Research Article
Molecular Modelling Analysis of the Metabolism of Venlafaxine
Discipline of Biomedical Science, School ofMedical Sciences, Faculty of Medicine, Cumberland Campus, C42, The University of Sydney, Lidcombe, NSW, Australia
Depression is one of the most common neuropsychiatric conditions affecting more than 120 million people worldwide (Willie et al., 2005). It is the leading cause of disability in the U.S. Depression is assumed to be involved in 50% of all suicide attempts in the Western world and 25% of severe depressed patients attempt suicide at least once in life (Uges and Conemans, 2000). Venlafaxine (1,2-dimethylamino)-1-(4-methoxyphenyl) ethyl-cyclohexanol hydrochloride; VEN) is a new phenylethylamine bicyclic antidepressant with distinctly different neuropharmacologic profile than existing antidepressants including tricyclic compounds. Its antidepressant activity is due to inhibition of neuronal uptake of norepinephrine, serotonin and to a lesser extent dopamine (Rogers and Schmidt, 2006). VEN lacks the adverse side effects common with tricyclic antidepressants (Ellingrod and Perry, 1994; Veefkind et al., 2000).
VEN is extensively metabolized in the human liver catalysed primarily by CYP2D6 and to a lesser extent by CYP3A4, CYP2C19 and CYP1A2, producing several metabolites. One of the metabolites, namely O-Desmethyl Venlafaxine (ODV), has activity profile similar to that of VEN (Hartter, 2006). Four other metabolites are N-desmethylvenlafaxine (NDV), N, N-desdimethylvalafaxine (NNDDV), N-desmethyl-O-desmethylvalafaxine (NDODV) and N, N-desdimethyl-O-desmethylvalafaxine (NNDDODV).
In this study, molecular modelling analyses have been carried out using the program Spartan ’02 (Spartan, 2002) to provide information on the relative toxicity of VEN and its metabolites and to explain why VEN has fewer side effects than tricyclic antidepressants.
COMPUTATIONAL METHODS
The geometries of VEN and its metabolites ODV, NDV, NNDDV, NDODV (Fig. 1) and NNDDODV have been optimised based on molecular mechanics, semi-empirical and DFT calculations, using the molecular modelling program Spartan ’02.
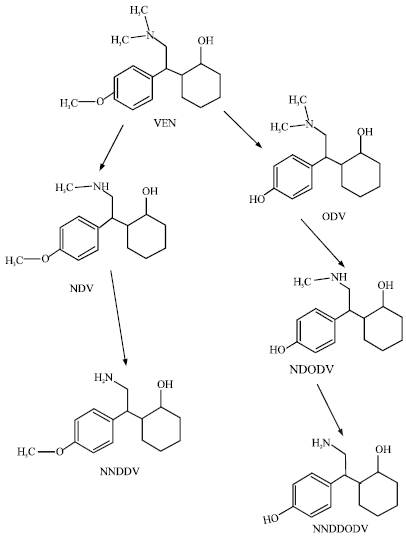 | |
| Fig. 1: | Metabolic pathways for VEN in rats and humans based on Shams et al. (2006) |
Molecular mechanics calculations were carried out using MMFF force field. Semi-empirical calculations were carried out using the routine PM3. DFT (Density Functional Theory) calculations were carried at B3LYP/6-31G* level. In optimization calculations, a RMS gradient of 0.001 was set as the terminating condition. For the optimised structures, single point calculations were carried out to give heat of formation, enthalpy, entropy, free energy, dipole moment, solvation energy, energies for HOMO (Hight Occupied Molecular Orbital) and LUMO (Lowest Occupied Molecular Orbital). The order of calculations: molecular mechanics followed by semi-empirical followed by DFT ensured that the structure was not embedded in a local minimum. To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structure corresponding to the global minimum or close to that could ultimately be reached in all cases. Although RMS gradient of 0.001 may not be sufficiently low for vibrational analysis, it is believed to be sufficient for calculations associated with electronic energy levels.
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, surface area, volume, dipole moment and energies of HOMO and LUMO as per both PM3 and DFT calculations for VEN and its metabolites ODV, NDV, NNDDV, NDODV and NNDDODV.
| Table 1: | Calculated thermodynamic and other parameters of ETV and its metabolites |
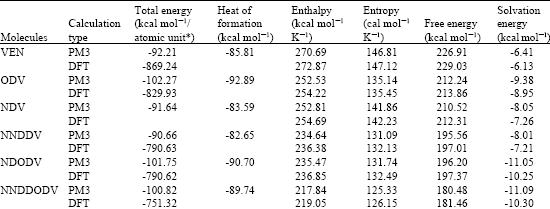 | |
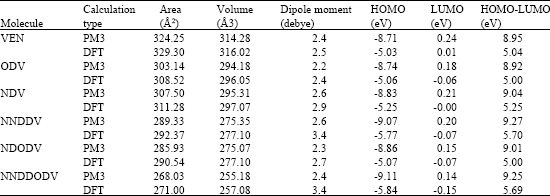 | |
| * In atomic units from DFT calculations | |
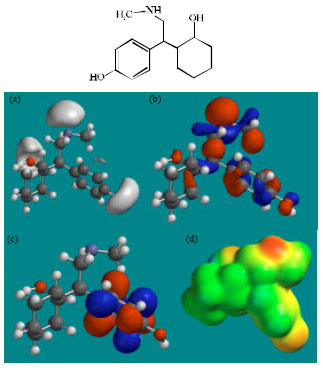
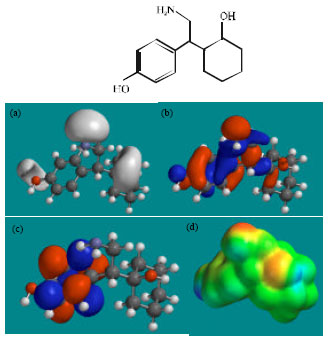
Fig. 2-7 give the regions of negative electrostatic potential (greyish-white envelopes) in (a), HOMOs (where red indicates HOMOs with high electron density) in (b), LUMOs in (c) and density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) in (d) as applied to optimised structures of VEN and its metabolites ODV, NDV, NNDDV, NDODV and NNDDODV.
The LUMO-HOMO energy differences for VEN and its metabolites from DFT calculations are found to range from 5.0 to 5.7 eV, indicating that the compounds would all be kinetically inert.
In the case of VEN, ODV, NDV, NNDDV, NDODV, NNDDODV, the electrostatic potential is found to be more negative around oxygen and nitrogen atoms and around the phenyl ring, indicating that the positions may be subject to electrophilic attack.
In the case of VEN, ODV, NDV, NNDDV, NDODV and NNDDODV, the HOMOs with high electron density are found to be slightly more localised than the LUMOs.
The overlap of HOMO with high electron density and region of negative electrostatic potential at some positions, gives further support to the idea that the positions may be subject to electrophilic attack.
The molecular surfaces of VEN and all its metabolites are found to possess significant amounts of electron-rich (yellow and red) and neutral (green) regions so that they may be subject to electrophilic and lyophilic attacks. The molecular surface of none of the compounds is found to abound in electron-deficient (blue) regions so that the compounds may not be subject to nucleophilic attacks such as those due to glutathione and nucleobases in DNA. This means that VEN and its metabolites may not cause DNA damage and oxidative stress associated with glutathione depletion.
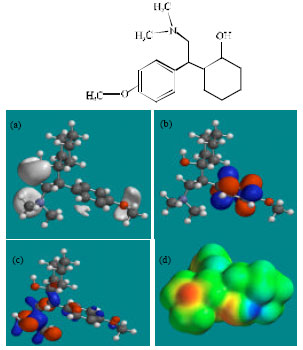 | |
| Fig. 2: | Structure of VEN giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
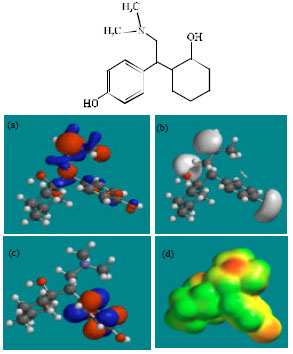 | |
| Fig. 3: | Structure of ODV giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
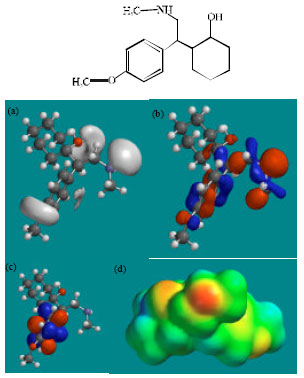 | |
| Fig. 4: | Structure of NDV giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
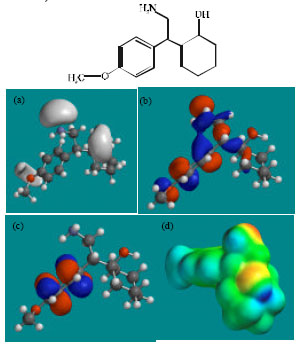 | |
| Fig. 5: | Structure of NNDDV giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
 | |
| Fig. 6: | Structure of NDODV giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
 | |
| Fig. 7: | Structure of NNDDODV giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
Venlafaxine (VEN) is a new phenylethylamine bicyclic antidepressant whose activity is due to inhibition of neuronal uptake of norepinephrine, serotonin and dopamine. It has lack side effects than tricyclic antidepressants. Molecular modelling analyses based on molecular mechanics, semi-empirical (PM3) and DFT (at B3LYP/6-31G* level) calculations show that VEN and its major metabolites have high LUMO-HOMO energy differences ranging from 5.0 to 5.7 eV, indicating that the compounds would all be kinetically inert. The molecular surface of neither nor any of its metabolites is found to abound in electron-deficient regions so that the compounds may not react with glutathione and nucleobases in DNA. This means that the compounds may not induce cellular toxicity associated with glutathione depletion and DNA damage associated with oxidation of nucleobases in DNA.
Fazlul Huq is grateful to the Discipline of Biomedical Science, School of Medical Sciences. The University of Sydney for the time release from teaching.