Research Article
Removal of Arsenic from Aqueous Solution by Photocatalytic UV Irradiation
Department of Environmental Health Engineering, Tehran Medical Sciences Branch, Islamic Azad University, Tehran, Iran
Arsenic is considered as one of the most toxic and carcinogenic elements (Basu et al., 2001). Arsenic compounds resemble in some respects those of phosphorus which occupies the same group (column) of the periodic table. However, arsenic is less commonly observed in the pentavalent state. The most common oxidation states for arsenic are: -3 in the arsenides such as alloy-like intermetallic compounds and +3 in the arsenites, arsenics (III) and most organoarsenic compounds (Bellack, 1971). Arsenic occurs in natural water in organic and inorganic forms. The species of inorganic arsenic are dependent on the redox conditions and the pH. Arsenic (As (V)) dominates in oxygen-rich waters and arsenite (As (III)) in reducing environments (Edwards et al., 1998). When heated in air, arsenic oxidizes to arsenic trioxide, the fumes from this reaction have an odour resembling garlic. Arsenic makes up about 1.5 ppm of the Earth’s crust. Soil contains 1-10 ppm of arsenic and Seawater has 1.6 ppb arsenic. Minerals with the formula MAsS and MAs2 (M = Fe, Ni, Co) are the dominant commercial sources of arsenic together with realgar (an arsenic sulfide mineral) and native arsenic. Arsenic also occurs in various organic forms in the environment (Bissen and Frimmel, 2003). Many countries and districts in Southeast Asia such as Vietnam and Cambodia, Western area of Iran (Kurdestan rural areas) and North Eastern and South Western coast area of Taiwan have geological environments conducive to generation of high-arsenic groundwaters (Mandal and Suzuki, 2002). Widespread arsenic contamination of groundwater has led to a massive epidemic of arsenic poisoning. It is estimated that approximately 57 million people in the many parts of the world are drinking groundwater with arsenic concentrations elevated above the World Health Organization’s standard of 10 ppb (USEPA 2002). Due to such harmful effects of arsenic on human health, new USEPA regulation lowered the Maximum Contaminant Level (MCL) for arsenic in drinking water from 50-10 μg L-1 (Sorg, 1997), therefore, it is necessary to find novel technologies to access this new regulation. A large number of technologies have been examined to remove arsenic from contaminated water (Choong et al., 2007). Many factors such as arsenic concentrations in water, presence of other constituents in water and expenses associated with a treatment process are contributed in selection of technology (Sorg, 1997). Arsenic removal methods include coagulation and precipitation (Hering et al., 1997), membrane separation (Choong et al., 2007), ion exchange (Waypa et al., 1997) and adsorption (Awual et al., 2008; Baba et al., 1999). Many of the waste streams contain As (III) which requires oxidation before it can be removed by common methods such as iron co-precipitation or ion exchange. Since the oxidation rate of dissolved As (III) by oxygen is extremely slow, oxidants such as chlorine, ozone or permanganate need to be used. Chlorine has traditionally been the oxidant of choice but there are growing concerns regarding the generation of organochloride byproducts from the naturally occurring organic matter which may also be present (USEPA, 2002). In this study Photocatalytic oxidation (PCO) was achieved with the combination of UV light rays and TiO2 nanoparticles. Nanoparticles have also solely known as effective adsorbents for some organic and inorganic substances. Therefore, this study evaluated the combination process of adsorption and advanced oxidation for arsenic removal. This process creates hydroxyl radicals and super-oxide ions, which are highly reactive electrons and designed to remove arsenic in water. The effects of other factors including initial concentration of As (III), pH and light intensity were also investigated.
TiO2 nanoparticles were purchased from Evonik-industrial company, Germany. The purity, especial surface area (BET) and sizes of the selected nanoparticles were 99.5%, 50±15 and 21 nm, respectively. Pulse Electrical Explosion (PEE) from nanoengineering and manufacturing company in Iran (model PEE10K) were used to characterize properties of TiO2 nanoparticles. Transmission Electron Microscope (TEM) image of selected TiO2 nanoparticles is shown in Fig. 1. The FTIR pattern of TiO2 nanoparticles before and after reaction is shown in Fig. 2.
 | |
| Fig. 1: | Transmission Electron Microscope (TEM) micrograph of TiO2 |
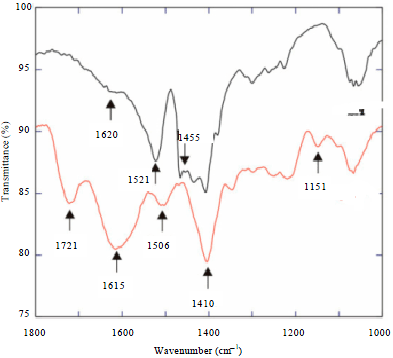 | |
| Fig. 2: | FTIR pattern of TiO2 nanoparticles before and after reaction |
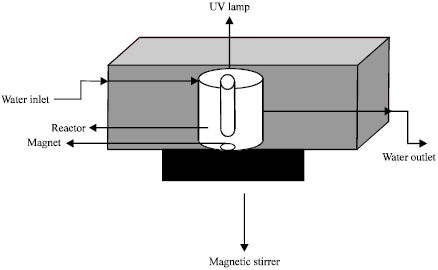 | |
| Fig. 3: | Schematic representation of photocatalytic reactor |
A 120 W UV lamp with irradiation 247 nm wave lenght inside a quartz jacket was submerged in the reactor to provide better irradiation of water samples. This reactor was operated continuously and stirred by use of a simple magnetic stirrer. The schematic diagram of the reactor used in the experiment is shown in Fig. 3. The experiments are conducted in the inner part (glass reactor) and the outer part designed to act as a cooling bath for circulating cold water and maintaining the temperature at 20°C. A 100 mg L-1 arsenic stock solution was prepared from Na2HAsO4.7H2O. The arsenic solutions, used in this study with 5, 15 and 20 mg L-1 concentration, were obtained by diluting the stock solution to desired concentration in distilled water. The effects of other parameters including initial pH (3, 7 and 11), arsenic concentration, amount of titanium oxide nanoparticles (0.25, 0.5, 0.75, 1 and 1.5 mg L-1) and reaction time were investigated. The batch adsorption experiments conducted for each concentration with different amounts of TiO2 nano particles. The photocatalytic reaction then undertaken by switching on UV lamp. All the experiments were done by maintaining one factor constant and changing other parameters. Samples then centrifuged (4000 rpm) and the amount of arsenic was detected by Hatch arsenic kits (Product No. 2822800) whose validity was approved previously (Edwards et al., 1998). The pH of the solutions was controlled by addition of NaOH or HCl. The pH of solution was measured using pH meter (Metrohm E520).
Effects of initial TiO2 concentration and adsorption time: The effects of different initial TiO2 nanoparticles on arsenic removal were investigated at arsenic initial concentration of 15 mg L-1 and pH 7. As shown in Fig. 4, as the adsorbent concentrations increased, arsenic removal enhanced. The initial sharp rise within 20 min of adsorption indicates the availability of readily accessible sites for adsorption. However, after 25 min a plateau is reached, indicating virtually no more sites remain available for further arsenic adsorption. Moreover, arsenic removal increased from 35-68% when TiO2 nanoparticles concentration increased from 0.25-1 mg L-1. No hefty removal in arsenic observed when adsorbent dosage increased to 1.5 mg L-1. Therefore, This value was chosen for the conduction of photocatalytic experiments.
Effects of initial pH in photocatalytic reaction: The effects of various pH were investigated and the results are shown in Fig. 5. The experiment conducted with arsenic initial concentration of 15 and 1 mg L-1 of TiO2 nanoparticles.
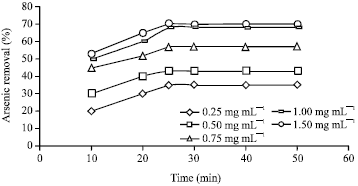 | |
| Fig. 4: | Effects of TiO2 concentrations on arsenic adsorption |
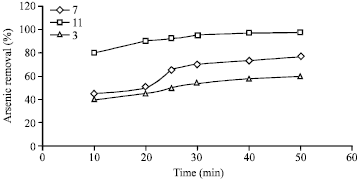 | |
| Fig. 5: | Effect of pH in photocatalytic reaction |
As shown in Fig. 5, maximum removal of arsenic (95%) was observed in pH 11. At pH 3, removal efficiencies decreased. The following equations shows arsenic photocatalytic oxidation with TiO2 (Aceituno et al., 2002).
 |
The zero charge point for TiO2 particles depending on their anatas form various from 5-7. If the mentioned pH is higher or lower, the TiO2 surface charge becomes negative and positive, respectively. As a result, TiO2 surface in basic conditions has negative charge and arsenic absorption on the negative surface of TiO2 is easier in basic pH. Decreasing pH gradually increases the electrostatic repulsion between TiO2 surface and arsenic which is positively charged at pH 5 (Hidaka et al., 2004).
Effects of initial arsenic concentration: The effects of initial arsenic concentration of its removal in pH 11 is shown in Fig. 6. It is shown that initial arsenic concentration strongly affected its removal. The removal observed decreased as initial arsenic concentration gradually increased from 5-20 mg L-1. When the amount of arsenic is small the photogenerated electrons efficiently transfer to TiO2. As the result better separation of electrons and holes would be achieved. These electrons could react with adsorbed oxygen molecular or surface Ti4+ to form reactive species O2 and reactive center surface Ti3+, respectively. This suggested that the recombination was slowed and the generation of O2 and surface Ti3+ was accelerated. In this situation the yield of OH would also be increased. However, higher amount of arsenic lead to its deposition which would cover more TiO2 surface and hinder the contact between TiO2 and arsenic which would increase diffuse distance and decrease the amount of received photons. Furthermore, as the radical densities were equal in all the solutions, a solution with low arsenic concentration with the same hydroxyl radical rate, would have higher transformation rate in comparison to solutions with higher density rates (Lopez et al., 2000).
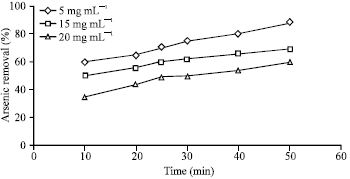 | |
| Fig. 6: | Effect of arsenic initial concentration (mg L-1) in photocatalytic reaction |
The obtained results indicate that the UV/TiO2 process is robust to remove arsenic by 1 mg L-1 TiO2 within 25 min. It is also proved in this study that arsenic removal using this process follows the first order kinetics. Moreover, results showed that arsenic was better in basic pH. Removal enhanced in lower concentration of arsenic as in higher concentration it deposited on UV lamps which subsequently reduced UV penetration on TiO2 area.
We are thankful to the Azad university, Tehran medical branch for the partial support of this study.