Research Article
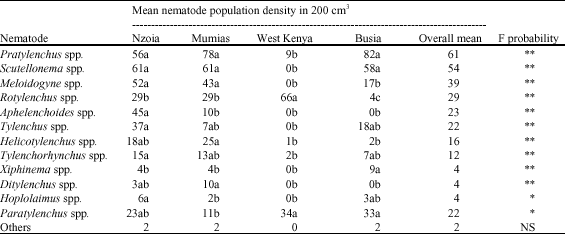
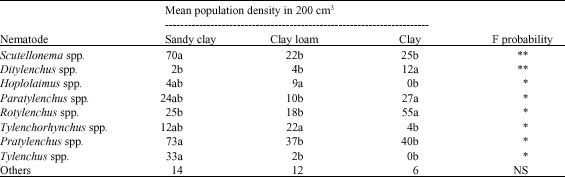
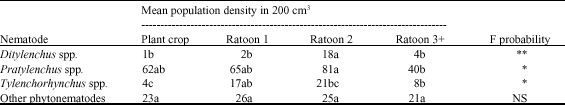
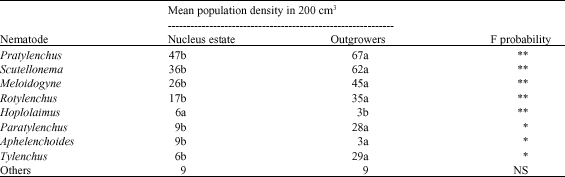
Abundance and Distribution of Plant Parasitic Nematodes Associated With Sugarcane in Western Kenya
Faculty of Agriculture, University of Nairobi, P.O. Box 30197-00100, Nairobi, Kenya
J.W. Kimenju
Faculty of Agriculture, University of Nairobi, P.O. Box 30197-00100, Nairobi, Kenya
F.M. Olubayo
Faculty of Agriculture, University of Nairobi, P.O. Box 30197-00100, Nairobi, Kenya
G.K. Mutua
Faculty of Agriculture, University of Nairobi, P.O. Box 30197-00100, Nairobi, Kenya