Research Article
Fecundity and Temporal Reproductive Cycle of Four Finger Threadfin (Eleutheronema tetradactylum) in Malaysian Coastal Water
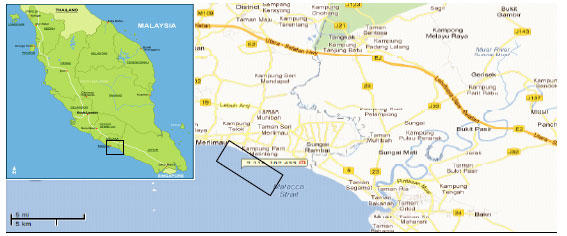
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 UKM, Bangi, Selangor, D.E., Malaysia
A. Samat
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 UKM, Bangi, Selangor, D.E., Malaysia
C.C. Zaidi
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 UKM, Bangi, Selangor, D.E., Malaysia
A.G. Mazlan
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 UKM, Bangi, Selangor, D.E., Malaysia
Gazi Mahabubul Alam
Academic Performance Enhancement Unit, University of Malaya, Jalan Panati Baharu, 59990 Kuala Lumpur, Malaysia
Abul Quasem Al-Amin
Faculty of Economic and Administration, University of Malaya, 50603 Kuala Lumpur, Malaysia
K.D. Simon
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 UKM, Bangi, Selangor, D.E., Malaysia