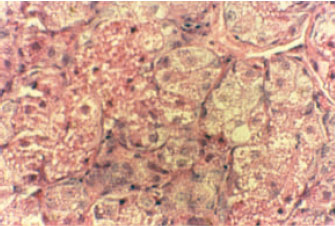
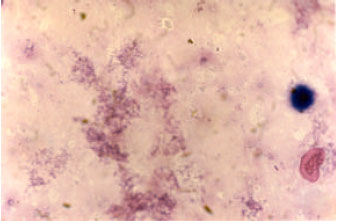
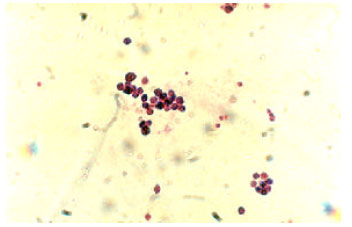
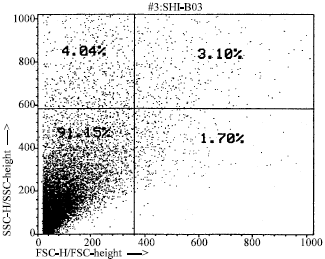
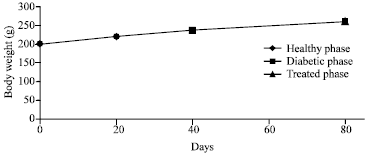
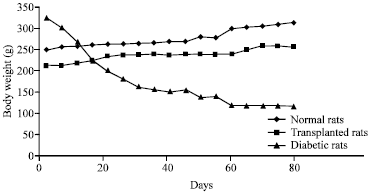
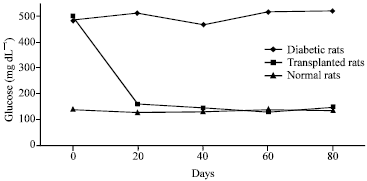
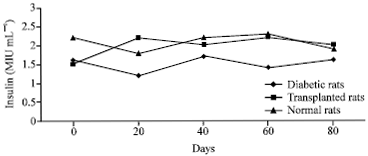
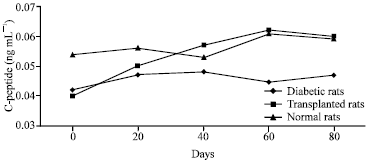
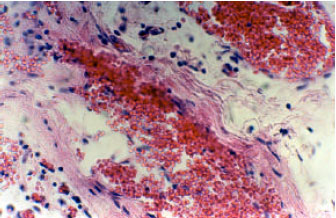
Research Article
Treatment of Streptozotocine Induced Diabetes Mellitus in Male Rats by Immunoisolated Transplantation of Purified Langerhans Islet Cells
Department of Pilot Biotechnology of Pasteur Institute of Iran, Tehran, Iran
LiveDNA: 98.4507
D. Noruzian
Department of Pilot Biotechnology of Pasteur Institute of Iran, Tehran, Iran
SH. Jamshidi
I.B.B. University of Tehran, Iran
A. Farhangi
Department of Pilot Biotechnology of Pasteur Institute of Iran, Tehran, Iran
M.R. Mehrabi
Department of Pilot Biotechnology of Pasteur Institute of Iran, Tehran, Iran
B. Lame Rad
Biochemistry Department of Alzahra University, Tehran, Iran
M. Mofidian
S.P.I. Institute, Karaj, Iran
A. Allahverdi
Department of Pilot Biotechnology of Pasteur Institute of Iran, Tehran, Iran