Research Article
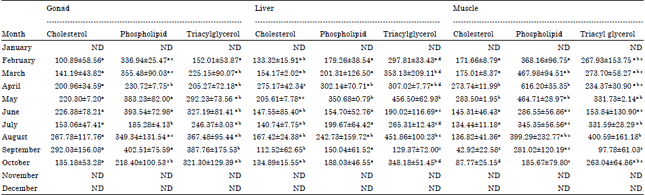
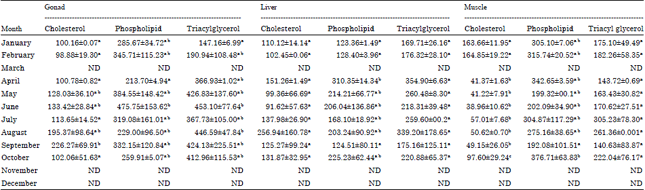
Lipid Changes in Relation to Maturation and Spawning of Tropical Double Spotted Queenfish, Scomberoides lysan (Forsskål, 1775)
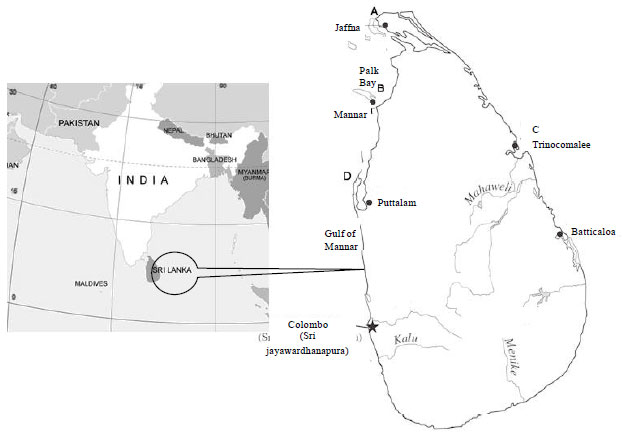
Department of Fisheries, University of Jaffna, Jaffna, Sri Lanka
K. Sivashanthini
Department of Zoology, University of Jaffna, Jaffna, Sri Lanka
W.S. Thulasitha
Department of Zoology, University of Jaffna, Jaffna, Sri Lanka