Research Article
The Hepatoprotective Effect of β-Carotene Against Cadmium Toxicity in Rats
Department of Pharmacology, National Research Center, Egypt
I. M. Alhazza
Department of Zoology, College of Science, King Saud University, Saudi Arabia
Cadmium (Cd) is a heavy metal of great environmental and human health concern. It is implicated in many industrial uses, such as in electroplating, paints, dyestuffs and mining industry and it is now a major threat to man`s environment. In animals, cadmium was shown to be toxic to all tissues. It has been reported that (Cd) causes morphological and functional damage in hepatic (Horiguchi et al., 2000) and renal tissues (El-Sharaky et al., 2007), testicular necrosis (Lorico et al., 2002), morphological and biochemical changes in lungs and gastrointestinal tract (Weisman, 1998). After entering into organism through food or water, Cd binds to albumins and erythrocytes in the blood and then is transferred into tissues and organs, where it is bound to proteins of low molecular mass producing metallothioneins (George et al., 1996).
The molecular mechanism that may be responsible for the toxicity of Cd involves oxidative stress by disturbing the antioxidant defense system and by producing Reactive Oxygen Species (ROS) (Thijssen et al., 2007), which include hydrogen peroxide (H2O2), superoxide (O2•–G), hydroxyl radical (OH•–G).
Some vitamins play an efficacious protective role through direct or indirect mechanisms which interfere with the intestinal absorption of heavy metals by increasing urinary excretion (Pleasants et al., 1993). Vitamin A resulted in a marked reduction in CCl4- induced hepatic damage in mice (Hooser et al., 1994). The primary antioxidant of vitamin A is to scavenge single oxygen; because single oxygen reacts with lipids to form lipid hydroperoxides and the removal of single oxygen prevents lipid peroxidation (Schafer et al., 2002). Vitmin A is obtained from β-carotene and it belongs to retinoid family. It exists in several chemical forms, such as retinoic acid and retinal. Interconversions between these chemical forms readily occur in the body. Administration of vitmin. A was observed to produce a significant regain in weight loss, growth depression and haematotoxicity resulted from exposure to gasoline (Uboh et al., 2008). Carotenoids are a group of phytochemicals that are responsible of different colors of the foods. They are recognized in the prevention of cancer (Rao et al., 1999), cardiovascular disease (Hung et al., 2008), osteoporosis (Rao, 2006) and hypercholesterolemia (Fuhramn et al., 1997). It was reported that the nutritional antioxidant, β-carotene ameliorated oxidative stress and loss of cellular antioxidants result from treatment of rats with Cd (El-Missiry and Shalaby, 2000). The present study has been designed to assess the effects of Cd on liver functions and hydroperoxide level as well as the efficacy of β-carotene treatment alleviating Cd toxicity.
Animals
Male Wister rats weighting 180-200 g were obtained from animal house, Faculty of Pharmacy, King Saud University. The rats were maintained under standard laboratory condition (12 h light, temperature 23±1°C). They fed dry ration ad lib. This study was conducted in Zoology Department, Faculty of Science, King Saud University, Saudi Arabia.
Chemicals
Cadmium as cadmium chloride (CdCl2) and β-carotene were purchased from Merck and Sigma, Germany, respectively.
Experimental Design
Seventy two male rats were randomly divided into three groups of twenty-four animals each. The groups were: group 1 served as control and received the equivalent volume of vehicle (corn oil), group 2 injected s.c. with 2.5 mg kg-1 body weight of cadmium chloride (Zhang and Jia, 2007) in 0.1 mL saline and group 3 injected intramuscularly with 10 mg kg-1 b.wt. of β-carotene (Salvadori et al., 1992) in 0.1 corn oil along with 2.5 mg kg-1 Cd (sc). The injection of β-carotene was 1 h prior to Cd injection in group 3. The injections were 3 times weekly for 6 weeks.
Collection of Samples
Eight blood samples were obtained from the eye of the rats in each group after 2, 4 and 6 weeks. The plasma were separated by centrifugation and stored frozen (-20°C) until further analysis. Eight animals from each group were sacrificed at the previous time intervals and liver samples were kept frozen.
Analytical Procedure
Blood hydroperoxide levels were evaluated using free radical analytical system (IRAM, PARMA, Italy). The test is a colorimetric test that takes advantage of the ability of hydroperoxides to generate free radicals after reacting with some transitional metals. When buffered chromogenic substance is added, a colored complex appears. The complex can be measured by a spectrophotometer.
Cd contents in liver cells were measured using atomic absorption spectrophotometer. Certified reference solutions were used to generate standard curve.
ALT, AST and ALP, activities were determined by kinetic methods using bio Merieux kits, France. Moreover, cholesterol concentration in plasma was evaluated colorimetrically.
Statistical Analysis
The data were expressed as Mean±SE of 8 rats in each group and statistical difference between groups were assessed by Student t-test.
Cd administered subcutaneously resulted in a significant increase (p<0.01) of hydroperoxide level by 50, 70 and 83% compared to control at 2, 4 and 6 weeks post-treatment (Table 1). The hydroperoxide level in β-carotene+Cd group increased significantly (p<0.01) by 27, 30 and 42%, respectively at the previous time intervals. It is evident that hydroperoxide level of rats treated with β-carotene and Cd was significantly (p<0.01) less than that of rats treated with Cd alone at the three time intervals. Hepatic Cd concentration (Table 1) showed a significant increase (p<0.01) in the treatment groups at all time intervals. Cd concentration of β-carotene+Cd group was less than in Cd group.
AST activity was significantly (p<0.01) increased in rats treated with Cd (Table 2) by 299, 360 and 437% at 2, 4 and 6 weeks post treatment.
AST activity increased in the rats given β-carotene+Cd significantly (p<0.01) by 173,132 and 153 % at previous intervals. The activity of the enzyme of rats treated with β-carotene+Cd was significantly (p<0.01) less than that of rats given Cd alone at all time intervals. A significant increase (p<0.01) in ALT activity was observed in the rats given Cd alone (Table 2) after 4 and 6 weeks from treatment. The activity of ALT appears normal in β-carotene+Cd group. Moreover, the values of β-carotene+Cd group were significantly less than those of Cd group.
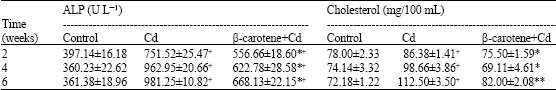
Alkaline phosphatase activity (Table 3) showed a significant increase (p<0.01) in animals treated with Cd or with β-carotene+Cd at all times intervals. The activity of the enzyme in β-carotene+Cd group was significantly less than that of Cd group. Moreover, cholesterol level of the rats treated with Cd elevated significantly (p<0.01) at all times intervals, while it increased significantly in the rats given β-carotene+Cd at 6 weeks, post treatment only.
| Table 1: | Effect of cadmium and β-carotene on blood hydroperoxide level and hepatic cadmium concentration |
 | |
| Values are Means±SE (n = 8), +Significantly different from control (p<0.01), *Significantly different from Cd group (p<0.01) | |
| Table 2: | Effect of cadmium and β-carotene on blood AST and ALT activities |
 | |
Values are Means±SE (n = 8), +Significantly different from control (p<0.01), *Significantly different from Cd group (p<0.01) | |
| Table 3: | Effect of cadmium and β-carotene on blood alkaline phosphatase and cholesterol concentration |
 | |
| Values are Means±SE (n = 8), +Significantly different from control (p<0.01), *Significantly different from Cd group (p<0.01) | |
Cadmium induces a broad spectrum of toxicological effects and biochemical dysfunctions constituting a serious hazard to health. The observed increase in hydroperoxide level of blood in rats treated with Cd may indicate oxidative stress which affects liver organelles. Cd participates in oxidation reactions associated with generation of some reactive species which interact with membrane lipids of liver cells to produce lipid peroxides (El-Demerdash et al., 2004). Reactive oxygen species such as superoxide radical and hydroxyl radical provoke sever changes at cellular level leading to cell death because of their extreme reactivity. They attack essential cell constituents such as proteins, lipids and nuclei acids. Also, they induce peroxidation of fatty acids (Stadtman and Berlett, 1997). Lipid peroxides that accumulate due to peroxidation of lipids are known to be harmful to cells and tissues (Linden et al., 2008). It was reported that subcutaneous administration of Cd to mice resulted in pronounced increase of lipid peroxidation in the liver accompanied by a depletion of hepatic GSH and catalase (Eybe et al., 2006). Liver has shown to be target organ of acute Cd toxicity. The relation between the hepatic oxidative damage and elevation of the relevant serum enzymes in Cd toxicity is well documented (Vicente-Sanchez et al., 2008). The observed increase in activities of plasma ALT, AST and ALP is likely due to lipid peroxidation of biomembranes which causes leakage of cellular components (Iguchi et al., 1993). Exposure of hepatocytes to Cd stimulates cellular production of H2O2 which affect permeability barrier of plasma membrane (Koizumi et al., 1996). It seems that, the increase in the liver enzymes of the present study may be due to accumulation of Cd in hepatic tissue which enhances formation of lipid peroxidation.
We observed a significant increase of the cholesterol level in plasma of Cd-treated rats when compared with control rats. Generally, heavy metals induce change in the activity of hydroxyl 3-methylglutaryl- Coenzyme A reductase, which alter cholesterol as well as lipid metabolisms. Cd exposure markedly increased the levels of inflammatory cytokines (Kayama et al., 1995), which might be responsible for the observed hypercholesterolemia In Cd-exposed rats. Murugavel and Pari (2007) reported that the increased levels of low density lipoprotein with decreased high density lipoprotein in Cd toxicity reflected the abnormalities in lipoprotein metabolism which may result in high levels cholesterol. Cd has been demonstrated to stimulate hypercholesterolemia and free radicals production (Kayama et al., 1995) that have been postulated to contribute to atherogenesis by variety of mechanisms including the inactivation of nitric oxide, the oxidation of low density lipoprotein and stimulation of cytokines (Harrison and Ohara, 1995).
Administration of biological antioxidants such as β-carotene suppresses lipid peroxidation elevation in the plasma of rats (Frusho et al., 2002) and mouse tissues (Iyama et al., 1996). β-carotene is a quencher of free radicals induced oxidative stress (Vardi et al., 2008). Carotenoids quench singlet oxygen primarily by physical mechanism, in which excess energy of single oxygen is transferred to carotenoids and then they relaxes into ground state, as a result carotenoids offer to protect against further oxygen radical and lipid peroxidation (Mc Nulty et al., 2008). In present investigation, β- carotene injections to rats given cadmium has a protective effect on the elevation of hydroperoxide level, which may indicate that β-carotene reduce the oxidative stress produced by Cd. Supplementation of the usual daily diet with β-carotene was shown to increase the plasma antioxidant capacity (Meydani et al., 1994). β-carotene improves the activities of antioxidative enzymes following oxidative stress (Zamora et al., 1991). Because β-carotene is lipophilic substance, it exerts its action in hydrophobic environment such as the lipid core of membranes. Thus, it is anticipated that natural β- carotene, a chain breaking antioxidants, can contribute to protect cell membranes from lipid peroxidation induced by Cd (El-Missiry and Shalaby, 2000).
In present experiment, pretreatment with β-carotene reduced Cd-induced elevation of ALT, AST and ALP. This may be due to lowering Cd levels in liver tissue by β-carotene. Some vitamins play an efficacious protective role through direct and indirect mechanisms, which interfere with the intestinal absorption of heavy metals by increasing urinary excretion (Pleasants et al., 1993). El-Demerdash et al. (2004) reported that the treatment of rats with β-carotene minimized the toxic effects of CdCl2. β-carotene can acts as antioxidant and anti-atherosclerotic agents (Stahl and Sies, 1996). The present experiment demonstrated that the values of plasma cholesterol of rats given β-carotene and Cd were less than of Cd group. From the present results, it can be concluded that use of β-carotene has the capability to alleviate many of harmful effects of CdCl2.
This study was supported by Research Center, Project No. Zoo /2007 /58, College of Science, King Saud University.