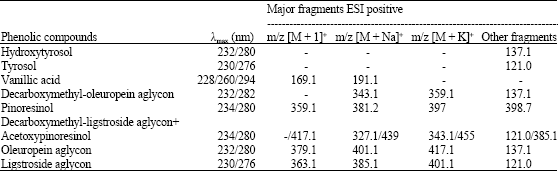
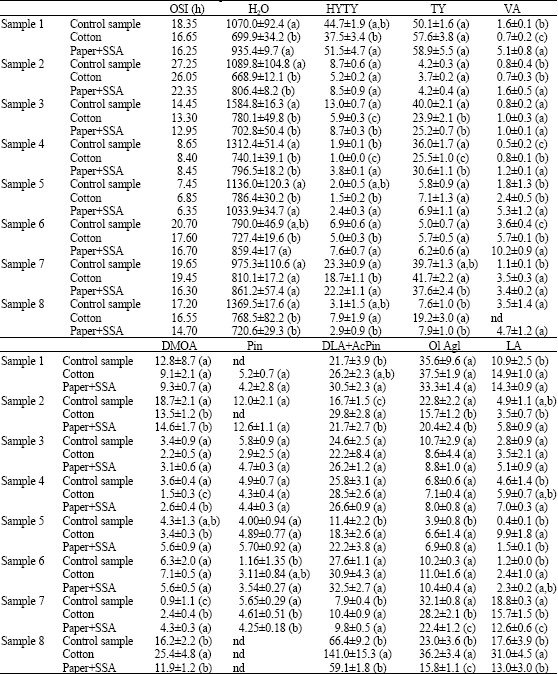
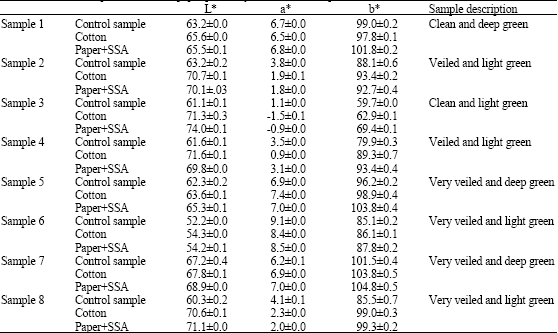
Research Article
Effect of Filtration Systems on the Phenolic Content in Virgin Olive Oil by HPLC-DAD-MSD
Department of Analytical Chemistry, Faculty of Sciences, University of Granada, C/Fuentenueva s/n, E-18071 Granada, Spain
L. Cerretani
Department of Food Science, University of Bologna, P.zza Goidanich 60, 47023 Cesena (FC), Italy
A. Bendini
Department of Food Science, University of Bologna, P.zza Goidanich 60, 47023 Cesena (FC), Italy
A. Segura-Carretero
Department of Analytical Chemistry, Faculty of Sciences, University of Granada, C/Fuentenueva s/n, E-18071 Granada, Spain
A. Fernandez-Gutierrez
Department of Analytical Chemistry, Faculty of Sciences, University of Granada, C/Fuentenueva s/n, E-18071 Granada, Spain
G. Lercker
Department of Food Science, University of Bologna, P.zza Goidanich 60, 47023 Cesena (FC), Italy