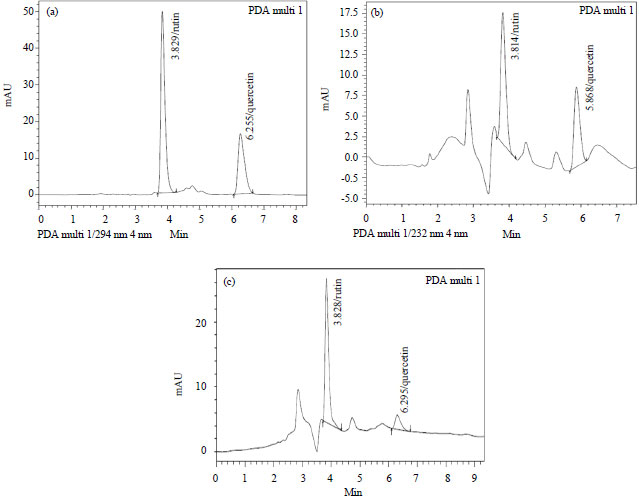
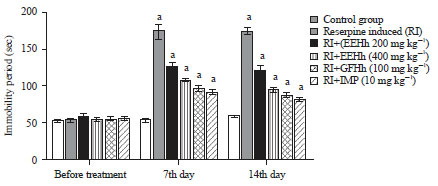
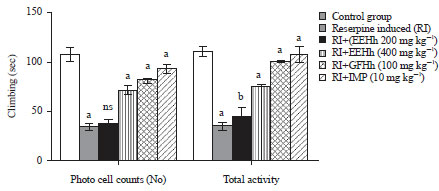
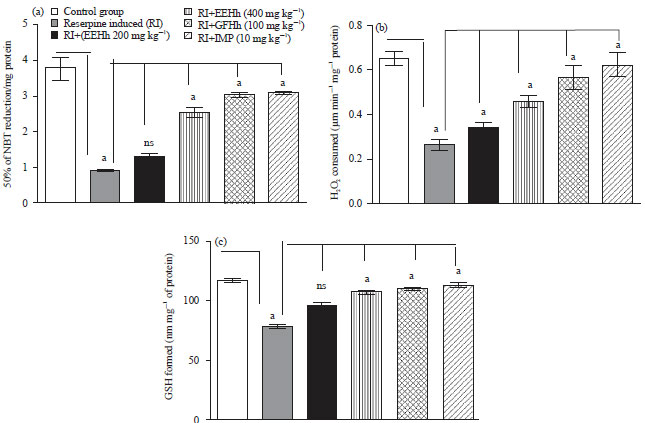
Research Article
Evaluation of Antidepressant like Effects of Ethanolic Hypericum hookerianum and its Glycosidic Flavonoid Enriched Extract in Reserpine Induced Swiss Albino Mice
Department of Biotechnology, School of Biotechnology and Health Sciences, Karunya University, Coimbatore, 641114, Tamil Nadu, India
S. Murugan
Department of Biotechnology, School of Biotechnology and Health Sciences, Karunya University, Coimbatore, 641114, Tamil Nadu, India
LiveDNA: 91.11728
P. Uma Devi
Department of Biology, School of Natural Science, Madawalabu University, Ethiopia