ABSTRACT
Tannery effluent has been associated with both organic and inorganic pollutants worldwide. The most critical area of interest within the tanning industry is the effluent treatment pathway. Indeed the anaerobic lagoons have traditionally been associated as a critical phase of such effluent treatments. For that matter pollutant load and toxicity levels of five interconnected anaerobic lagoons were determined using both chemical and bioassay techniques. The results indicated that high organic and inorganic pollutants were observed. Moreover depletion of oxygen levels resulted to increased anaerobism which induced the conversion of sulphates to the highly toxic sulphide levels as determined by the biosensors. Thus efficacy of anaerobic lagoons is dependent to the chemistry of the effluent and pre-treatment of the waste. However the use of indigenous organisms as biosensor should in future be used to complement the techniques used in this study. This suggested approach will enable the tenets of adaptability and acquired resistance overtime be established.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/rjet.2007.167.175
URL: https://scialert.net/abstract/?doi=rjet.2007.167.175
INTRODUCTION
Pollution from the leather processing industries which is deemed as the largest polluter in the world (Khwaja, 1998) also has a negative long term impact on the economic growth potential of a country irrespective of the immediate profit accruals intended (Mwinyihija and Killham, 2006). Tanning is a major polluter worldwide and tannery wastewater, in particular, is a potential environmental pollutant (Ros and Ganter, 1998; Mwinyihija et al., 2006). It can cause a serious environmental impact on water systems with its high oxygen demand, discolouration and toxic chemical constituents (Song et al., 2000; Mwinyihija et al., 2005a).
Therefore the major public concern over tanneries has traditionally been about odour and water pollution from untreated or poorly treated discharges. Important pollutants associated with the tanning industry include chlorides, tannins, chromium, sulphate and sulphides as addition to trace organic chemicals and increasing use of synthetic chemicals such as pesticides, dyes and finishing agents, as well as from the use of newer processing chemical solvents (Anonymous, 2003). These substances are frequently toxic and persistent and affect both human health and the environment (Anonymous, 1994; Mwinyihija et al., 2005a).
This contamination can interfere with subsequent land use (agriculture, building) as well as potentially contribute to groundwater pollution. Groundwater contamination occurs when wastewater and chemicals seep through the soil from unlined ponds, pipes and drains, or from dumps and spills. In addition to deposition of solids, raw unsettled tannery wastewaters can cause encrustation (of calcium carbonate) and serious corrosion of metals as well as concrete sewers due to H2S biological oxidation to H2SO4 (Balusubramanian and Pugalenthi, 2000). Cleaning up of such environmental impacts will require expenditure of funds, which could have promoted positive and sustainable development (Mwinyihija and Killham, 2006).
The complex nature of waste emanating form the tanning industry has generally resulted for this industry to be considered by far as the largest source of hazardous wastes. However, to date there are no reliable estimates of the quantity and types of hazardous waste generated in most developing countries. Approximately 10-15% of the wastes produced by the industry overall are likely to be hazardous, increasing at a rate of 2-5% per year (Chaaban, 2001).
Because the tannery wastewater contains a complexity of pollutants including chromium and chlorinated phenols, it is vital to comprehend the toxic nature of such wastewater to further understand its impacts and identify both environmental managemental aspects and potential remediation strategies. Tannery wastewater is generally treated by various physico-chemical or biological methods (includes the anaerobic lagoons) and by a combination of both techniques (Reemste and Jekel, 1997). Physical and chemical processes are frequently employed to treat contaminated sites including application of ameliorants, but often do not destroy the contaminants (Bouwer et al., 1994; Gatima et al., 2006).
The aim of this study was therefore to use chemical techniques and bioassay to assess and determine the efficacy of the anaerobic lagoons in reducing the toxic nature of effluents from a tanning industry site in Kenya. The effluent at the study site was estimated to take six days in each lagoon before being discharged.
MATERIALS AND METHODS
All areas of the study site were marked using a twelve channel GPS (eTrexTM) personal navigator, part number 190-00205-00 Rev. B (Sept. 2000) GARMIN International Inc. USA). This was carried out to obtain waypoints and altitude at identified sampling points. Sediments and effluent samples from the anaerobic lagoons were collected in 20 mL super polyethylene vials (in triplicates every sampling date) in a tannery site in Kenya (2004/05 period). Bottles were filled to the brim and carefully capped without air bubbles. During storage and transportation from the field, they were stored in the dark at 4°C and tested within four weeks of sampling.
Preparation of Sediment and Effluent Samples
Sediment extraction was performed using a method adapted from that described by Mathews and Hastings (1987). Samples (10 g) of sediment were weighed into centrifuge tubes and 20 mL aliquots of deionised water (Millipore-Q, Quantum™ EX ultra pure organex cartridge) added (in triplicate). The pH of the samples was measured using a portable, standard, pH electrode (Hanna HI 8424, Norlab Instrument Ltd., Aberdeen, UK). The samples were placed in an end-over-end shaker for 30 min and then centrifuged at 1100 g for 15 min. Parallel extractions for toxicity determination were carried out in triplicate and samples taken from the supernatants immediately prior to the bioassay. All chemicals used were of analytical-reagent grade and all test solutions were prepared using double deionised water.
Bioassay
Lux-marked bacterial biosensors were used during the study and the preparation of the biosensor and luminometer measurements were carried out as described. One hundred microliter of the resuscitated biosensor suspension was added to the samples at 15 sec intervals, accurately timed for measurement in the Bio Orbit 1253 luminometer (Labtech International, Uckfield, UK). Each sample was exposed to the sensor for exactly the same time. Samples were incubated for 15 min before light output measurements were carried out at 15 sec intervals. This ensured the same exposure time to the potentially toxic elements for cells in each of the cuvettes.
Sample Digestion (Sediments Samples)
Aliquots (2.5 mL) of concentrated HNO3 (69% Analar grade) were added to 200 mg dried sediments samples obtained from the anaerobic lagoons, which had been weighed into 75 mL digestion tubes. The mixture was then allowed to stand overnight at 15°C. The following day, the digestion tubes were placed on a heating block and the temperature was gradually raised to 100°C for 8 h. The samples were allowed to digest for 3 h, after which the volume was reduced to 3-4 mL. The digest was cooled at room temperature and diluted to 10 mL with double deionised water in graduated tubes. Total concentrations of Pb, Cu, Zn, Fe, Ni, Cd and Cr were determined using atomic absorption spectrometry (Perkin Elmer Analyst 100).
Chemical Analysis
A total concentration of Cr, Ni, Cu, Zn, Cd and Fe in each tannery effluent sample was determined (acidified with 1% HNO3) by Atomic Absorption spectrometry (Perkins Elmer Analyst 100).
Biological Oxygen Demand (BOD)/Dissolved Oxygen (DO) Determination
Levels of BOD and DO were determined using standard protocols (APHA, 1965) and carried out in an accredited laboratory (Inveresk Laboratory, Scotland). Samples of water were incubated at 20°C for 5 days in a dark water bath. Every day for five days, DO was determined. The difference between initial value and the value at each time (t) period (i.e., oxygen demand) was plotted as the BODt (mg L-1).
Samples for the determination of BOD and DO were tested within 10 days of collection to avoid degradation. The protocol included adding 1.0 mL of manganous sulphate reagent followed immediately by 1.0 mL of alkaline-iodide-azide solution to the BOD bottle (300 mL). The bottle was restoppered immediately and the contents mixed by shaking vigorously for at least 20 sec or until the precipitated manganous and manganic hydroxide is evenly dispersed. After 2 to 3 min of shaking again, the precipitate in the sample was allowed to settle for 1 h. By means of a two-way pipette and vacuum system, 100 mL of solution was transferred from the BOD bottle to a specially-painted Erlenmeyer flask containing a magnetic stirring bar. Titration was carried out immediately with thiosulphate solution until the solution turned to pale straw colour. Four drops of starch solution was added. Titration was continued until the blue colour disappeared. The dissolved oxygen was calculated using normality and volume of sodium thiosulphate with BOD values obtained as explained earlier.
Sulphate and Chloride Determination
Appropriate standards for the determination of sulphate and chloride using ion exchange chromatography (Dionex, series 4500i-Autosampler AS40) were prepared in various concentration ranges (0, 2, 5 and 10 mg L-1) and a calibration curve obtained. For sulphate determination, a stock solution of 1000 mg L-1 SO4 was prepared by dissolving 1.818 g of potassium sulphate (K2SO4) in 1 L of deionised water. A chloride stock standard solution of 1000 mg L-1 Cl was prepared by dissolving 1.648 g of sodium chloride (NaCl) in 1 L of deionised water. The eluent was prepared by weighing out 0.95 g of Na2CO3 and 0.71 g of NaHCO3. The preparation was then dissolved and made up to 5 L with deionised water. The regenerant was prepared by adding 3.5 mL of concentrated H2SO4 to approximately 300 mL of deionised water and making up to 5 L with deionised water. The following instrumentation conditions were maintained:
| • | Eluent |
| Eluent flow rate | : | 1.2 mL min-1 |
| Suppressor | : | H2SO4 |
| Background conductivity | : | 17.3 μS |
| • | Analyte |
| Analyte flow rate | : | 1.2 mL min-1 |
| Temperature compensation | : | Selectable compensation between 0.0 and 3.0% per 1.7°C |
| Pressure | : | 2900 psi |
| Limit | : | 5000 psi |
Total Phenols
A spectrophotometric determination of phenols based on a multicommuted flow system with a 100 cm optical path flow cell was used to determine total phenols. All solutions were prepared with distilled and deionised water and used analytical grade chemicals. Phenol reference solutions within 10.0 and 100 g L-1 were prepared by appropriate dilutions of a 1.00 g L-1 stock solution. Reagent R1 was prepared by dissolving 50.0 mg of 4-aminoantipyrine (4-AAP) in 50 mL of a buffer containing 5.2 g L-1 NaHCO3, 5.8 g L-1 H3BO3 and 6.2 g L-1 KOH (pH 10.0). Reagent R2 was a 0.20% m/v K3[Fe(CN)6] solution prepared in water. Water was used as the carrier. The analysis was carried out by an accredited laboratory (Robertson Laboratories, UK).
| Vi | = | Three-way solenoid valves. |
| B | = | Reaction coil (80 cm). |
| D | = | Long pathlength flow cell (100 cm optical path). |
| C | = | Water carrier (5.4 mL min-1). |
| S | = | Sample (6.0 mL min-1). |
| R1 | = | 0.10% (m/v) 4-AAP buffered at pH 10.0 (0.8 mL min-1). |
| R2 | = | 0.2% (m/v) K3[Fe(CN)6] (0.6 mL min-1). |
| W | = | Waste. |
Solutions containing 7.0 mol L-1 phenol, m-cresol, p-cresol, p-chlorophenol, catechol, hydroquinone, p-aminophenol and p-nitrophenol were employed for evaluation of the relative response for different phenols.
These data were then collated and analysed using Microsoft Excel. Statistical analysis involved two way analysis of variance (ANOVA) and treatment effects determined by Least Significant Difference (LSD).
RESULTS AND DISCUSSION
Generally, before any manipulations were carried out (untreated samples), high toxicity (p≤0.05) was observed for the samples obtained from the effluent treatment pits (Table 1) leading to the anaerobic lagoons. Tannery samples from the beam house, chrome stripping and general sedimentation pit showed extreme toxicity. Results from the different effluent treatment pits were all significantly different (p≤0.05). This observation was similar for the analyzed samples obtained from the anaerobic lagoons (except for lagoon 3) (Table 2). When the organic matter contents was compared to the bioluminescence values, a reduction in the value of the biosensor (indicating increased toxicity) similarly demonstrated a general reduction in the organic matter content as indicated in Table 2. Studies elsewhere suggested that strong binding of the pollutants (e.g., Chromium, Mecury etc.) capability towards humic acid (a major component of the organic matter content) is most probably due to sulfhydryl and thiophenolic groups (Miretzky et al., 2005). Therefore, the presence of natural humic substances in sediments reduces the availability (thus the toxicity) of the pollutants leached from the sediments to the aquatic systems. The environmental dynamic mechanism could involve both complexation and reduction and most probably occurring simultaneously. The overall effect of this is the reduction of the sediment strength in the affected lagoons as shown in Table 2. Moreover Schumacher (2002) reported that the quality of organic matter in sediments is critical to the partitioning and bioavailability of sediments-associated contaminants. Specifically Talley et al. (2002) found out that PAH’s clay/silt sediment fraction were more mobile and available and thus posed a great concern.
| Table 1: | Percentage maximum bioluminescence of effluent treatment pits |
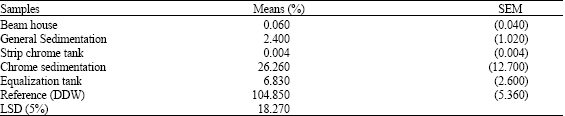 | |
| DDW: Double Deionised Water, n = 9 | |
| Table 2: | Percentage maximum bioluminescence, Total Organic Compounds (TOC) and Organic Matter (OM) of anaerobic effluent treatment lagoons |
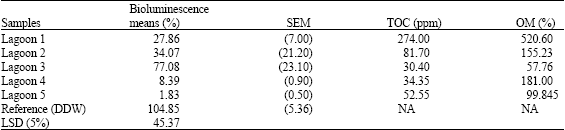 | |
| DDW: Double Deionised Water, NA: Not Available, n = 9 | |
Bioassay
Biosensors have been used to monitor environmental contamination (Steinberg et al., 1995; van de Lelie et al., 1994; Mwinyihija et al., 2005b, 2006) by heavy metals (Paton, 1995) and organic contaminants, as well as toxicity in soils and water contaminated by industrial effluents (Brown et al., 1996), BTEX (benzene, toluene, ethlybenzene, xylene) compounds (Sousa et al., 1998) and chlorinated aromatics (Steiert and Crawford, 1985). Toxicity is assessed by examining a decrease in light output from the bacterium when it is exposed to environmental samples or chemicals (Steinberg et al., 1995). The reduction (due to a toxic substance) in light output is proportional to the bioavailable concentration present (Sousa et al., 1998). Therefore, maximum percentage bioluminescence for all the analysed samples calculated against a blank of double deionised water at pH 5.5. The choice of the lux-marked biosensor in this work offered great environmental relevance in determining the bioavailability of various groups of the pollutants and the toxic nature of the effluent from the Kenyan tanning industry. Samples from the effluent treatment pits (Table 1) and anaerobic lagoons (lagoons 1, 2 and 3) (Table 2) were associated with this type of toxicity. The increase of luminescence in this study suggested that considerable toxicity was caused by volatile organics. However lagoons 3 and 4 indicated extreme toxicity with bioluminescence values of 8.39 and 1.83%, respectively. Available metals are generally in the form of soluble cations and their tendency to be present in ionic form increases with increasing acidity (Sposito, 1989). In addition the high toxicity values could possibly be linked with the increased sulphide levels (Table 4). The toxicity relationship with the organic matter content and potential impact has been discussed earlier. Sarin (2000) reported the toxicity response of lux-marked E. coli HB101 to a range of metals thus its suitability in the present study.
Toxicity in samples such as treatment effluents, anaerobic lagoons and downstream riverine sampling points (Table 1 and 2) was attributed to high concentrations of chromium and phenols (Table 3). For example, although in tannery wastewater Cr3+ is the most expected Cr form, the Redox reactions occurring in the sludge can increase the concentration of the hexavalent form (Kotaś and Stascika, 2000). Most metals show increased solubility with decreased pH (Artiola, 1996), indicating increased bioavailability (chemical assimilation and possible toxicity) of organic/inorganic compounds (Steinberg et al., 1995; Shaw et al., 2000; Alexander, 2000). Under slightly acidic or neutral pH conditions in this type of wastewater, the poorly soluble Cr(OH)3 aq should be the preferred form, but a high content of organic matter originating from the hide material processing is effective in forming soluble organic Cr3+ complexes (Stein and Schwedt, 1994; Walsh and O’Halloran, 1996a, b).
| Table 3: | Total phenols, pH and metal concentrations of tannery effluent treatment pits (mg L-1), anaerobic lagoons (mg L-1) and riverine sediments (mg kg-1) (Mwinyihija et al., 2005a) |
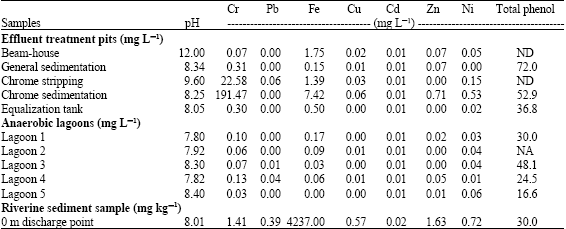 | |
| The Limit of Detection (LOD) was determined on the basis of five blank samples at average blank signal plus three and ten times the standard deviation, NA: Not Available, ND: None Detected | |
Other related studies investigating the fractionation of chromium toxicity in water using E. coli HB101 pUCD607 showed that speciation of chromium at different pH levels and a synergistic effect with other metals (e.g., copper and zinc) contributed to its toxicity (Wararatananurak, 2000). This observation suggested that chromium is frequently a constraint to bioremediation in contaminated environments (Killham, 2006. Personal Communication).
The choice of the organism (E. coli HB101 pUCD607) was suitable for the study as it is found in a variety of habitats including sewage sludge and contaminated river water and it functions over a wide pH range. However, the future use of indigenous organisms as biosensors should complement this study and enable exploration of adaptability and acquired pollutant resistance over time to be addressed.
Chemical Analysis
Total phenols were present at elevated concentrations within most of the effluent treatment pits, lagoons and at the discharge point. However, a gradual decrease in phenol concentration was observed downstream (Table 3). The samples with the highest total phenols were the general sedimentation, chrome sedimentation, equalisation tank, lagoons 1 and 3 and the discharge point at the river (>30 mg L-1). The presence of high total phenols within the sampled areas was demonstrated by increased (except lagoon 3) bioluminescence (Table 2). Increase in bioluminescence is related to the uncoupling of the proton gradient (release of the fatty acid substrate for the luciferase enzyme) by chlorinated phenols, causing an increase in electron transfer rate and therefore an increase in the respiration rate, resulting in an increase in light output.
To ascertain the impact of the pollutants all the treatment phases before the anaerobic lagoons were determined. Indeed metals such as chromium and iron showed high concentrations as shown in Table 3. Chromium stripping (22.58 mg L-1) and chrome sedimentation (191.47 mg L-1) samples were, not surprisingly, associated with high chromium concentrations, hence linked to decreased luminescence of the sensor (Table 3) calculated against a blank of double deionised water at pH 5.5. When Table 3 was further scrutinized and the effluent treatment pits compared to the anaerobic lagoons, chromium unlike the other parameters was mostly affected. The chrome stripping (22.58 mg L-1) and chrome sedimentation (191.47 mg L-1) pits demonstrated high concentration values whereas within the anaerobic lagoons very low concentration levels (≤0.13 mg L-1) were observed. On the other hand total phenol did not necessarily show any difference between effluent treatment pits and the anaerobic lagoons (the pH values were at most >7.8).
| Table 4: | Results depicting analysis of various ecotoxicological parameters related to the three main stages of tannery waste water treatment flow; raw effluent, sedimentation tank and final effluent at the discharge point (mg L-1) (n = 3) |
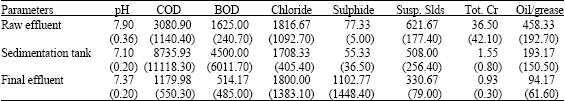 | |
| Susp. Slds: Suspended solids, Values in parentheses are SEM (n = 9) | |
This could be attributed to the behavior of chlorinated phenols where in conditions of ambient pH constant pKa, chlorophenols can donate protons to form phenolate ions (Schellenberg et al., 1984). Therefore the effect is based on the tendency of donating the protons to increase along with increasing chlorination of the benzene ring. Thus the phenolate form is more water soluble than its non-ionized counter part (Lyytikäinen, 2004). Furthermore the tendency of phenolate ions to sorb on soil or sediment is 15-30 times lower compared to their conjugated acids (Lagas, 1988). This overall result (Table 3) was critical in understanding the role of indigenous and habitat microorganism in breaking down of metals and organic contaminants within the anaerobic lagoons sediment matrices.
In Table 4 results showed that all the investigated parameters were quite erratic as depicted through the standard errors of the mean most probably due to the different types of cured raw material and diverse processing regimes. In particular, the Sulphide content increased (from 77.33±5.0 to 1102.77±1448.4 mg L-1) sequentially through the effluent treatment pits as BOD levels reduced (except for the sedimentation pits), likewise demonstrating a depletion of oxygen levels within the treatment pits. In addition the depletion of dissolved oxygen encourages anaerobic activity, which leads to release of noxious gases (Pepper et al., 1996).
Sulphates are reversed to sulphides through reduction by anaerobic bacteria thus increased anaerobic conditions with in the lagoons resulted to an increase in Sulphide contents (Table 4). This observation showing increases in sulphide concentration in correlation to increased anaerobic conditions has been observed in earlier studies by Balusubramanian and Pugalenthi (2000). However sulphide is not only toxic for higher organisms, it is also known as an inhibiting substance in anaerobic microbial processes (Wiemann et al., 1998).
CONCLUSIONS
It is important to note that anaerobic lagoons cause sedimentation of suspended solids, partial conversion of COD to methane gas, conversion of sulphides into sulphates and production of sludge. In retrospect, it was demonstrated in this study that depleted levels of oxygen occurred due to biological decomposition of organic matter and other tannery waste, imparting anaerobic conditions observed. Thus the study demonstrated that anaerobic lagoons by themselves have very little impact and could be the potential source of Hygrogen sulphide re-generation and methylation. The lagoons efficacy can only be assured if the waste is pretreated effectively and the passage of effluent doesn’t exceed more than 6 days in moving from one pit to another to avert anaerobic conditions setting in. Furthermore the study successfully demonstrated the potential of lux-marked biosensors to determine toxicity of tannery effluent. Indeed appropriate chemical analysis was used to further confirm the biosensor-based identification of 'cause and effect' of toxicity and potential constraints to remediation. The success of identifying the primary contaminants within the anaerobic lagoons prompts the need to further investigate the impact of effluent emanating from the tanning industry to either terrestrial or aquatic ecosystem when discharged or the dynamics of methylation within the anaerobic lagoons.
REFERENCES
- Alexander, M., 2000. Ageing, bioavailability and overestimation of risk from environmental pollutants: Critical review. Environ. Toxicol. Chem., 34: 4259-4265.
Direct Link - Balusubramanian, S. and V. Pugalenthi, 2000. A comparative study of the determination of sulphide in tannery wastewater by Ion Selective Electrode (ISE) and iodimetry. Water Res., 34: 4201-4206.
Direct Link - Bouwer, E., N. Durant, L. Wilson, W. Zhang and A. Cunningham, 1994. Degradation of xenobiotic compounds in situ: Capabilities and limits. FEMS Microbiol. Rev., 15: 307-317.
Direct Link - Brown, J.S., E.A.S. Rattray, G.I. Paton, G. Reid, I. Caffoor and K. Killham, 1996. Comparative assessment of the toxicity of a paper mill effluent by respiratory and luminescence based bacteria assay. Chemosphere, 32: 1553-1561.
CrossRefDirect Link - Chaaban, M.A., 2001. Hazardous waste source reduction in materials and processing technologies. J. Mater. Process. Technol., 119: 336-343.
Direct Link - Gatima, E., M. Mwinyihija and K. Killham, 2006. Determination of adsorption efficiency based on cation exchange capacity related to red earth, bone meal and pulverized fly ash as ameliorants to lead contaminated soils. Int. J. Environ. Sci. Technol., 3: 269-280.
Direct Link - Kotas, J. and Z. Stasicka, 2000. Chromium occurrence in the environment and methods of its speciation. Environ. Pollut., 107: 263-283.
CrossRefDirect Link - Matthews, J.E. and L. Hastings, 1987. Evaluation of toxicity test procedure for screening treatability potential of waste in soil. Toxicol. Assess., 2: 265-281.
CrossRef - Miretzky, P., W.F. Jardim, C.M. Bisnoti and C.J. Rocha, 2005. Factors affecting Hg(II) adsorption in soils from the Rio Negro basin (Amazon). Quim. Nova, 28: 438-443.
Direct Link - Mwinyihija, M. and K. Killham, 2006. Is the Kenyan Tanning industry integral to prioritized environmental sustainability targets set in the quest to industrialization by 2020?. Environ. Sci., 3: 113-134.
CrossRef - Mwinyihija, M., A. Meharg, J. Dawson, N.J.C. Strachan and K. Killham, 2006. An ecotoxicological approach to assessing the impact of tanning industry effluent on river health. Arch. Environ. Contam. Toxicol., 50: 316-324.
Direct Link - Reemste, T. and M. Jekel, 1997. Dissolved organics in tannery wastewaters and their alteration by a combined anaerobic and aerobic treatment. Water Res., 31: 1035-1046.
Direct Link - Ros, M. and A. Ganter, 1998. Possibilities of reduction of recipient loading of tannery waste Slovenia. Water Sci. Technol., 37: 145-152.
Direct Link - Schellenberg, K., C. Leuenberger and R.P. Schwarzenbach, 1984. Sorption of chlorinated phenols by natural sediments and aquifer materials. Environ. Sci. Technol., 18: 652-657.
CrossRef - Shaw, L.J.Y., L.A. Glover, K. Killham, D. Osborn and A.A. Meharg, 2000. Bioavailability of 2,4- dichlorophenol associated with soil water-soluble humic material. Environ. Sci. Technol., 34: 4721-4726.
Direct Link - Song, Z., C.J. Williams and R.G.J. Edyvean, 2000. Sedimentation of tannery wastewater. Water Res., 34: 2171-2176.
CrossRefDirect Link - Sousa, S., C. Duffy, H. Weitz, A.L. Glover, E. Bar, R. Henkler and K. Killham, 1998. Use of a lux-modified bacterial biosensor to identify constraints to bioremediation of BTEX-contaminated sites. Environ. Toxicol. Chem., 17: 1039-1045.
CrossRefDirect Link - Steiert, J.G. and R.L. Crawford, 1985. Microbial degradation of chlorinated phenols. Trends Biotechnol., 3: 300-305.
CrossRef - Stein, K. and G. Schwedt, 1994. Chromium speciation in the wastewater from a tannery. Fresenius J. Anal. Chem., 350: 38-43.
Direct Link - Steinberg, S.M., E.J. Poziomek, W.H. Engelman and K.R. Rogers, 1995. A review of environmental applications of bioluminescence measurements. J. Chemosphere, 30: 2155-2197.
Direct Link - Talley, J.W., U. Ghosh, S.G. Tucker, J.S. Furey and R.G. Luthy, 2002. Particle-scale understanding of the bioavailability of PAHs in sediment. Environ. Sci. Technol., 36: 477-483.
Direct Link - Van de Lelie, D., P. Corbisier, W. Baeyens, S. Wuertz, L. Diels and M. Mergeay, 1994. The use of biosensors for environmental monitoring. Res. Microbiol., 145: 67-74.
Direct Link - Walsh, A.R. and J. O`Halloran, 1996. Chromium speciation in tannery effluent-1: An assessment of techniques and role of organic Cr(III) complexes. Water Res., 30: 2393-2400.
Direct Link - Walsh, A.R. and J. O`Halloran, 1996. Chromium speciation in Tannery effluents-II: Speciation in effluent and in a receiving estuary. Water Res., 30: 2401-2412.
Direct Link - Wiemann, M., H. Schenk and W. Hegemann, 1998. Anaerobic treatment of tannery wastewater with simultaneous sulphide elimination. Water Res., 32: 774-780.
Direct Link








