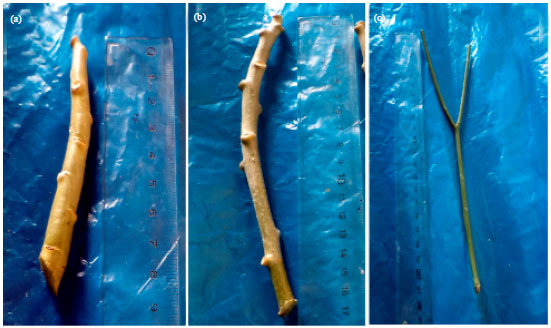
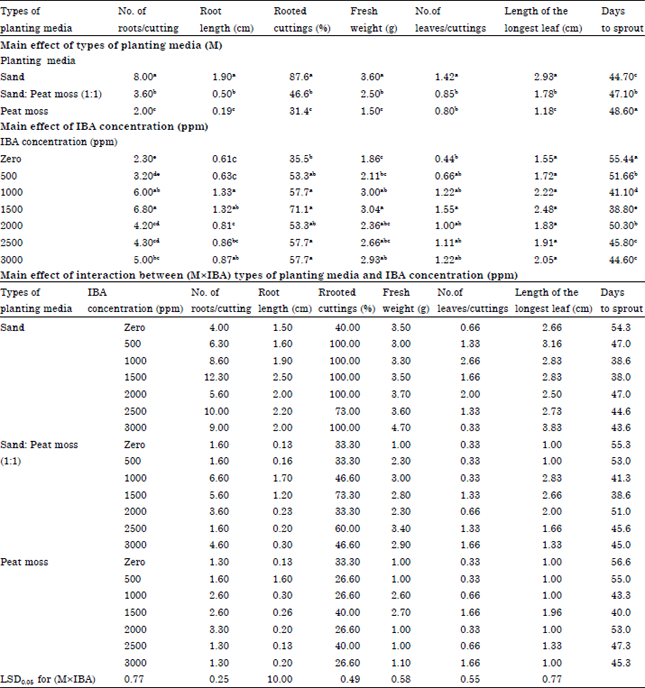
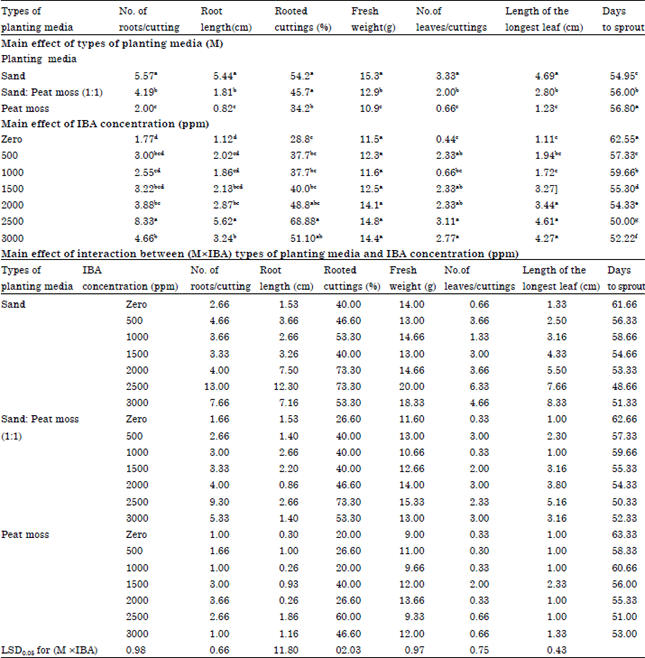
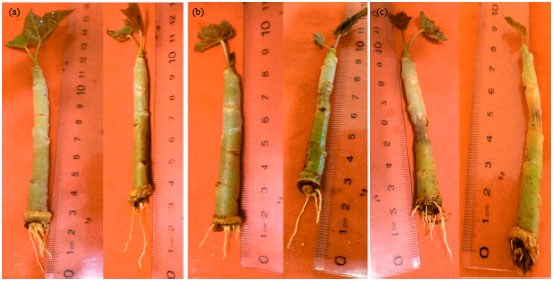
Research Article
Rooting of Jatropha curcas L. and Euphorbia tirucalli Cuttings in Response to IBA and Planting Media in North Egypt: A Potential Source for Tomorrow’s Oil, Biodiesel and Biofuels
Department of Horticulture, Faculty of Agriculture, Damanhour University, Egypt
Hany M. El-Naggar
Department of Floriculture, Faculty of Agriculture, Alexandria University, Egypt