Research Article
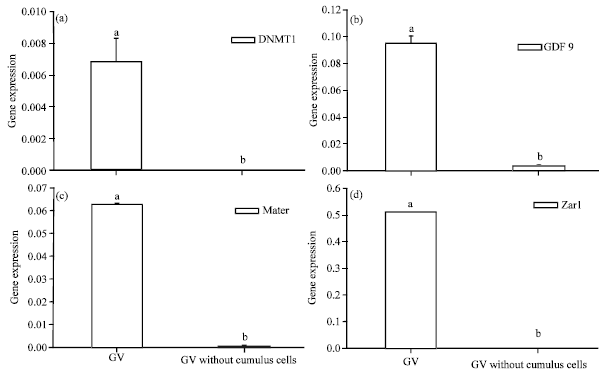
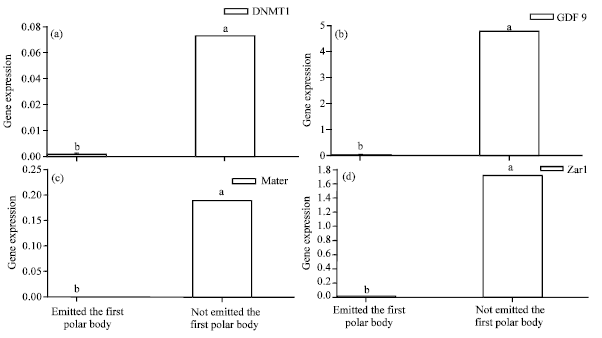
Relations Between GDF9, Zar1, Mater and DNMT1 Gene Relative mRNA Abundance and Oocytes Quality of Ovine Oocytes
The Key Lab of Animal Biotechnology of Xinjiang, Urumqi, People�s Republic of China
Liqin Wang
The Key Lab of Animal Biotechnology of Xinjiang, Urumqi, People�s Republic of China
Yuncheng Zhao
The Key Lab of Animal Biotechnology of Xinjiang, Urumqi, People�s Republic of China
Min Hou
The Key Lab of Animal Biotechnology of Xinjiang, Urumqi, People�s Republic of China
Jing Wang
The Key Lab of Animal Biotechnology of Xinjiang, Urumqi, People�s Republic of China
Juncheng Huang
The Key Lab of Animal Biotechnology of Xinjiang, Urumqi, People�s Republic of China