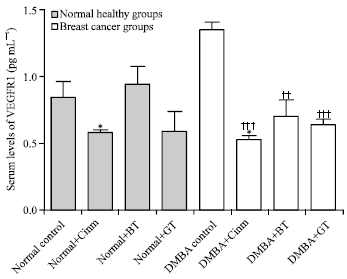
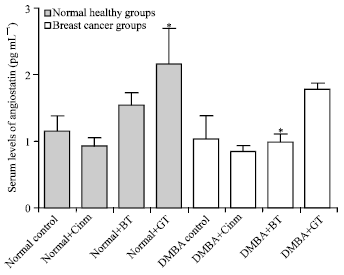
Research Article
Antiangiogenic Activities of Cinnamon, Black and Green Tea Extracts on Experimentally Induced Breast Cancer in Rats
Department of Biochemistry, Faculty of Pharmacy, Al-Azhar University, Assiut, Egypt
Sherif W. Mansour
Department of Physiology, Biomedical Science, College of Clinical Pharmacy, King Faisal University, Al-Hofuf, Kingdom of Saudi Arabia
Mueen A. Khalil
Department of Parmacogonosy, Pharmaceutical Science, College of Clinical Pharmacy, King Faisal University, Al-Hofuf, Kingdom of Saudi Arabia