ABSTRACT
A method for the vegetative propagation of Dillenia suffruticosa with important cultural, medicinal and ecological values was developed. Juvenile stem cuttings were collected from wild populations in Brunei Darussalam. The cuttings with or without leaves were treated with Indole Butyric Acid (IBA), Naphthaleneacetic acid (NAA), or Indole Acetic Acid (IAA) at 0, 0.01, 0.05, 0.10, or 0.20% (w/v) using a quick-dip method, planted in rooting media and kept in a misted greenhouse. The survival and rooting percentages, number and length of new roots produced, number and area of leaves and Relative Growth Rate (RGR) based on stem length and stem diameter were assessed at a 4-week interval for 24 weeks. The leafless cuttings and leafy cuttings treated with distilled water after week 4 and 16, respectively, did not survive. All of the leafy stem cuttings treated with 0.10 and 0.20% NAA and 0.20% IAA survived for 24 weeks. Leafy stem cuttings treated with 0.10 and 0.20% NAA and not ABA had formed significantly more and longer roots than cuttings in the other treatments at week 8. Leafy stem cuttings treated with 0.10% IAA had more new leaves and greater leaf area, than cuttings in the other treatments at week 8. There were no significant differences in the RGR of leafy stem cuttings among the different treatments at week 8. This study demonstrates that treatments with 0.10-0.20% NAA and 0.10% IAA and not IBA resulted in the development roots and shoots from leafy stem cuttings and can be used for the mass production of D. suffruticosa plantlets, which will prevent the over exploitation of wild stocks.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=rjb.2015.73.87
INTRODUCTION
Dillenia suffruticosa (Griff. ex Hook. f. and Thomson) Martelli belong to the family Dilleniaceae, which contains approximately 12 genera and 350 species (Cullen, 2001). In Brunei Darussalam, D. suffruticosa is a common evergreen shrub that is known by various local Brunei names, for example simpur air, jingin and tengering (Said et al., 2003). Its flower has been selected as the national flower and is featured on Brunei’s one-dollar note (Thurman and Cheong, 2008; LaFrankie, 2010). The flower represents favorable economic growth and the green leaf represents the progress of Brunei’s economy.
Besides its ornamental values, D. suffruticosa also has high potential values as a medicinal and herbal plant. Its roots were shown to exhibit anticancer activity and have antioxidant activity and stronger cytotoxic activity than extracts from other parts of the plant (flower, fruit and leaf) (Armania et al., 2013; Tor et al., 2014). However, the leaf extract showed antibacterial and antiviral properties (Wiart et al., 2004; Wiart, 2006; Muliawan, 2008). Moreover, the fruit of D. suffruticosa can be used as treatment for cancerous growths (Ahmad and Holdsworth, 1995). In folk remedies, the compressed leaves and boiled roots of D. suffruticosa have been used to ease stomach cramps (Adfa, 2005).
As well as its medicinal and bioactive properties, D. suffruticosa is one of the best potential plants to rehabilitate and stabilize slopes to prevent landslides (Mafian et al., 2009). This plant is also an important component of the ecosystem as the fruits are food for wild birds and mammals and the pollen is an important food source for Xylocopa (carpenter bees) species (Thurman and Cheong, 2008). The large leaf of D. suffruticosa is often made into nests by birds.
Even though D. suffruticosa is a very common plant in Brunei Darussalam, its seeds have a very low germination rate. Consequently, it is very difficult to obtain seedlings via seed propagation (Besar, 1995; Manjul, 2013). In this respect, Baskin and Baskin (2001) reported that the seeds germinated readily after imbibing water. However, Manjul (2013) obtained the opposite result and showed that seeds failed to germinate after imbibing water. The problems of low germination percentages and slow germination rates may be overcome by using vegetative propagation techniques, for example, using stem cuttings, because such techniques do not rely on seeds (Munjuga et al., 2008).
Auxins are plant growth regulators that are used to induce root formation from cuttings. Various auxins have been shown to improve overall rooting percentages, hasten root initiation, increase the number and quality of roots (Blythe et al., 2007) and promote the development of uniform roots (Boyer et al., 2013). Auxins induce root formation and establish hormonal signals that control root development (Went, 1938; Calenza et al., 1995). Different auxins have been used in the vegetative propagation of tropical shrubs and trees with medicinal and timber values (Husen, 2008; Kipkemoi et al., 2013). Auxin-treated stem and leaf cuttings form adventitious roots; making auxins the most commonly used plant hormones in vegetative propagation (Wiesman et al., 1988; Overvoorde et al., 2010). However, the responses to different types and concentrations of auxins vary among plant species and are affected by genotype (Guo et al., 2009).
The aim of this research was to develop methods for the conservation of D. suffruticosa. If this plant is to be used for its cultural, medicinal and ecological values, it is important to have a large and reliable source of plant materials so that no plants will be collected from wild populations. Treating stem cuttings with rooting hormones, such as auxins, is a traditional and promising method for the multiplication and vegetative propagation of this plant.
The specific objectives of this study were to determine the effects of different types and concentrations of auxins on the survival, rooting and growth of stem cuttings and to determine whether the presence of leaves on cuttings is important for their survival, rooting and growth.
MATERIALS AND METHODS
Sampling sites: Stock plants of D. suffruticosa (average height = 100±0.05 cm) were selected from wild populations growing in the coastal areas of Brunei Darussalam (Table 1). In total, 260 randomly chosen juvenile stems (one stem cutting per plant) were sampled from March to June 2014. One two-node stem cutting 8-12 cm long and 0.6-1.2 cm in diameter was sampled from each plant to reduce over harvesting of wild plants. Healthy and uniform stem cuttings were collected at all sampling sites in the early morning (07:00) to minimize drying.
| Table 1: | Global positioning system coordinates and elevations of sampling sites of wild populations of Dillenia from which stem cuttings were obtained. All wild populations were located in coastal areas of Brunei Darussalam |
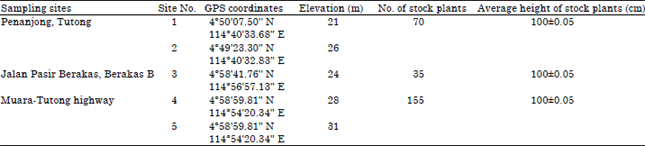 | |
| One juvenile stem cutting was collected from each plant. Sampling was conducted from March to June 2014 | |
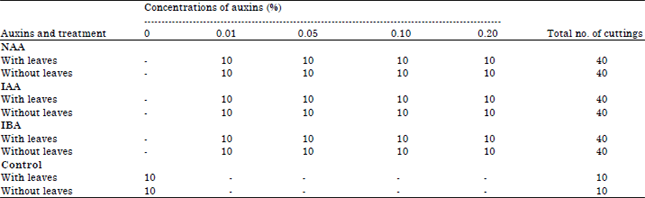
| Table 2: | Total number of cuttings in different types and concentrations of auxins and types of stem cuttings |
 | |
Type of stem cuttings: Two types of tip stem cuttings were prepared according to Aminah et al. (1995). The first is leafy cutting in which two-thirds of the leaves were removed and the second is cutting without leaves. All flowers, flower buds and fruits on the stem cuttings were removed and the basal parts of the stems were trimmed just below the node.
Types and concentrations of auxins: Three types of auxins were used: 1-naphthaleneacetic acid (NAA), 3-indoleacetic acid (IAA) and Indole-3-Butyric Acid (IBA). The treatment method was that described by Guo et al. (2009). A 0.20% (w/v) stock solution of each auxin was prepared and then 0.01, 0.05 and 0.10% w/v solutions were prepared by dilution. Ten cuttings (n = 10) were used for each treatment, including the control (double distilled water) (Table 2). Cuttings were dipped quickly (5-8 sec) into the test solution (5 mL) and then planted in square plastic pots (36 cm2×6.0 cm) containing a 1:1 mixture of black soil and perlite (1 cutting per 625 g soil). The cuttings were grown in this medium until roots started to appear. Both the soil and perlite were autoclaved before potting to prevent fungal and microbial growth. The cuttings were placed in a hole in the soil medium that was made with a pencil and the soil was pressed firmly around the cutting. After 6 weeks, the plants were re-potted into larger square pots (121 cm2×13 cm) with the same growth medium (800 g). After 10 weeks, the medium was replaced with black soil (950 g) supplemented with slow-release fertilizer (N-P-K; 15:15:15).
Preparation of growth media and growth conditions: The cuttings were kept in a greenhouse at the Biology Lab, Universiti Brunei Darussalam for the first 10 weeks of this experiment. Pots were arranged in two rows on benches inside the greenhouse. Misting units were located 80 cm above the benches and released water mist for 1 min at 5 min intervals. The pots containing the cuttings were covered with a plastic sheet to prevent water loss from the soil and to maintain high atmospheric humidity. The stem cuttings were grown at 22-29°C, relative humidity of 80-90% and light at 300 μmol m–2 sec–1 (measured with an Apogee Quantum MQ-200 PAR Meter, Apogee Instruments, UK). The pots were placed in a random arrangement on the benches and were moved to new random positions each week. After 10 weeks, the cuttings were moved to a shade house and grown at 22-34°C, relative humidity of 60-75% and light at 450 μmol m–2 sec–1. The cuttings were watered twice a day.
Assessment of cuttings: The following parameters for stem cuttings in each treatment were calculated according to Aminah et al. (1997): survival percentage (%), rooting percentage (%), number of roots, total length of roots (cm), number of new leaves and leaf area (cm2). These parameters were analyzed and recorded at every 4 week intervals from week 0 (initial measurement) to week 24. Stems that turned brown were considered dead and discarded to assess the survival percentage, the number of cuttings surviving at each sampling time was divided by the total number of cuttings per treatment. Stem cuttings with root length of 1 cm or longer were considered to be rooted, as described by Baul et al. (2011). The rooting percentage was determined at every 4 week intervals by dividing the number of rooted cuttings by the total number of cuttings in each treatment.
The numbers of new roots and leaves on each rooted cutting were counted and total root length was measured (cm) using a vernier caliper. All newly produced leaves were traced onto blank paper. The leaf shapes were cut from the paper and leaf areas were measured (in cm2) using a leaf meter (ADC, BioScientific Ltd., England).
Relative Growth Rates (RGRs) based on the change in stem diameter and stem length of seedlings over time were calculated as described by Hunt (1982), as follows:
![]()
where, W2 and W1 represent the final and initial stem diameter for RGRstem diameter and the final and initial stem length (cm) for RGRstem length and t2 -t1 was 8 weeks. The calculations of RGR values were based on stem diameter and stem length measured at week 8. Although the rooting percentage, number and length of new roots produced, number and area of leaves of leafy stem cuttings were measured at every 4 weeks until week 24, only data collected at week 8 was used in the statistical analysis, due to high mortality of leafy stem cuttings after week 8.
Statistical analysis: The survival and rooting percentage data was arcsine-square transformed before statistical analysis. The number of roots, total root length, number of new leaves, leaf area, RGRstem diameter and RGRstem length were compared between the two cutting types (leafy and no-leaf cuttings) and among the three auxin types (NAA, IAA, IBA) and four auxin concentrations (0, 0.01, 0.05, 0.10 and 0.20% w/v) using a three-way analysis of variance (ANOVA), followed by Tukey’s Honest Significant Difference (Tukey’s HSD) tests. However, since none of the no-leaf stem cuttings survived, two-way ANOVA and Tukey’s HSD tests were conducted. Assumptions of normality and equal variances were checked during the two-way ANOVA and were not violated. All statistical analyses were conducted using R version 3.1.2 (R Development Core Team, 2014).
RESULTS
Survival percentages of stem cuttings: Both treated and non-treated leafless stem cuttings did not survive after week 4 (data not shown), whereas all of the leafy stem cuttings that were treated with different auxins and water (control) survived (Fig. 1), giving higher survival rates than that of the control after 8 weeks of growth. In the control, leafy stem cuttings showed an 80% survival rate at week 8 but all of them died from week 16 onwards (Fig. 1). Leafy stem cuttings treated with NAA at 0.05, 0.10 and 0.20% or with IAA or IBA at 0.10 and 0.20% showed 100% survival at week 8 (Fig. 1). From weeks 16-24, the survival rates of leafy stem cuttings treated with IBA at 0.01% (20-30%) or IAA at 0.05% (30%) were higher than that of leafy stem cuttings treated with NAA (Fig. 1). None of the leafy stem cuttings treated with a low concentration of NAA (0.01%) survived after week 16, however, those treated with higher concentrations of NAA (0.10 and 0.20%) or IAA (0.20%) showed a 100% survival rate at week 24 (Fig. 1). Because of the high mortality rates of cuttings treated with low auxin concentrations, the statistical analyses were based on data obtained at week 8. This allowed us to compare survival rates and rooting percentages among all of the types and concentrations of auxins (Fig. 1).
Rooting percentages and number and length of roots on stem cuttings: The leafy stem cuttings that were treated with auxins or distilled water (control) were able to form roots in the first 4 weeks after treatment. However, because of the high mortality rates of leafy stem cuttings from week 8-24, especially those treated with low auxin concentrations, root data obtained at week 8 were used for the statistical analyses. The leafy stem cuttings treated with auxins rooted better (formed more roots and longer roots) than control cuttings.
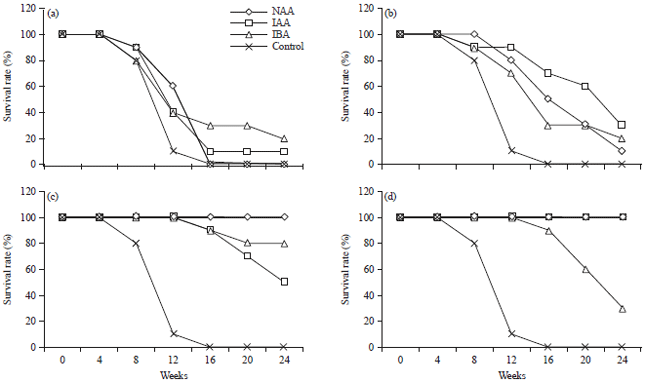 | |
| Fig. 1(a-d): | Survival percentages of leafy stem cuttings (initial number of stem cuttings = 10) of Dillenia suffruticosa after treatments with water (control) or with different concentrations (a) 0.01, (b) 0.05, (c) 0.10 and (d) 0.20% w/v of auxins (NAA, IAA, or IBA) over 24 weeks |
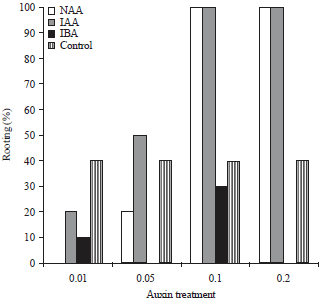 | |
| Fig. 2: | Rooting percentages of leafy stem cuttings (initial number of stem cuttings = 10) of Dillenia suffruticosa at 8 weeks after treatments with different concentrations (0.01, 0.05, 0.10, or 0.20%) of auxins (NAA, IAA, or IBA) |
| Table 3: | F-statistics and p-values following two way analysis of variance of parameters of Dillenia suffruticosa treated with different types auxins (NAA, IAA, or IBA) at various concentrations (0.01, 0.05,0.10, or 0.20%) |
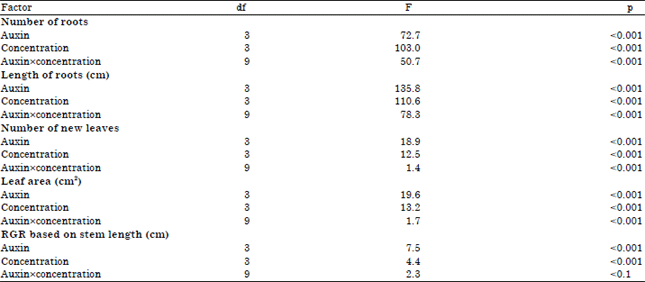 | |
| Data collected at week 8 were used in statistical analyses, df: Degrees of freedom, NS: Not significant | |
In the control, 40% of the leafy) stem cuttings had formed roots by week 8 (Fig. 2), whereas 100% of those treated with high concentrations of NAA or IAA (0.10 and 0.20%) had formed roots at week 8 (Fig. 2).
There were significant differences in the mean number of roots and root length among the different types of auxins (p<0.001) and among the different concentrations of auxins (p<0.001). There was also a significant interaction between the types and concentrations of auxins (p<0.001) (Table 3).
Similar to the survival percentages, high concentrations of NAA resulted in significantly more roots per cutting (0.10% NAA = 2.10±0.23; 0.20% NAA = 4.70±0.30) and longer roots (0.10% NAA = 29.8±4.6 cm and 0.20% NAA = 58.9±3.7 cm) compared with other auxin treatments and the control (p<0.05) at week 8 (Fig. 3).
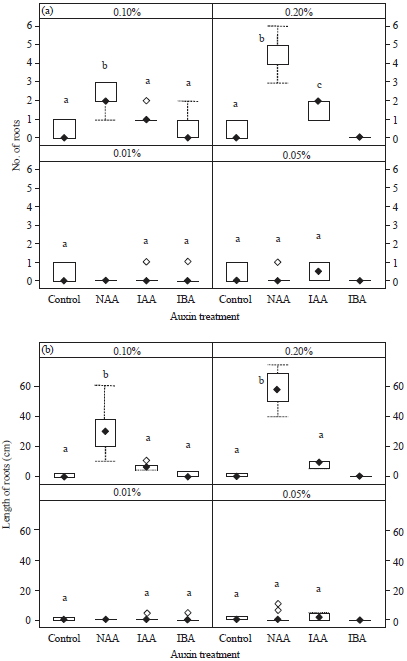 | |
| Fig. 3(a-b): | (a) Number of roots and (b) Length of roots on leafy stem cuttings (n = 10) of Dillenia suffruticosa at 8 weeks after treatment with different concentrations (0.01, 0.05, 0.10, or 0.20%) of auxins (NAA, IAA, or IBA). Different letters within each auxin concentration indicate significant differences compared with control (p<0.05; one-way ANOVA followed by Tukey’s HSD). No letter indicates that cuttings did not form roots; therefore, these treatments were excluded from statistical analyses |
The cuttings treated with 0.20% NAA showed the highest mean number of new roots and the longest roots but stem cuttings treated with a lower NAA concentration (0.01%) produced no new roots (Fig. 3). Cuttings treated with IBA or IAA produced few or no roots (Fig. 3).
Number of new leaves and leaf areas of stem cuttings: There were significant differences in the mean number of leaves and leaf area among the different auxins (p<0.001) and the different concentrations of auxins (p<0.001). For these parameters, there was also a significant interaction between the types and concentrations of auxins (p< 0.001) (Table 3). The cuttings treated with IAA formed more new leaves (0.20±0.13 at 0.01%, 1.40±0.22 at 0.20%) than did the cuttings treated with the other auxins. The IAA treated cuttings also showed the largest new leaf area (23.54 ±2.99 cm2 at 0.01%, 299.60±47.89 cm2 at 0.20%). The stem cuttings treated with 0.10 and 0.20% NAA formed some new leaves but those treated with distilled water, 0.01 and 0.05% NAA and 0.05 and 0.20% IBA did not form any new leaves (Fig. 4).
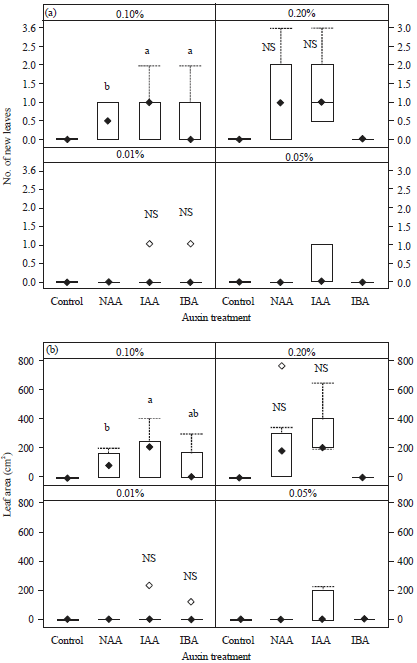 | |
| Fig. 4(a-b): | (a) Number of new leaves and (b) Areas of leaves on leafy stem cuttings (n = 10) of Dillenia suffruticosa at 8 week after treatment with different concentrations (0.01, 0.05, 0.10, or 0.20%) of auxins (NAA, IAA, or IBA). Different letters within each auxin concentration indicate significant differences compared with control (p<0.05; one-way ANOVA followed by Tukey’s HSD test or t-test). No letter indicates that cuttings did not produce new leaves, so these treatments were excluded from statistical analyses |
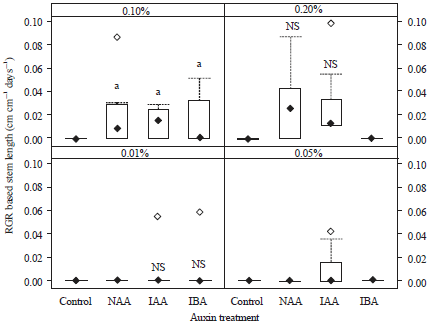 | |
| Fig. 5: | Relative growth rate based on stem length of leafy stem cuttings (n = 10) of Dillenia suffruticosa at 8 weeks after treatment with different concentrations (0.01, 0.05, 0.10, or 0.20%) of auxins (NAA, IAA, or IBA). Different letters within each auxin concentration indicate significant difference compared with control (p<0.05; one-way ANOVA followed by Tukey’s HSD test). No letter indicates that cuttings had not shown an increase in growth and so these treatments were excluded from statistical analyses |
Relative growth rate based on stem length and stem diameter of stem cuttings: The RGR based on stem diameter was not included in statistical analyses because there was no noticeable increase in stem diameter during the experiment. The stem diameter measured at 4 week interval was the same as the initial stem diameter for each leafy stem cuttings.
The mean RGR based on the stem length (cm cm–1 day–1) at week 8 differed significantly among the three types of auxins (p<0.001) and among the different auxin concentrations (p<0.001). The interaction between the types and concentrations of auxins was not significant (p>0.05) (Table 3). However, when the RGR data was analyzed using one way ANOVA or t-test within each auxin concentration, there were no significant differences among the three types of auxins (p>0.05, Fig. 5). The highest mean RGRstem length was in stem cuttings treated with 0.20% NAA (0.03±0.01 cm cm–1 day–1) (Fig. 5). All of the leafy cuttings treated with auxins at 0.10% showed higher RGRs based on stem length than that of the control cuttings (Fig. 5).
DISCUSSION
Importance of presence of leaves and auxins in vegetative propagation techniques: In this study, none of the stem cuttings without leaves that are treated with auxins or not survived after 4 weeks. Several other studies have reported that stem cuttings without leaves showed a high mortality rate in vegetative propagation (Badji et al., 1991; Ofori et al., 1996; Akinyele, 2010). This is because an active leaf is necessary to provide energy for the growth of stem cuttings, as there are very little reserves in the stem (Reuveni and Raviv, 1980). According to Reuveni and Raviv (1980), leafy cuttings of avocado (Persea americana Mill.) showed a higher rooting percentage and sucrose content at the base than that of cuttings without leaves. For many species, the presence of leaves on cuttings has been shown to stimulate root initiation (Hartmann et al., 1990). Leaves are important because they promote root formation and manufacture carbohydrates via photosynthesis (Sandhu et al., 1989). Rooting is stimulated by high levels of available carbohydrates provided by the leaves. Therefore, for cuttings to form new shoot and root tissues, it is important that they are supplied with appropriate amounts of light and water, since both are required for photosynthesis (Wage, 2001).
According to Wage (2001), a misting system is important for vegetative propagation to ensure sufficient moisture in the rooting media and provides high humidity conditions for stem cuttings. The current study presented that treating leafy stem cuttings with auxins was also important for obtaining high quality planting stocks of D. suffruticosa, because none of the non-treated leafy stem cuttings, which are those treated with only water, survived after week 16. The function of auxins in root initiation is important for the survival of stem cuttings. Uniyal et al. (1993) found that the formation of roots on cuttings treated with auxins was important for survival, as cuttings in the control that did not form roots wilted and died. Similarly, Srivastava et al. (1986) reported that the majority of dipterocarp shoots wilted if roots failed to form.
Effects of auxin types and concentrations on survival and rooting percentages and growth: The results of this study illustrate the importance of identifying appropriate types and concentrations of auxins to promote rooting in vegetative propagation. According to Sandhu et al. (1989), root formation is induced more effectively by auxins than by other plant growth regulators including cytokinins, gibberellins and ethylene. Plant growth regulators are applied exogenously to produce a variety of growth effects, because endogenous hormones are usually at sub-optimal levels, as low as 1 μM in plant tissues (Pessarakli, 2002). Auxins are also known to enhance the hydrolysis of nutritional reserves in the plant. In this study, the types and concentrations of auxins significantly affected the survival and rooting percentages of stem cuttings and higher concentrations of NAA and IAA resulted in greater survival percentages at 24 weeks. Low concentrations of NAA and IAA resulted in no rooting or low rooting percentages. Exogenous auxins were shown to increase the availability of carbohydrates at the site of root development (Middleton et al., 1980). According to Middleton et al. (1980), auxins may directly control the mobilization of carbohydrates from the leaves to the hypocotyl and roots.
All of the stem cuttings of D. suffruticosa treated with 0.10 and 0.20% NAA and IAA formed roots within 8 weeks. Higher concentrations of NAA and IAA resulted in more roots per cutting and longer roots. The IBA was less effective than IAA and NAA at inducing the formation of healthy roots, especially at high concentrations. High IBA concentrations may have been toxic to the cuttings, as was the case for Holarrhena pubescens cuttings (Baul et al., 2010).
Based on the survival and rooting percentages, the results of this study confirmed that NAA (0.10 and 0.20%) and IAA (0.20%) treatments were appropriate for rooting and survivorship of D. suffruticosa cuttings. Numerous studies ( De Klerk et al., 1997; Thirunavonkkarasu and Saxena, 1997; Soyler and Arslan, 2000; Boyer and Graves, 2009; Avci et al., 2010) also reported that IAA was the best auxin to induce root formation on stem cuttings.
However, various species respond differently to different types of auxins (Brown, 2008). The initiation and development of roots on stem cuttings is controlled by complex ecophysiological and biochemical processes, as well as anatomical factors (Uniyal et al., 1993). According to Veeraragavathatham et al. (1983), the rooting of stem cuttings is also dependent on endogenous growth regulators and the inactivation of various enzymes. In a previous study, different types of roots formed in response to NAA and IBA treatments; IBA induced a strong fibrous root system to form, while NAA resulted in a bushy root system (Brown, 2008). Dillenia suffruticosa has very bushy adventitious roots, making NAA the most appropriate hormone for its rooting.
In this study, leafy cuttings treated with 0.10% IAA formed the most new leaves and produced the largest new leaf area. The leafy cuttings in this treatment also showed a high rooting percentage, indicating that better leaf development was related to better rooting. Seran and Umadevi (2011) reported that stem cuttings of Citruslimon L. treated with IAA were able to produce a large number of new leaves because they had developed a vigorous rooting system. Stancato et al. (2003) showed that roots enabled cuttings to absorb more nutrients and produce more leaves. Cuttings with more new leaves and more shoots showed higher survivorship rates when grown in natural conditions (Pessarakli, 2002).
Juvenile D. suffruticosa cuttings were used in this study because other studies have shown that juvenile shoot cuttings are more responsive than mature ones to plant growth regulators (Ab Shukor and Liew, 1994). Compared with mature stems, juvenile stems contain negligible amounts of rooting inhibitors and have less differentiated cells and more actively dividing cells (Paton et al., 1970; Hartmann et al., 1990; Lakshmanan et al., 1995). Several other studies have reported that juvenile cuttings of tropical plants such as Dryobalanops oblongifiolia, Shorea acuminata, S. parvifolia and S. leprosula formed new roots more readily than did cuttings from mature plants (Penguang, 1978; Alias, 1984; Ab Shukor and Liew, 1994). The condition of stock plants from which cuttings are taken is also important, because only healthy plants that are free of disease and insect damage will produce high quality planting stocks (Wage, 2001). However, stems need to be cut at the right position to allow for subsequent root formation. They should be cut below the nodes to include the very active dividing cells that are more likely to form roots (Wage, 2001).
At 8th week, the RGR values based on stem length and stem diameter did not vary significantly among the different treatments. The cuttings produced roots and new leaves based on the types and concentrations of auxins but their stem length/stem diameter were not affected. This result differs from those of Memon et al. (2013), who reported that cuttings of Bougainvillea treated with NAA showed a greater increase in stem length per cutting than did those in the control.
CONCLUSION
A method for the vegetative propagation of D. suffruticosa was developed and the presented results showed that juvenile leafy stem cuttings treated with auxins formed roots and new leaves. Stem leafy cuttings treated with 0.10 and 0.20% NAA showed high survival and rooting percentages. The stem leafy cuttings treated with 0.20% IAA showed a 100% survival rate and strong shoot development (highest number of new leaves and largest new leaf area). At week 8, cuttings treated with 0.10 and 0.20% NAA had formed significantly more roots and longer roots, than cuttings in the other treatments. All of the leafy cuttings treated with NAA or IAA at 0.10 and 0.20% formed roots. None of the treatments resulted in differences in RGRstem length and RGRstem diameter at 8or 24 weeks, suggesting that 24 weeks may not be long enough to observe differences in growth related to the different auxin treatments. The results of this study showed that NAA and IAA are suitable for the mass propagation of D. suffruticosa cuttings. In particular, 0.10-0.20% NAA and 0.10% IAA and not ABA treatments led to root and shoot development from stem cuttings. Rooting of stem cuttings of D. suffruticosa is a viable propagation method to produce sufficient resources for habitat rehabilitation and for research on the medicinal properties of this important plant.
ACKNOWLEDGMENTS
This work was funded by Universiti Brunei Darussalam. We thank the staff of the Environmental and Life Sciences Programme, Faculty of Science and the Department of Forestry, Brunei Darussalam for their assistance with field and laboratory work.
REFERENCES
- Adfa, M., 2005. Survey etnobotani, studi senyawa flavonoid dan uji brine shrimp beberapa tumbuhan obat tradisional suku Serawai di Propinsi Bengkulu. Gradien, 1: 43-50.
Direct Link - Ahmad, F.B. and D.K. Holdsworth, 1995. Traditional medicinal plants of sabah, Malaysia Part III. The rungus people of Kudat. Int. J. Pharmacogn., 33: 262-264.
CrossRefDirect Link - Akinyele, A.O., 2010. Effects of growth hormones, rooting media and leaf size on juvenile stem cuttings of Buchholzia coriacea Engler. Ann. For. Res., 53: 127-133.
Direct Link - Memon, N., N. Ali, M.A. Balochi and Q. Chartar, 2013. Influence of Naphthalene Acetic Acid (NAA) on sprouting and rooting potential of stem cuttings of Bougainvillea. Sci. Int., 25: 299-304.
Direct Link - Aminah, H., J.M. Dick, R.R.B. Leakey, J. Grace and R.I. Smith, 1995. Effect of Indole Butyric Acid (IBA) on stem cuttings of Shorea leprosula. For. Ecol. Manage., 72: 199-206.
CrossRefDirect Link - Aminah, H., J.M. Dick and J. Grace, 1997. Rooting of Shorea leprosula stem cuttings decreases with increasing leaf area. For. Ecol. Manage., 91: 247-254.
CrossRefDirect Link - Armania, N., L.S. Yazan, I.S. Ismail, J.B. Foo and Y.S. Tor et al., 2013. Dillenia suffruticosa extract inhibits proliferation of human breast cancer cell lines (MCF-7 and MDA-MB-231) via induction of G2/M arrest and apoptosis. Molecules, 18: 13320-13339.
CrossRefDirect Link - Avci, S., S. Cocu, M. Aasim, C. Sancak and S. Ozcan, 2010. Effects of treating with auxin solutions on rooting of cuttings of sainfoin (Onobrychis viciifolia). Trop. Grasslands, 44: 123-127.
Direct Link - Badji, S., I. Ndiaye, P. Danthu and J.P. Colonna, 1991. Vegetative propagation studies of gum arabic trees. 1. Propagation of Acacia senegal (L.) Willd. using lignified cuttings of small diameter with eight nodes. Agroforest Syst., 14: 183-191.
CrossRefDirect Link - Baul, T.K., M. Mezbahuddin, M.M. Hossain and M. Mohiuddin, 2010. Vegetative propagation of Holarrhena pubescens, a wild tropical medicinal plant: Effect of indole-3-butyric acid (IBA) on stem cuttings. For. Stud. China, 12: 228-235.
CrossRefDirect Link - Baul, T.K., M.M. Hossain, M. Mezbahuddin and M. Mohiuddin, 2011. Vegetative propagation of Litsea monopetala, a wild tropical medicinal plant: Effects of indole-3-butyric acid (IBA) on stem cuttings. J. For. Res, 22: 409-416.
CrossRefDirect Link - Boyer, N.Z. and W.R. Graves, 2009. NAA is more effective than IBA for rooting stem cuttings of two Nyssa spp. J. Environ. Horticult., 27: 183-187.
Direct Link - Calenza, J.L. Jr., P.L. Grisafi and G.R. Fink, 1995. A pathway for lateral root formation in Arabidopsis thaliana. Genes Dev., 9: 2131-2142.
PubMedDirect Link - De Klerk, G.J., J.T. Brugge and S. Marinova, 1997. Effectiveness of indoleacetic acid, indolebutyric acid and naphthaleneacetic acid during adventitious root formation in vitro in Malus Jork 9. Plant Cell Tissue Organ Cult., 49: 39-44.
CrossRefDirect Link - Guo, X.F., X.L. Fu, D.K. Zang and Y. Ma, 2009. Effect of auxin treatments, cutting's collection date and initial characteristics on Paeonia Yang Fei Chu Yu cutting propagation. Scientia Horticulturae, 119: 177-181.
CrossRefDirect Link - Husen, A., 2008. Clonal propagation of Dalbergia sissoo Roxb. and associated metabolic changes during adventitious root primordium development. New For., 36: 13-27.
CrossRefDirect Link - Kipkemoi, M.N.R., N.P. Kariuki, N.V. Wambui, O. Justus and K. Jane, 2013. Macropropagation of an endangered medicinal plant Strychnos henningsii (gilg) (loganiaceae) for sustainable conservation. Int. J. Med. Plants Res., 2: 247-253.
Direct Link - Lakshmanan, P., C.S. Loh and C.J. Goh, 1995. An in vitro method for rapid regeneration of a monopodial orchid hybrid Aranda Deborah using thin section culture. Plant Cell Rep., 14: 510-514.
CrossRefDirect Link - Mafian, S., B.B.K. Huat, N.A. Rahman and H. Singh, 2009. Potential plant species for live pole application in tropical environment. Am. J. Environ. Sci., 5: 759-764.
CrossRefDirect Link - Middleton, W., B.C. Jarvis and A. Booth, 1980. The role of leaves in auxin and boron-dependent rooting of stem cuttings of Phaseolus aureus roxb. New Phytol., 84: 251-259.
CrossRefDirect Link - Muliawan, S.Y., 2008. Effect of Dillenia suffruticosa extract on dengue virus type 2 replication. Universa Med., 27: 1-5.
Direct Link - Overvoorde, P., H. Fukaki and T. Beeckman, 2010. Auxin control of root development. Cold Spring Harbor Perspect. Biol., Vol. 2.
CrossRefDirect Link - Paton, D.M., R.R. Willing, W. Nicholls and L.D. Pryor, 1970. Rooting of stem cuttings of Eucalyptus: A rooting inhibitor in adult tissue. Aust. J. Bot., 18: 175-183.
CrossRefDirect Link - R Development Core Team, 2014. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria.
Direct Link - Reuveni, O. and M. Raviv, 1980. Importance of leaf retention to rooting of avocado cuttings. J. Am. Soc. Hortic. Sci., 106: 127-130.
Direct Link - Sandhu, A.S., S.N. Singh, P.P.S. Minhas and G.P.S. Grewal, 1989. Rhizogenesis of shoot cuttings of raspberry (Physalisperuviana L.). Indian J. Horticult., 46: 376-378.
Direct Link - Seran, T.H. and T. Umadevi, 2011. Influence of Indole Acetic Acid (IAA) on the establishment of stem cuttings in lemon (Citrus limon L.). J. Agric. Res., 49: 517-524.
Direct Link - Ab Shukor, N.A. and T.S. Liew, 1994. Effects of plant materials, cutting positions, rooting media and IBA on rooting of shorea leprosula (Dipterocarpacea) Cuttings. Pertanika J. Trop. Agric. Sci., 17: 49-53.
Direct Link - Soyler, D. and N. Arslan, 2000. The effects of some plant growing regulators on the rooting of capers (Capparis spinosa L.). Turk. J. Agric. For., 24: 595-600.
Direct Link - Stancato, G.C., F.F.A. Aguiar, S. Kanashiro, A.R. Tavares, E.L.M. Catharino and R.B. de Aguiar Silveira, 2003. Rhipsalis grandiflora Haw. (Cactaceae) propagation by setem cuttings. Scientia Agricola, 60: 651-656.
CrossRefDirect Link - Tor, Y.S., L.S. Yazan, J.B. Foo, N. Armania and Y.K. Cheah et al., 2014. Induction of apoptosis through oxidative stress-related pathways in MCF-7, human breast cancer cells, by ethyl acetate extract of Dillenia suffruticosa. BMC Complement. Altern. Med., Vol. 14.
CrossRefDirect Link - Veeraragavathatham, D., V.N.M. Rao and K.N. Shanmugavelu, 1983. Enzyme inactivity as a limiting factor in the rooting phenomenon of Jasminum auriculatum Vahl. Cv. Parimullai. Ind. J. Plant Physiol., 26: 108-111.
Direct Link - Went, F.W., 1938. Specific factors other than auxin affecting growth and root formation. Plant Physiol., 13: 55-80.
Direct Link - Wiart, C., S. Mogana, S. Khalifah, M. Mahan and S. Ismail et al., 2004. Antimicrobial screening of plants used for traditional medicine in the state of Perak, Peninsular Malaysia. J. Fitotera, 75: 68-73.
CrossRefPubMedDirect Link - Wiesman, Z., J. Riov and E. Epstein, 1988. Comparison of movement and metabolism of indole-3-acetic acid and indole-3-butyric acid in mung bean cuttings. Physiol. Plant, 74: 556-560.
CrossRefDirect Link - Ofori, D.A., A.C. Newton, R.R.B. Leakey and J. Grace, 1996. Vegetative propagation of Milicia excelsa by leafy stem cuttings: Effects of auxin concentration, leaf area and rooting medium. For. Ecol. Manage., 84: 39-48.
CrossRefDirect Link








