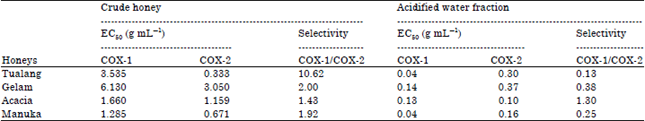
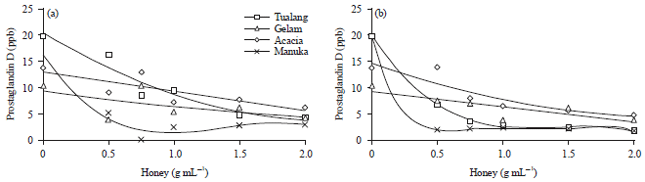
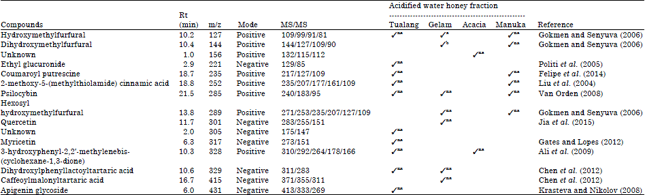
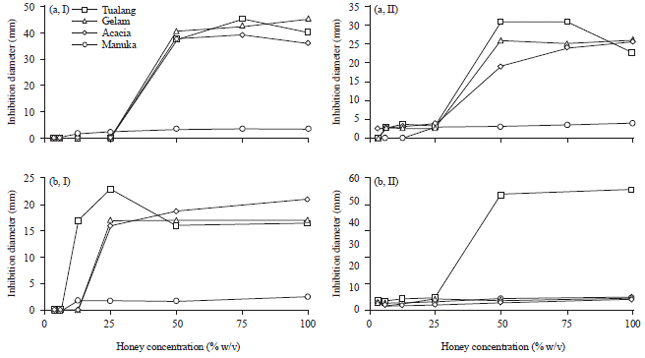
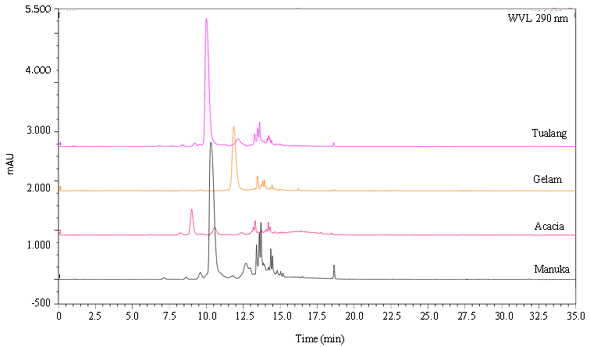
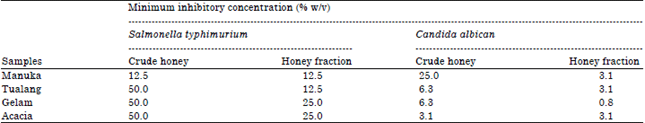
Research Article
Anti-Inflammatory and Anti-Microbial Activities of Selected Honey Samples
Department of Bioprocess Engineering, Faculty of Chemical Engineering, Metabolites Profiling Laboratory, Institute of Bioproduct Development, Universiti Teknologi Malaysia, 81310, UTM Skudai, Johor Bahru, Johor, Malaysia
Nurul Izzati Mohd Ismail
Department of Bioprocess Engineering, Faculty of Chemical Engineering, Metabolites Profiling Laboratory, Institute of Bioproduct Development, Universiti Teknologi Malaysia, 81310, UTM Skudai, Johor Bahru, Johor, Malaysia