Research Article
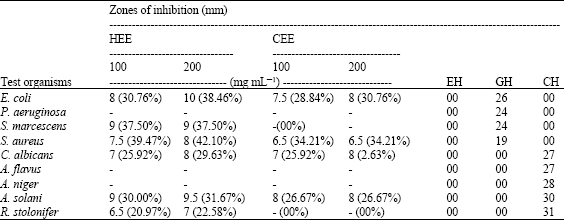
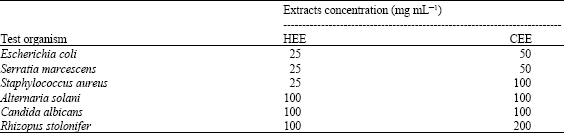
In vitro Study on the Antimicrobial Activity and Phytochemical Analysis of Ethanolic Extracts of the Mesocarp of Voacanga africana
Department of Biology, Federal University of Technology,P.M.B. 1526, Owerri, Nigeria
N.E. Onyedineke
Department of Biology, Federal University of Technology,P.M.B. 1526, Owerri, Nigeria
LiveDNA: 234.745