Research Article
Micropropagation of Water Chestnut (Trapa sp.) Through Local Varieties of Rajshahi Division
Not Available
A. Nahar
Not Available
M.A. Razvy
Not Available
M.K. Biswas
Not Available
A.H. Kabir
Not Available
Water Chestnut (Trapa sp.) is an annual aquatic dicotyledonous plant bearing starchy fruit. It grows in shallow fresh water basin in the tropical, sub-tropical and temperate zone of the world (Daniel et al., 1983; Kumar et al., 1985). It has been commercially cultivated in Japan, China, Pakistan, Srilanka and different parts of Southeast Asia including Bangladesh. Now it is going to be very popular for its rich nutritional food value (Pandit and Quadri, 1986). The edible part of water chestnut is its cotyledon. The starchy fruit contains starch, protein, fat and significant amount of calcium, vitamins, phosphorus, iron, potassium, zinc and ascorbic acid (Khan and Chughtai, 1955; Bargale et al., 1987). For human consumption water chestnut is eaten as a vegetable or flavor, or is added in other dishes. It is used also to make colored powder and dye. Beside this, it has a great medicinal value (Kosuge et al., 1985).
Water chestnut can be cultivated in paddy field as well as in natural fresh water pond or lake. Efficient large-scale production of water chestnut in rice paddy field requires mass supply of uniform and disease free propagules. If we can increase the multiplication of seedlings through in vitro technique, then it can easily provide more plantlet from a few seeds. Micropropagation is an important biotechnological approach to achieve this goal. However, reports on micropropagation of water chestnut are few in number (Zhou et al., 1983; Agrawal and Mohan Ram, 1995; Hoque et al., 2001). Furthermore, seasonal effect on seed dormancy can be overcome through in vitro micropropagation. This study describes a simple method of micropropagation for obtaining large-scale disease free seedling through local varieties of Rajshahi Division. This results adequate supply of uniform and disease free propagules of water chestnut with a view to manipulate for different purposes.
Plant material: Mature and fresh fruit of water chestnut were harvested from a water chestnut aquatic field at Rajshahi, Naogaon and Natore district of Bangladesh. The excess unnecessary parts were removed from the collected materials and the remaining parts were then cut into convenient size (1-1.5 mm). The materials were washed thoroughly under running tap water for several times to reduce the dust and surface contaminants. Then the fresh materials were stored in controlled environment at 5°C without light.
Sterilization and callus induction from embryonal explant: Fruits were rinsed in distilled water containing few drops of tween 80 and savlon for 15-20 min. Then the fruits were washed 6-8 times for removing these chemical substances. The apical end of the fruit partially covering the large cotyledon, plumule, small cotyledon and hypocotyl, was excised with aseptic tools (7-8 mm3) in front of laminar air-flow cabinet. The fruits were then surface-sterilized with 0.1% HgCl2 solution for 7 min with continuous shaking. Then the sterilized explants were washed 6-7 times with double-distilled water immediately to remove all traces of HgCl2.
After proper disinfection, the pericarp was removed and the embryonal explants (2-3 mm long) were excised and cultured on semi-solid (0.61% agar) MS (Murashige and Skoog, 1962) medium supplemented with different combination and concentration of 2,4-D (0.1, 1.0, 1.5 mg L-1), BA (0.5, 1.0, 1.5 mg L-1) and NAA (0.5, 1.0 mg L-1). The response to callus induction varied greatly with respect to different combination and concentration of growth regulators. Usually callus proliferation was started from the cut surface of the explant and finally covered the whole explant. Among all treatment, callus induction was found to be best in medium containing 2.5 mg L-1 2,4-D and 1.0 mg L-1 BA in all varieties. These calli are light green in color but the degree of callus formation was moderate. Phloroglucinol (PG) and casein hydrolyzate (CH) were also added to the medium with different concentration to reduce the accumulation of phenolic compounds (Hoque et al., 2002). Callus induction from embryonal explants was recorded after 4 weeks of culture.
Shoot proliferation from callus: For organogenesis, embryo derived callus were subcultures into media containing half strength MS medium supplemented with various concentration of BA (1.1, 2.2 mg L-1) alone or in combination with NAA (0.5, 1.1 mg L-1) and GA3 (0.5 mg L-1). Callus were inoculated directly into solid, semi-solid and liquid medium for comparison. Comparative analysis of data showed that liquid medium was responsible for highest number and length of shoot. Culture vessels and filter paper bridge were used for inoculation in case of solid or semi-solid and liquid medium, respectively. In all cases, pH of the media was added to 5.7 with 1 N NaOH and 0.5 N HCl solution prior to autoclave sterilization.
Rooting and transfer of plantlets: Rooting of in vitro derived shoots was obtained on half strength liquid medium supplemented with IBA and GA3. It has been observed that 0.1 mg L-1 IBA was found to be most effective for root formation. For transferring to soil rooted plantlet were thoroughly washed to eradicate all traces of the medium. Then the rooted micro-cutting were successfully transplanted to paddy field and the survival rate was found to be 95%.
Callus induction and subsequent regeneration of plantlet in water chestnut was a difficult task because of the secretion of phenolic substances by explant. Therefore, blackening of explant and medium was reported (Wiermann, 1981). Accumulation of phenolic substances was reduced and callus was promoted by the addition of PG (Hoque and Arima, 2000). A similar role of PG was reported in Piper nigrum (Sarasan and Nair, 1991) and in Aristolochia indica (Manjula et al., 1997).
Effects of different concentrations and combinations of growth regulators on callus induction are shown in Table 1. In this study, inhibition in callus formation has been prevented by the use of PG and CH (Fig. 1A).
| Table 1: | Effect of different concentration and combination of growth regulators on callus induction from embryogenic explants. Data were recorded after 6 weeks of culture |
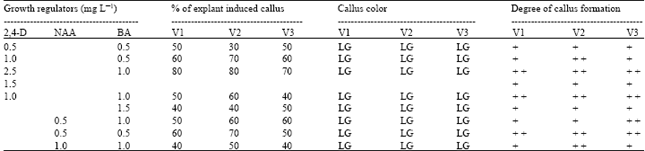 | |
| LG = Light Green, + + + = Massive callus, + + = Moderate callus, + = Slight callus, V1 = Trapa quadrispinosa, V2 = T. bispinosa, V3 = T. acornis | |
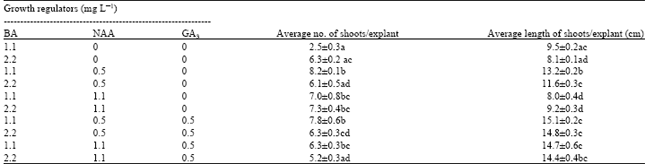
| Table 2: | Effect of different concentration and combination of growth regulators on shoot multiplication and elongation from embryonal explants of water chestnut after 4 weeks of culture |
 | |
| Table 3: | Effect of different concentration and combination of growth regulators on root induction from callus derived shoots. Data were recorded after 3 weeks of inoculation |
 | |
| Comparison of mean values obtained in treatments were made by using Duncan’s multiple range test; Values with different letter(s) within column are significantly different at p<0.05 | |
 | |
| Fig. 1: | Different stages of micropropagation technique of water chestnut. A- Overcoming phenolic compounds with the addition of CH and PG. B- Callus induction of embryonal explant in the medium supplemented with 2.5 mg L-1 2,4-D and 1 mg L-1 BA. C- Proliferation of shoots from callus in the medium supplemented with 1.1 mg/ BA and 0.5 mg L-1 NAA. D- Rooting of in vitro derived shoots in the medium supplemented with 1.1 mg L-1 IBA. E- Mass propagation of water chestnut in natural water |
The best medium for callus induction in this study was half strength MS medium containing 2.5 mg L-1 2,4-D and 1 mg L-1 BA for all the varieties treated. The calli in this medium showed 80% frequency and grew quickly than other combination of growth regulators treated. T. bispinosa showed satisfactory callus induction on medium supplemented with 0.5 mg L-1 NAA and 0.5 mg L-1 BA, but the degree of callus formation was not best. Induced calli were light green in color (Fig. 1B). Degree of callus formation was massive in 2.5 mg L-1 2,4-D and 1 mg L-1 BA containing medium.
Shoots were regenerated when the calli were transferred to regeneration medium. Various concentrations and combinations of BA, NAA and GA3 were tested in the shoot inducing medium to determine the best combination and concentration for shoot induction. The summarized results are presented in Table 2. Highest average shoot number per explants was observed 1.1 mg L-1 BA + 0.5 mg L-1 NAA containing medium. Highest average length of shoots per explant was found in 1.1 mg L-1+0.5 mg L-1 NAA+ 0.5 mg L-1 GA3 containing medium. BA is the most effective cytokinin for enhancing organogenesis in various plant (Kane et al., 1993; Hoque et al., 2000; Jenks et al., 2000). In this experiment it has been found that BA along with NAA and GA3 showed satisfactory result in shoot growth (Table 2). Best shoot multiplication was found on MS half strength medium containing 1.1 mg L-1 BA, 0.5 mg L-1 NAA and 0.5 mg L-1 GA3 (Fig. 1C). GA3 is known to have stimulatory effect on stem elongation in different plants (Phinney, 1984; Manjula et al., 1997). There were no significant different among the three varieties used in this study with respect to shoot multiplication.
For root proliferation, mainly auxiliary shoots were excised from the shoots of the primary culture and placed in the liquid rooting medium. Efficient rooting from the base of excised shoots was observed and summarized in Table 3. In response to various concentrations of IBA and GA3, IBA was most effective for root proliferation. In most cases, two roots emerged from the node of nearest cut end of the stem within 7-10 days in presence of 1.1 mg L-1 IBA (Fig. 1D). Trapa quadrispinosa showed higher number of root per shoot and the length of root was also highest in this variety. Rooted plantlets were brought out from the culture chamber and rooted micro-cutting were transferred to plastic pot lacking stress condition to acclimatize them in ex vitro condition. After a few weeks micro-cutting were successfully transplanted to the paddy field (Fig. 1E).
The methods of callus induction and in vitro regeneration of plantlets presented in this paper could open a new way of making large-scale production of propagule of water chestnut for nursery purpose in our Rajshahi region. Moreover, it is an efficient method for obtaining disease free water chestnut as well as to develop a genetic transformation system for varietal improvement of water chestnut.