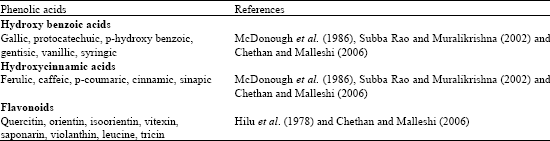
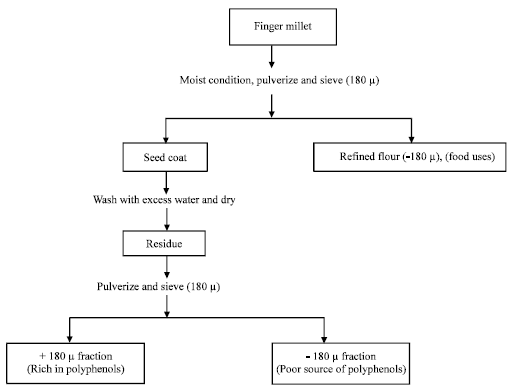
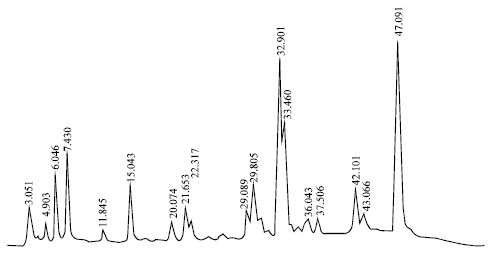
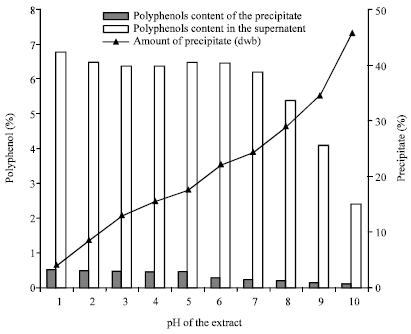
Research Article
Finger Millet Polyphenols: Characterization and their Nutraceutical Potential
Department of Grain Science and Technology, Central Food Technological Research Institute, Mysore 570 020, India
N.G. Malleshi
Department of Grain Science and Technology, Central Food Technological Research Institute, Mysore 570 020, India