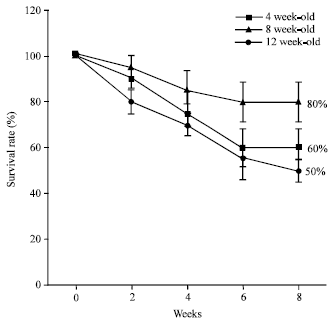
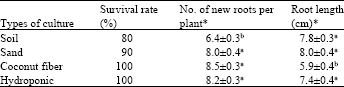
Research Article
Influence of Plantlet Age and Different Soilless Culture on Acclimatization of Stemona curtisii Hook.f.
Department of Biology, Faculty of Science, Chiang Mai University, Chiang Mai, 50200, Thailand
Srisulak Dheeranupattana
Department of Biology, Faculty of Science, Chiang Mai University, Chiang Mai, 50200, Thailand
Araya Jatisatienr
Department of Biology, Faculty of Science, Chiang Mai University, Chiang Mai, 50200, Thailand
Sunanta Wangkarn
Department of Chemistry, Faculty of Science, Chiang Mai University, Chiang Mai, 50200, Thailand
Pitchaya Mungkornasawakul
Department of Chemistry, Faculty of Science, Chiang Mai University, Chiang Mai, 50200, Thailand
Stephan Pyne
School of Chemistry, University of Wollongong, New South Wales, 2522, Australia
Alison Ung
School of Chemistry and Forensic Science, University of Technology Sydney, Sydney, 2007, Australia
Thanapat Sastraruji
School of Chemistry and Forensic Science, University of Technology Sydney, Sydney, 2007, Australia