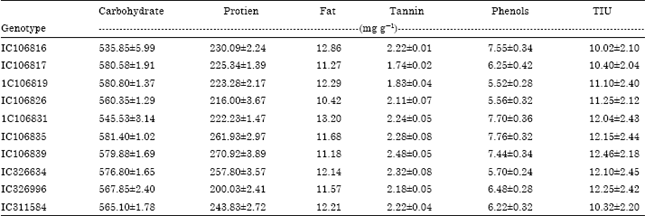
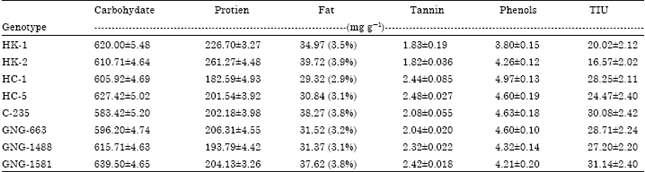
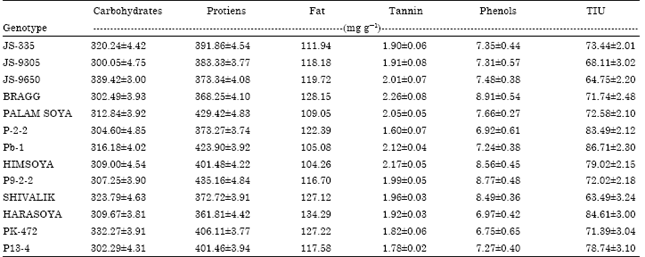
Research Article
Biochemical Basis for Bruchid Resistance in Cowpea, Chickpea and Soybean Genotypes
Parasitology and Entomology Laboratory, Department of Biosciences, Himachal Pradesh University, Shimla, 171005, India
D.R. Thakur
Parasitology and Entomology Laboratory, Department of Biosciences, Himachal Pradesh University, Shimla, 171005, India