Research Article
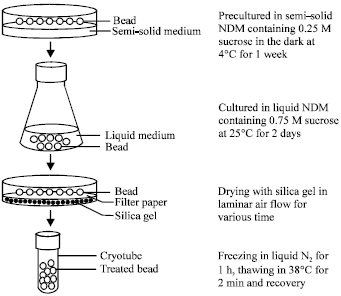
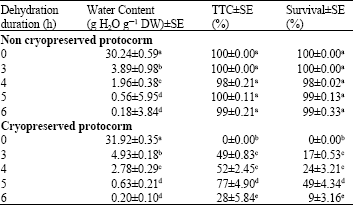
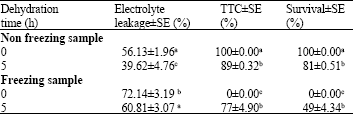
In vitro Conservation of Cleisostoma areitinum (Rchb.f.) Garay, Rare Thai Orchid Species by an Encapsulation-Dehydration Method
Applied Taxonomic Research Center, Department of Biology, Faculty of Science, Khon Kaen University, Khon Kaen 40002, Thailand
S. Bunnag
Applied Taxonomic Research Center, Department of Biology, Faculty of Science, Khon Kaen University, Khon Kaen 40002, Thailand
M. Monthatong
Applied Taxonomic Research Center, Department of Biology, Faculty of Science, Khon Kaen University, Khon Kaen 40002, Thailand