. Cowley, N.L. Finch, K.L. Chadwick, L.D. Ermolov and M.J.S. Riffle, 2004. Special strategies system-revised observations: A useful tool for educational research and evaluation. Appalachia Educational Lab Inc. (AEL), Charleston, WV., USA. http://edvantia.ehclients.com/site-assets/04SSOS-R.pdf.
- Nicol, D.J. and J.T. Boyle, 2003. Peer instruction versus class-wide discussion in large classes: A comparison of two interaction methods in the wired classroom. Stud. Higher Educ., 48: 457-473.
CrossRefDirect Link - Orosan, P.G., A.M. Weine, L.A. Jason and J.H. Johnson, 1992. Gender differences in academic and social behavior of elementary school transfer students. Psychol. Schools, 29: 394-402.
Direct Link - Overton, T.L., 2001. Teaching chemists to think: From parrots to professionals. Univ. Chem. Educ., 5: 62-68.
Direct Link - Marks, R. and I. Eilks, 2009. Promoting scientific literacy using a sociocritical and problem-oriented approach to chemistry teaching: Concept, examples experiences. Int. J. Environ. Sci. Educ., 4: 231-245.
Direct Link - Sampson, V., 2004. The science management observation protocol: Using structured observations to improve teachers' management of inquiry-based classrooms. Sci. Teacher, 71: 30-33.
Direct Link - Shulman, L.S., 1986. Those who understand: Knowledge growth in teaching. Educ. Researcher, 15: 4-14.
Direct Link - Smith, T.E., 1992. Gender differences in the scientific achievement of adolescents: Effects of age and parental separation. Soc. Forces, 71: 469-484.
CrossRefDirect Link - Tan, O.S., 2004. Students' experiences in problem-based learning: Three blind mice episode or educational innovation? Innov. Educ. Teach. Int., 41: 169-184.
CrossRefDirect Link - White, H.B., 2002. Commentary: Problem-based learning and disciplinary boundaries. Biochem. Mol. Biol. Educ., 30: 420-420.
CrossRefDirect Link - Zoller, U., 1993. Are lecture and learning compatible? Maybe for LOCS: Unlikely for HOCS. J. Chem. Educ., 70: 195-197.
CrossRefDirect Link - Achor, E.E., S.M.S. Kurumeh and C.A. Orokpo, 2012. Gender dimension in predictors of students' performance in MOCKSSCE practical and theory chemistry xaminations in some secondary schools in Nigeria. Educ. Res., 2: 16-22.
CrossRefDirect Link - Allen, D.E., B.J. Duch and S.E. Groh, 1996. The power of problem-based learning in teaching introductory science courses. New Directions Teach. Learn., 1996: 43-52.
CrossRefDirect Link - Bayraktar, S., 2011. Turkish preservice primary school teachers' science teaching efficacy beliefs and attitudes toward science: The effect of a primary teacher education program. Sch. Sci. Math., 111: 83-92.
CrossRef - Bybee, R. and B. McCrae, 2011. Scientific literacy and student attitudes: Perspectives from PISA 2006 science. Int. J. Sci. Educ., 33: 7-26.
CrossRefDirect Link - Johnstone, A.H., S.J. Sleet and J.F. Vianna, 1994. An information processing model of learning: Its application to an undergraduate laboratory course in chemistry. Stud. Higher Educ., 19: 77-87.
CrossRefDirect Link - Johnstone, A.H., A. Watt and T.U. Zaman, 1998. The students' attitude and cognition change to a physics laboratory. Phys. Educ., 33: 22-29.
CrossRef - McManus, S.M. and M. Gettinger, 1996. Teacher and student evaluations of cooperative learning and observed interactive behaviors. J. Educ. Res., 90: 13-22.
Direct Link - Osborne, J. and S. Collins, 2001. Pupils' views of the role and value of the science curriculum: A focus-group study. Int. J. Sci. Educ., 23: 441-467.
CrossRef - Samuelowicz, K. and J.D. Bain, 2001. Revisiting academics' beliefs about teaching and learning. Higher Educ., 41: 299-325.
CrossRef - Savin‐Baden, M., 2004. Understanding the impact of assessment on students in problem‐based learning. Innovations Educ. Teach. Int., 41: 221-233.
CrossRef - Sirhan, G., C. Gray, A.H. Johnstone and N. Reid, 1999. Preparing the mind of the learner. Univ. Chem. Educ., 3: 43-46.
Direct Link - Wilkerson, L., 1996. Tutors and small groups in problem-based learning: Lessons from the literature. New Directions Teach. Learn., 1996: 23-32.
CrossRef








