Research Article
Controlling of Alternaria Leaf Spot Disease on Faba Bean Using some Growth Substances
Department of Leguminous and Forage Crops Disease, Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt
Faba bean (Vicia faba L.), which has common names (broad bean, fava bean, horse bean and field bean) native to North Africa and Southwest Asia. It is grown mainly for its green pods and dried seeds, which are rich in protein, vitamins, carbohydrates, dietary fibers, minerals and secondary metabolites such as phenolics. Thus, it is an important component of the human nutritional consumption (Randhir et al., 2002; Sahile et al., 2011). Furthermore, it improves the soil fertility by fixing atmospheric nitrogen leads to reducing costs and minimizing impact on the environment. Therefore, increasing the plant crop production is one of the major targets of Egypt Agriculture policy (Mahmoud et al., 2004; Bendahmane et al., 2012).
Faba bean is suffering from many destructive diseases. It is attacked by more than 100 pathogens in the Mediterranean region. Diseases can inflict great losses in faba bean production. During the last years, Alternaria leaf spot disease caused by Alternaria alternata cause serious damage to the plant and decrease of yield production due to changing in temperature degrees (by increasing from natural values) in Egypt (Honda et al., 2001; Reis et al., 2007; Juroszek and von Tiedemann, 2011). The lesions first appear on the lower leaves as small, brown circular spots which slowly increase in size and develop concentric brown rings with dark margins. Occasionally, an old rust pustule is found in the center of lesion, indicating that the leaf spot fungus may have entered through dead pustule tissue. Alternaria leaf spot generally occurs late in the season as plants start to mature (Bernier et al., 1993).
It is an urgent need to apply alternative safe efficient methods against fungal plant diseases due to the hazard of fungicides on public health and environmental balance. It is well known that normal plant growth and development are partially controlled by chemical substance produced by the plant. Recently, attempts have been made to use growth regulator substances not only to increase yield but also to control or minimizing the damage and loss caused by plant pathogens. For this reasons, both growth promoters (IBA and GA3) and growth retardants (Ethrel and PBZ) used as fungicides that control fungal plant diseases (Abdalla, 2001; Abd El-Hai, 2001; Khalifa, 2003; Metwally et al., 2006; Abd El-Hai et al., 2010).
The aim of this research is an attempt to study the effect of growth substances on some morphological, physiological traits and yield. In addition to reducing the negative effects of Alternaria leaf spot disease on faba bean.
Plant material and growth substances: Faba bean seeds cv. Maser 1 were obtained from Legume Crops Research Depart, Agricultural Research Center, Giza, Egypt. Meanwhile, Indole butyric acid 99% ("IBA" Aldrich Chemical Company, England), Gibberellic acid 92% ("GA3" Berelex tablets, Imperical Industries Plc, plant protection division "ICI', England), Ethrel (2-chloroethyl phosphoric acid) obtained from BDH chemicals LTD, U.K and Paclobutrazol (PBZ) "2RS-3RS-1-(4-chlorophenyl)-4,4-dimethyl -2-(1,2,4-triazol-1-yl)-pentan-3-01) as well as chemical fungicide (Tridex-80% wp "Mancozeb group") were used in this investigation.
Isolation of Alternaria leaf spot causal pathogen: Alternaria alternata was isolated from naturally infected faba bean leaves collected from different fields of Dakhlia Governorate. The isolated fungus was identified based on cultural and microscopic morphological characters according to the key given by Barnett and Hunter (1977).
In vitro: This experiment aimed to study the effect of growth substances and Tridex-80 % on linear growth of A. alternata. The tested growth substances (IBA, GA3, Ethrel and PBZ) at 50, 100 and 150 ppm as well as Tridex-80% at 2.5 g L–1 were separately mixed with PDA before solidification, then poured in a sterile petri dishes. Four plates for each concentration were inoculated with fungal disc of A. alternata which had been cut from the periphery of 7 days old culture. Four plates inoculated with A. alternata only without any amendment served as control. The plates were incubated at 20°C, linear growth was observed daily and diameter of fungal colonies were recorded when particular control filled of petri dishes with fungal mycelia.
In vivo: Experiments were carried out at Tag El-Ezz Agricultural Research Station, Dakahlia, Egypt under natural infection during 2013/2014 and 2014/2015 winter seasons. The experiments aimed to study the effects of foliar spray treatments of growth substances and Tridex-80% on reduce Alternaria leaf sot disease of faba bean and some physiological aspects, growth and yield. Seeds of faba bean were sown in 23 and 15 November in the first and second seasons, respectively. At the age of 50, 65 and 80 days in tow seasons, the plants were sprayed until dripping using small pressure pump with one of the concentration of treatments. A complete randomized block design with three replicates was used in the two mentioned seasons.
Disease assessment: Disease Severity (DS) of Alternaria leaf spot was recorded at 90 days from sowing using the scale of Vakalunakis (1990), based on the leaf area infected (class rate from 0-4), then calculated as disease severity %, using the following formula:
| NPC | = | No. of plants in each class rate |
| CR | = | Class rate |
| NIP | = | No. of infected plants |
| MSC | = | Maximum severity class rate |
The Disease Incidence (DI) of Alternaria leaf spot as, a disease percentage was determined according to the following formula:
Physiological traits: After 80 days from sowing, the following physiological activities were determination:
| • | Photosynthetic pigments (chlorophyll a, b, total chlorophylls and carotenoids) were determined in the blade of terminal leaflet of third leaf from plant tip according to Mackinney (1941) |
| • | Total phenolic compounds were determined in fresh shoot using the Folin-Ciocalteau reagent according to Malik and Singh (1980) |
| • | Total soluble carbohydrate were extracted from 2 g shoot dry matter by ethanol 70% for overnight at laboratory temperature (Kayani et al., 1990) then determined by the Anthron method (Sadasivam and Manickam, 1996) |
Morphological characters and yield components: At harvest, plant height, number of branches/plant, pods number/plant and weight of 100 seed were recorded.
Statistical analysis: All data were subjected to statistical analysis of software CoStat (2005) using analysis of variance technique (Gomez and Gomez, 1984).
In vitro
Antifungal activity of growth substances: Great variations in the linear growth of the pathogen were recorded with different types of growth substances (Fig. 1). PBZ at any concentration and high concentrate of Ethrel (150 ppm) completely inhibited the growth of Alternaria alternata.
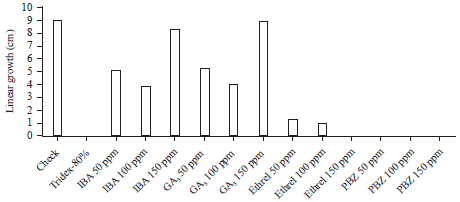 | |
| Fig. 1: | Effect of growth substances on Alternaria alternata growth |
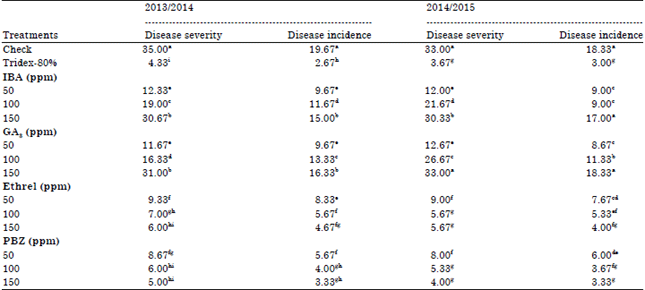
| Table 1: | Effect of growth substances on disease severity and disease incidence of Alternaria leaf spot in faba bean plant |
 | |
| Different letters within a column indicate significant difference at p≤0.05 | |
These results come online with this recorded on the fungicide Tridex-80%. While, the high level of IBA and GA3 (150 ppm) had no significant effect in fungal growth. The inhibitory effect of growth retardants (Ethrel and PBZ) on fungal growth may be due to the inhibition of ergestrol biosynthesis and blocking C-14 demethylation reaction in fungi (Fletcher and Hofstra, 1990). Ergestrol is essential for maintenance of fungal membrane structure and without it the physical stability of membranes, as well as, the activity of membrane bound enzymes can be affected adversely (Brenneman and Murphy, 1991). Meanwhile, plant growth promoter (IBA and GA3) increased both of pathogen growth and host growth (Al-Masri et al., 2002). Moreover, Reddy and Strzelczy (1989) stated that GA3 is growth stimulators for hyphae and stimulated spore germination of many pathogenic fungi.
In vivo
Disease assessment: Data of disease severity and disease incidence of Alternaria leaf spot of faba bean under field condition were recorded in Table 1. Application of growth substances at any concentration used and Tridex-80% significantly reduced Alternaria leaf spot DS and DI as compared with check. Except for fungicide Tridex-80%, the maximum reduction of disease severity and incidence were recorded by PBZ at 150 ppm followed by 100 ppm then Ethrel at 150 ppm. While, IBA and GA3 at 150 ppm came to late in both seasons. It is worthy to mention that, no significant differences among Tridex-80%, PBZ (100 or 150 ppm) and Ethrel 150 ppm.
Similar results were previously reported that using triazoles in controlling of potato early blight. PBZ increased epicuticular-wax which acts as protective waxy layer on the surface of the plant organs and decrease the size of stomatal openings (Fletcher and Hofstra, 1990). The outer thick cuticle of plant organs seem to be the barriers to inward movement of fungal mycelium (Singh et al., 1980). Moreover, PBZ increased activity of antioxidants enzymes that protect cell organelles from oxidative damage (Sankhla et al., 1992). In addition, PBZ increased Ca content that increase the rigidity of plant cell walls by complexing with wall matrix polysaccharides, which lead to be more resistant against fungal invasion (Abdalla, 2001). In addition to, the inhibitory effects growth substances on fungal diseases may be due to its effects on fungal growth, sporulation and sclerotial formation (Khalifa, 2003). Abd El-Hai et al. (2010) added that, Ethrel and IBA increased chlorophyll, total phenol and proline content. Proline and phenolics are used as indicator for controlling any stress conditions. Moreover, chlorophyll content is a good parameter reflecting the health condition of any plant. More, phenolic are well known as antifungal, antibacterial and antiviral. Gogoi et al. (2001) reported that, the first step of plant defence mechanism involves a rapid accumulation of phenols at the infection site, which restricts or slows the growth of the pathogen. With respect to Ethrel effects, Bovio et al. (1987) stated that, ethylene releasing from Ethrel induced plant resistance. Chowdhury (2003) found that treated plants with growth regulators lead to increase in total phenol, calcium content and chatechol oxidase activity, which protect plants against pathogen stress. Meanwhile, growth substances reduced the activity of pectolytic enzymes. The GA3 decrease cell size that makes them denser, thus increasing resistance by forming a biological barrier (Ketykova et al., 1985). In addition to, GA3 at high concentration of gibberellic acid caused increasing in activity of lytic enzymes (Reddy and Strzelczy, 1989).
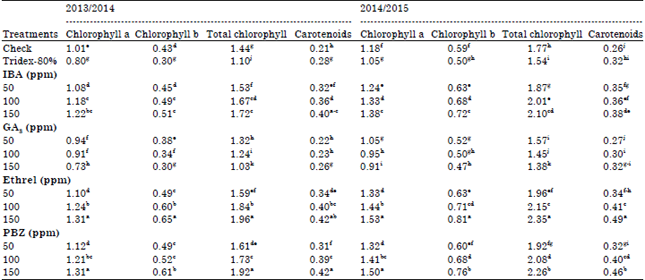
Physiological traits: Data in Table 2 show that all photosynthetic pigments (chl. a, b and carotenoids) increased significantly under application of IBA, Ethrel and PBZ.
| Table 2: | Effect of growth substances on photosynthetic pigments (mg g–1 fresh weight) of faba bean leaves |
 | |
| Different letters within a column indicate significant difference at p≤0.05 | |
There are appositive relationship between concentrations and its effects. The PBZ followed by Ethrel was more effective in this respect. While, GA3 at any concentrate and Tridex-80% decreased chl. a and b, but increased carotenoids content. The high level of GA3 (150 ppm) was more effective.
Carotenoids are known as major endogenous plant antioxidants. In addition, chlorophyll is a good parameter of plant healthy. The reduction in chlorophylls (a and b) by GA3 application may be due to destruction of pigments, reduced rate of synthesis, or dilution of pigments as a result of increased cell size and rate of cell expansion induced by GA3. In addition, GA3 induced chloroplast degradation and apparently interferes directly with chlorophyll metabolism (Straub and Lichtentholer, 1973). On the other side, the increase in photosynthetic pigments due to Etherl may be explained by the stimulation of chlorophyll synthesis and the delay of chlorophyll destruction by increased cytokinin content in the leaves. Cytokinin causes an increase in the number of chloroplasts in the leaf by increasing both intensity of cell growth phytohormones and the activity of cytoplasm ribosome, thus chlorophyll synthesis is stimulated (El-Zawily et al., 1994). Paclobutrazol also increased cytokinin content (Izumi et al., 1988), which delaying chlorophyll destruction (Dalziel and Lawrence, 1984). Moreover, Fletcher and Arnold (1986) stated that the increase in photosynthetic pigments by PBZ may be due to a simulation of chlorophyll production due to higher level of cytokinins content, reduction in chlorophyll degradation and delaying the senescence.
Total phenols and total soluble carbohydrate in faba bean plants were recorded in Table 3. The application of growth substances at any concentrate increase significantly total phenols and total soluble carbohydrates compared with check. Ethrel came to first followed by PBZ then IBA, hence GA3 came late. In contrast, fungicide Tridex-80% decreased significantly total soluble carbohydrate in faba bean shoot in both seasons. While, total phenols increased using fungicide treatment.
Phenols accumulation rapidly at the infection site for restricts or slows the growth of the pathogen due to its action as antioxidants, antimicrobial and photoreceptor (Lamba et al., 2008). In our study, growth substances (IBA, Ethrel and PBZ) increased photosynthetic pigments consequently increased total soluble carbohydrates. In addition, Ethrel and PBZ increased cytokinin content in plant that accelerates the translocation of C-14 assimilated to storage parts (Mauk et al., 1986).
| Table 3: | Effect of growth substances on total phenol (mg/100 g fresh weight) and total soluble carbohydrate (mg g–1 dry weight) of faba bean plant |
 | |
| Different letters within a column indicate significant difference at p≤0.05 | |
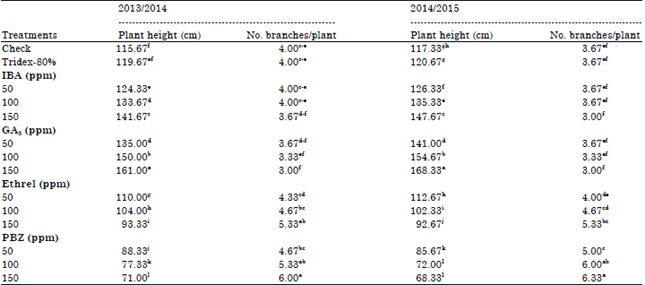
| Table 4: | Effect of growth substances on some morphological characters of faba bean plant |
 | |
| Different letters within a column indicate significant difference at p≤0.05 | |
Moreover, Rashad (1998) reported that GA3 increase the activity of amylase, invertase and sucrose synthesis. These enzymes play an important role in increasing hydrolysis and translocation of carbohydrate from shoot to root. Because the accumulation of carbohydrates in shoot may lead to inhibition of photosynthesis (Paul et al., 1992).
Morphological characters and yield components: As shown in Table 3 and 4, spraying faba bean plants with growth promoter (Ethrel and GA3) increased significantly plant height, while growth retardants (Ethrel and PBZ) inverse this. The highest values of plant height occurred under GA3 at 150 ppm. In contrast, PBZ give the lowest values. In the other side, number of branches/plant increased significantly under the application of growth retardants (Ethrel and PBZ), PBZ was the most effective.
Concerning the effects of growth substances on number of pods and weight of 100-seed, data in Table 5 shows that number of pods/plant was increased significantly by any level of growth substances used. Ethrel give the maximum values followed by PBZ then GA3. Moreover, weight of 100-seed was increased significantly under all growth substances used except, the low level of IBA (50 ppm) which showed no significant effect. PBZ give the highest values followed by Ethrel.
The stimulatory effect of GA3 on plant height may be due to increase in cell division, number of internodes and/or elongation of each internode (Bruce, 1990; Abdel-All, 1994; Deotale et al., 1998). Whilest, the suppression in plant height due to Ethrel my have been resulted from a reduction of cell elongation and/or through inhibition of cell division and elongation of sub apical meristems (Sen and Naik, 1977). Moreover, Anderson (1971) stated that the decrease in plant height and increase in number of branches due to Ethrel might be attributed to the liberation of ethylene. Ethylene is a natural growth regulator, which causes among many biological effects, inhibition of terminal bud growth and stimulation of lateral shoot development (Helaly et al., 1984). In addition to, Ethrel decreasing auxin levels which in turn inhibition of both cell division and elongation (Ibrahim et al., 1990). They added that the effect of GA3 on plant height closely related to an increase in IAA. The increase in number of branches by the application of PBZ may be due to the role of PBZ on stimulation of lateral shoot development (Davis et al., 1988).
| Table 5: | Effect of growth substances on yield components of faba bean plants |
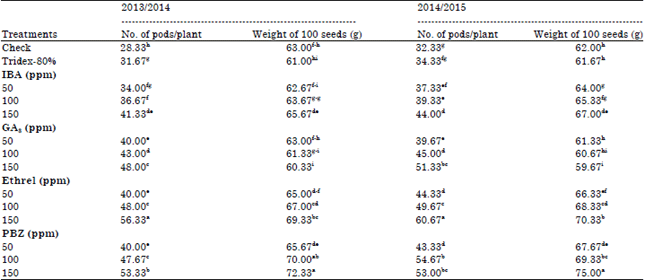 | |
| Different letters within a column indicate significant difference at p≤0.05 | |
The GA3 increased number of pods/plant due to increase in flowering period (Castro et al., 1987). Moreover, the stimulating effect of Ethrel on number of pods/plant might be related to enhancing photosynthetic pigments formation in the leaves and cyclic photophosphorylation. Ethrel also stimulating axillary buds development giving rise to more lateral branches. The increase in seed weight due to Ethrel may be the result of a certain role on stimulating the formation of conducting elements allowing the passing of more nourishing materials to be accumulated in the developing seeds (Arafa, 1982). The PBZ and GA3 increased number of inflorescences/plant, number of pods and seed number (Abd El-Hai, 2001), thus increased plant yield. Also, PBZ increased cytokinin content which delay chlorophyll destruction and senescences leading to increase plant yield (Izumi et al., 1988).
It could be concluded hat foliar spraying of PBZ, Ethrel and IBA at 150 ppm during the growth of faba bean is recommended for reducing Alternaria leaf spot disease and improving growth and yield.