ABSTRACT
Archetypal fungal resistance is marked by deform macroscopic growth on artificial medium supplemented with fungicides and the overexpression of resistance proteins weaponries at the molecular level. The interaction of Fusarium oxysporum f.sp. dianthi. with fungicides viz., copper oxychloride, metalaxyl-mancozeb and, mancozeb under heat-stress condition was studied. We evidence the pathogen interrelatedness to elude combine heat and fungicides stress is chiefly governed by the differential over expression of a 22.9 kDa Resistant Heat Shock Protein (RHSP) and a 26.3 kDa housekeeping protein (HKP). Of the assayed fungicides, resistant protein suppression was a function of temperature rise acting as a positive variance for copper oxychloride; and as a negative variance for mancozeb and metalaxyl-mancozeb. This first report suggests predicting field temperature prior to application of fungicide of choice is keyed to avert resistance resurgence. We infer within the context of global warming, the interaction of F. oxysporum f.sp. dianthi with fungicides inherently fluctuates and lends credence for the expression of adaptive RHSP responsible for its resiliency, a predicament for the farmers cry.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=ajppaj.2012.19.24
INTRODUCTION
Fusarium wilts, caused by soilborne Fusarium oxysporum f.sp. dianthi. (Prill. and Delacr.) W.C. Snyder and H.N. Hans, is the most calamitous phytopathogen for tuberous crops (Agrios, 1988). In India alone, 80-100% yield loss is associated with the F. oxysporum species (Anjani et al., 2004). Tubers incessantly deteriorate in store-house once the pathogen gain access to host even after pretreatment with agrochemicals. Performing fungicides in the field harbours carbamate and phenylamide derivatives as active ingredient notably for mancozeb and metalaxyl, respectively. Despite the harmful effects of fungicides such as resistance and cross-resistance within fungi, insects and weeds population (FRAC, 2009), fungicides form the scaffold strategy for extensive seed treatment (Khanzada et al., 2002), while the used of some bacterial isolates to control F. oxysporum f.sp. radicis-lycopersici (Kerkeni et al., 2008) and F. oxysporum f.sp. psidii (Srivastava et al., 2011) proved to be effective.
Pioneer post genomics sequencing and comparative genomics studies revealed pathogenicity mobility on Fusarium chromosomes (Ma et al., 2010), providing substantial clue that Fusarium spp. can adapt to weird conditions. Unquestionably, profiling resistance protein weaponries under chronic stress conditions provide invaluable insight on F. oxysporum f.sp. dianthi. inherent adaptability to key fungicides and global warming trend. Reports on fungicides efficiency on F. oxysporum strains (Amini and Sidovich, 2010; Taskeen-Un-Nisa et al., 2011) focuses on variability in antifungal potential and sensitivity. The lack of information how the pathogen proteome changes and pilot adaptation to global heat-stress trends has hoodwinked farmers on the choice of fungicides considering the global heating scenario. Given the commercialization of registered metalaxyl-mancozeb blend (MAGNET® 8/64%w/w, RPC AgroIndustries, India), we hypothesize its molecular effectiveness to halt resistant proteins expression to be superior over mancozeb (INDOFIL Chemicals®, India, 75%WP) and inorganic copper oxychloride (INDOFIL Chemicals® India, 75%WP) in the context of global warming. This study was conducted to analyse the expression of adaptive proteins in the virulent isolate F. oxysporum f.sp. dianthi. obtained from sampling S. tuberosum L. farms in India (West Bengal, Burdwan region) under combined heat- and fungicide- stress.
MATERIALS AND METHODS
Between June 2010 to December-2010, Fusarium isolates were recovered from infected potato (Solanum tuberosum L.) tubers on Yeast Extract Agar Peptone (YEAP) medium amended with 0.75 mg L-1 chloramphenicol and 0.25 mg L-1 ampicillin, within the state of West Bengal-India, Burdwan District. Subsequent to pathogenicity test on healthy tubers, the most virulent strain was identified by the Division of Plant Pathology-IARI, New Dehli (Ref. No. 3095/2010) and commercialized registered copper oxychloride, mancozeb and metalaxyl-mancozeb were assayed. Fifty millilitre sterile Liquid Potato Sucrose Medium (LPSM) were inoculated with 5 mm diameter mycelium plug in 100 mL Erlenmyer flask. Each fungicide concentration per flask were maintained at 10 and 50 mg kg-1 and incubated at 39 and 24°C, respectively for a photoperiod of 16/8 h day and night for 5 days with occasional shaking.
A second lot of controls: (1) Positive control void of fungicide but heat stress at 39°C and 2) negative control void of fungicide and heat stress in LPSM were cultured as mentioned above. Homogeneous fresh mycelium growth were harvested with a triple-fold cheesecloth and air-dried for 10 min on Whatmann paper N°1. One gram of each sample was crushed in a pre-chilled mortar and pestle, containing 0.1 g silica gel-G (Merck), 10 mg SDS (sodium dedocylsulphate) and 100 mM, Tris-HCl pH 7.2 buffer system [containing 0.1% β-mercaptoethanol, 0.1% ascorbic acid and 0.1% cysteine-HCl and 2 mM PMSF (phenylmethylsulphonylfluoride)]. Crushed samples were vortexed at 10,000 rpm/10 min/4°C and supernatants precipitated with 30% tricarboxylic acid. Pelleted samples were triple washed using 98.8% acetone and aliquots stored in 100 mM, Tris-HCl pH 7.2 buffer containing 2 mM PMSF at -20°C.
Proteome change analysis by SDS-PAGE was done as follows: Protein concentrations were determined by the standard Bradford (1976) method using Biorad® bovine serum albumin for calibration. All protein samples were profiled on a 15% polyacrylamide gel (Merck) (Lemmli, 1970) at constant voltage for approximately 6 h. The gels were stained using 50% methanol and 7% glacial acetic acid, 0.2% Coomassie Blue R250 overnight. The gels were destained in two steps: first with 50% methanol, 7% acetic acid for 1 to 2 h and completed with 7% methanol and 7% acetic acid. The gels were analyzed using Bangalore-genie precision molecular weight markers and photographed using the ST4 Quantum Biogel documentation system. The experiment was carried out in quadruplet and choice profile is represented.
Fungicides antifungal potential were evaluated using the food poison technique (Grover and Moore, 1962) on YEAP at 24°C and percentage radial growth inhibition were calculated for triplicates using Pandey et al. (1982) method. Data were analyzed using Statistical Package for Social Science (SPSS) 17.0 for the determination of mean and significant differences. One way ANOVA and Tukey’s test for estimation of smallest significant difference (p = 0.05) were applied.
RESULTS
Mancozeb (M) exhibited a 100% radial growth inhibition at 500 mg kg-1, while the maximum of metalaxyl-mancozeb (MM) and copper oxychloride (Ce) stood at 58.77 and 76.01%, respectively at 500 mg kg-1. Hence, metalaxyl-mancozeb blend emerged less active at 24°C compared to mancozeb as depicted in Fig. 1a and b.
Figure 2a and b depicts the impact of heat stress on F. oxysporum f.sp. dianthi. interaction with fungicides. It shows a differential expression pattern manifested by the overexpression of a 26.3 kDa housekeeping protein (HKP) at all stress conditions. Amongst assayed fungicides, metalaxyl-mancozeb blend at 24°C/50 mg kg-1 considerably deactivated the expression of the 26.3 kDa HKP but induced a 56.7 kDa resistant protein Fig. 2a (lane 2). Moreover, none of the fungicides at 24°C/50 mg kg-1 suppressed the constitutively expressed 128.6 kDa protein (Fig. 2a; lane 2, 3, 4). No major inducible protein was observed with mancozeb (lane 3) at 24°C/50 mg kg-1 but also failed to suppress the 26.3 kDa HKP. The pathogen reacted violently with copper oxychloride at 24°C/50 mg kg-1 (Fig. 2a, lane 4) leading to the induction of a 77.4 kDa resistant protein. On the other hand, Fig. 2b (lane 2 and 4) revealed heat-stress single-handedly suppressed resistance proteins encoders except the 22.9 kDa RHSP encoder.
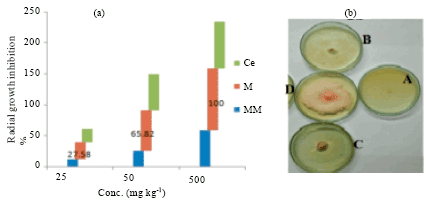 | |
| Fig. 1(a-b): | (a) Antifungal potentials of fungicides against F. oxysporum f.sp. dianthi. Ce: copper oxychloride, M: Mancozeb, MM: Metalaxyl-mancozeb. Significant inhibition at 500 mg kg-1; where no significant difference exist at p = 0.05 (5%) according to Tukey’s test. (b): Radial growth inhibition after 5 days, 24°C at 500 mg kg-1. A: Mancozeb (M), B: Metalaxyl-mancozeb (MM), C: Copper oxychloride (Ce), D: Control, no fungicide and heat-stress |
 | |
| Fig. 2(a-b): | (a) Lane 1 and 6-Control at 24°C, fungicide free. Lane 2-MM at 50 mg kg-1. Lane 3-M at 50 mg kg-1. Lane 4-Ce at 50 mg kg-1. Lane 5 and 10-Molecular weight marker. All 50 mg kg-1 were cultured at 24°C in LPSM. Lane 7-MM at 10 mg kg-1. Lane 8-M at 10 mg kg-1. Lane 9-Ce at 10 mg kg-1. All 10 mg kg-1 were cultured at 39°C in LPSM. (b): Lane 1-Molecular weight marker. Lane 2 and 4-heat stress (39°C), fungicide free. Lane 3-Heat stress free and fungicide free |
However, the pathogen at 24°C-fungicide-free proliferated abundantly by producing an array of proteins with major ones being 77.2, 69.9, 64.8, 56, 49.8, 45.9, 30.1, 26.3, 22.9, 18.2 and 17.7 kDa as depicted in Fig. 2b (lane 3).
DISCUSSION
Despite the huge application of fungicides, S. tuberosum L. is enticed to low yield in some regions even when the crop escape foliar damages mediated by the early and late blight diseases due to the presence of soil-borne Fusarium spp. Its been proposed coupling abiotic inducers to pathogen elicitors is a better as alternative to fungicides (Alkahtani et al., 2011). In this study we described the first report evidencing that F. oxysporum f.sp. dianthi. can effectively eludes carbamates, phenylamides and, the blend of the two derive fungicide while responding to heat-stress using principally a 22.9 kDa RHSP.
The present analysis shows more proteins are suppressed by copper oxychloride at 39°C/10 mg kg-1 than at 24°C/50 mg kg-1 vis-à-vis mancozeb and metalaxyl-mancozeb blend at same concentration. Implying, the pathogen sensitivity under heat-stress decreases for mancozeb and metalaxyl-mancozeb blend than copper oxychloride. Following, FRAC (2009) reports, phenylamides (or metalaxyl) targets RNA polymerase I and show resistance and cross resistance to the Oomycetes. This may imply inability of phenylamides to halt de novo synthesis of rRNA in F. oxysporum f.sp. dianthi. and also its structural determinant role in protein synthesis is blended with mancozeb; inferring either resistance or antagonism occurs with a rise in temperature. Furthermore, FRAC (2009) reported carbamates such as mancozeb targets β-tubulin engaged in mitosis; but also exhibit resistance in many fungal species. Gel profile illustrates expression of resistance proteins expression manifested by overexpression of the 22.9 kDa RHSP and 26.3 kDa HKP with respect to metalaxyl-mancozeb and mancozeb (Fig. 2a).
Mandal and Sinha (1992) reported copper chloride, ferric chlorides, manganese sulfate, were efficient in controlling Fusarium oxysporum f.sp. lycopersici. in accordance with our studies with copper oxychloride. The inability for phenylamides and carbamates derivatives to suppress the 26.3 kDa HKP and the 22.9 kDa RHSP indicates with the current scenario of global warming, farmers may experience further poor yield if the sequel reliance on these fungicides are maintained vis-à-vis the ubiquitous nature of F. oxysporum f.sp. dianthi. in the soil if field temperature is not considered.
The low antifungal potential of metalaxyl-mancozeb blend at 24°C/500 mg kg-1 indicate some degree of antagonism exist blending metalaxyl and mancozeb with regard to F. oxysporum f.sp. dianthi. Contrary to this inhibitory result; lane 2 (Fig. 2a) indicates many proteins were suppressed by the blended fungicide at 24°C/500 mg kg-1; explicitly, inhibitory potential should be higher. This divergence may probably be due to suppression of genes not directly linked to the pathogen vital metabolic processes. Moreover, the induction of the 56.8 kDa resistant protein by this blend, could explain the ambiguity observed. Mancozeb possess manganese and zinc in its structure and copper oxychloride contain copper, indicative transitional metal fungicides under heat-stress may enhance the pathogen vulnerability.
CONCLUSION
Indubitably, F. oxysporum f.sp. dianthi. thermotolerance poses severe threat to tuberous crop production as global temperature increases. Overcoming field resistance and understanding adaptive strategies depends chiefly on how we probe into pathogens interaction with fungicides under heat stress conditions. Temporarily, while relying on fungicides for the time being, a keen prediction of the field temperature will serve as an indicator for the choice of fungicides with respect to the targeted predominant pathogen for a purposeful antifungal activity optimization. The simplicity of copper oxychloride, cost effectiveness, efficiency make’s it suitable for controlling F. oxysporum f.sp. dianthi. under heat stress conditions; irrevocably, lends credence for curbing the farmers cry.
ACKNOWLEDGMENT
This research was funded by the Third World Academy of Sciences (TWAS), Trieste-Italy and the Department of Biotechnology (DBT), New Dehli, India in program FR number: 3240223450. We thank the Division of Plant Pathology-IARI, New Dehli for identifying the pathogen.
REFERENCES
- Kerkeni, A., M. Daami-Remadi, N. Tarchoun and M.B. Khedher, 2008. Effect of bacterial isolates obtained from animal manure compost extracts on the development of Fusarium oxysporum f. sp. radicis-lycopersici. Asian J. Plant Pathol., 2: 15-23.
CrossRefDirect Link - Anjani, K., M.A. Raoof, P.A.V. Reddy and C.H. Rao, 2004. Sources of resistance to major castor (Ricinus communis) diseases. Plant Genet. Resour. Newslett., 137: 46-48.
Direct Link - Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-254.
CrossRefPubMedDirect Link - Amini, J. and D.F. Sidovich, 2010. The effects of fungicides on Fusarium oxysporum f. sp. Lycopersici associated with Fusarium wilt of tomato. J. Plant Prot. Res., 50: 172-178.
CrossRefDirect Link - Khanzada, K.A., M.A. Rajput, G.S. Shah, A.M. Lodhi and F. Mehboob, 2002. Effect of seed dressing fungicides for the control of seedborne mycoflora of wheat. Asian J. Plant Sci., 1: 441-444.
CrossRefDirect Link - Ma, L.J., H.C. van der Does, K.A. Borkovich, J.J. Coleman and M.J. Daboussi et al., 2010. Comparative genomics reveals mobile pathogenicity chromosomes in Fusarium. Nature, 454: 367-373.
CrossRefDirect Link - Alkahtani, M., S.A. Omer, M.A. El-Naggar, E.M. Abdel-Kareem and M.A. Mahmoud, 2011. Pathogenesis-related protein and phytoalexin induction against cucumber powdery mildew by elicitors. Int. J. Plant Pathol., 2: 63-71.
CrossRefDirect Link - Srivastava, S., V.P. Singh, R. Kumar, M. Srivastava, A. Sinha and S. Simon, 2011. In vitro evaluation of carbendazim 50% WP, antagonists and botanicals against Fusarium oxysporum f. sp. psidii associated with rhizosphere soil of Guava. Asian J. Plant Pathol., 5: 46-53.
CrossRefDirect Link - Taskeen-Un-Nisa, A.H. Wani, M.Y. Bhat, S.A. Pala and R.A. Mir, 2011. In vitro inhibitory effect of fungicides and botanicals on mycelial growth and spore germination of Fusarium oxysporum. J. Biopestic., 4: 53-56.
Direct Link








