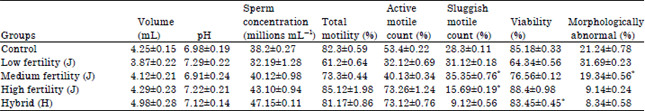
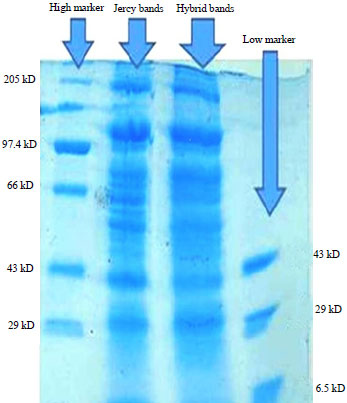
Research Article
Analysis of Seminal Plasma Proteins of South Indian Jersey and Hybrid Bulls and their Correlation with Semen Quality
School of Biosciences and Technology, VIT University, Vellore, 632014, Tamil Nadu, India
LiveDNA: 91.13005
V. Devi Rajeswari
School of Biosciences and Technology, VIT University, Vellore, 632014, Tamil Nadu, India
LiveDNA: 91.11193
M. Ramesh Pathy
School of Biosciences and Technology, VIT University, Vellore, 632014, Tamil Nadu, India
T.B. Sridharan
School of Biosciences and Technology, VIT University, Vellore, 632014, Tamil Nadu, India