ABSTRACT
In this study we taken two samples of kaolin, Iron Kaolin (IK) and White Kaolin (WK) of Tamazert in El-Milia and we have execute a study of the distribution of the different mineralogical phases, as well as the determination of the chemical composition in the different slices of grains concerning the two samples. The analyses by X-ray Diffraction show that the analyzed samples constituted mainly of kaolin with similar content, of Mica (Muscovite-Illite), as well as of Quartz in heavy proportion in the two samples (IK and WK). The existence of some elements as the Goethite and Hematite, as well as the Anatase and the Rutile, generate the disorder in the chemical composition of the Kaolinite. These modifications of structural order are for consequence of change of the quality raw materials of the kaolin extracted from the Tamazert deposit. We remark that these variations are more important for the Iron Kaolin (IK); they are explaining by the presence of more elevated content in silica. However, the increase of these contents is less important for White Kaolin (WK).
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2006.1020.1027
URL: https://scialert.net/abstract/?doi=jas.2006.1020.1027
INTRODUCTION
The kaolin is one of many types of clay and commonly known as clay of China or Clay of paper. It is Silica hydrated with a composition approximately 46% SiO2, 40% Al2O3 and 14% H2O. The general formula is Al2 (OH) 4Si2O5 (US Geological Survey, 2001). It is composed of a two-layer arrangement, where the geometries around Aluminum and Silicon central atoms are octahedral and tetrahedral, respectively. The structure of kaolinite is showing in Fig. 1.
Kaolinite is one of the most abundant minerals in the Earth crust. Some of its properties (viz. color, particle size, morphology surface chemistry and charge) are exploited by the papermaking, paint and pharmaceutical industries, among others (Mohammad and Al-Trawneh, 2004).
The layer of TAMAZERT is knowing since 1923, was the subject of a traditional exploitation since 1932, then industrial but reduced, the kaolin not treated delivered as raw matter to the ceramics nationals units of productions, as unit of GUELMA, MILA and MAGHNIA. (Some Algerians Cities) (Laraba, 2001).
In 1975 was prevision of the ore utilization treated, the ore is sandy type, with presence of Mica (Muscovite) and Quartz and Oxide of Iron.
Currently, the unit of kaolin of El-Milia, encounter some problems bound to the fluctuations of the quality of the raw material, by the frequent apparition of elevated content in hematite (Fe2O3) and of dismissals rich in Silica, (ENOF, 1999).
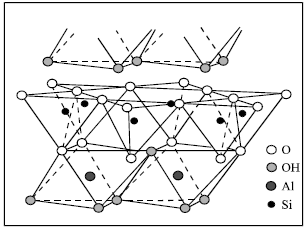 | |
| Fig. 1: | Structure of a kaolinite layer (Modified from USGS) |
Therefore, to solve these problems and especially the problems concerning the quality of the raw material dragging in the production of kaolin, it is necessary to realize a mineralogical analysis of the samples of this kaolin. We know that small quantities as low as 0.4% of ferric Iron may be sufficient enough to impart color to the kaolin deposit (Ambikadevi and Lalithambika, 1999) the removal of these associated impurities becomes imperative to make the material value-added. For this reason we have taken two samples of kaolin from Tamazert deposit in El-Milia and we have realize a study of the distribution of the different mineralogical phases, as well as, the determination of the chemical composition in different slices of grains concerning the two types of samples; Iron Kaolin (IK) and White Kaolin (WK); this last, let's to know the very important impurities generate the tow kaolin types. Finally this study can help the other researchers in the enrichment domain to ameliorate the chain of present treatment of the society of kaolin in Tamazert and how they can remove maximally the Oxide of Iron (Fe2O3) and other impurities from this kaolin.
GEOGRAPHICAL SITUATION OF TAMAZERT
The kaolin of Tamazert deposit is situated in a region of NE of Algeria, being part of the Kabylie of Collo (Fig. 2). It is limit in the west by the city of Jijel (45 km flight of bird), to the North by the Mediterranean Sea, to the East by the city of Collo and to the South by the city of Sidi-Maarouf.
The layer is situated to 17 km in the North of El-Milia, it is located between the longitude 6° 5" and boreal latitude 36°5''. The region makes part of a mountainous massif in the center of the mountains of Kabylie of Collo. The point culminating of the region is in Aine-Sra has 701 m; the relief's are relatively abrupt.
GEOLOGY AND SETTING
The layer of Tamazert is characterized by the existence of two facies: the first is sandy kaolin formed by superficial sedimentary layers, which cover by the second facies of kaolinitic gneiss situated in depth (ENOF, 1999-2000).This kaolinitic massive generally forms itself by a transformation of minerals as the silica (SiO2), what explains its abundance in the layer.
The three kaolin bodies (central body, Sidi Kader body and north body) are grouped in a space squared of 2000 m2 of rating; they are situated in a powerful gneissetic formation of general orientation. NE-WS has middle poundage directed toward the SO, this formation is Flanked on its pour South by a set of micaschistic and garnets.
The whole deposit has result from the change of a gneissetic facies that localizes in the superior part of the relief, the gneiss of kaolinite can also contain some intercalations of Quartzites, these last; generally constitute the saturates of the kaolin.
In the sector situated to the East of Sidi-Kader, contrarily that is observed to the Oust, the gneiss plunge to the Southbound it is possible that we have to make two compartments separated by a faille, as well as can lets support of the mylanitises rocks observed on the south flank of Sidi Kader.
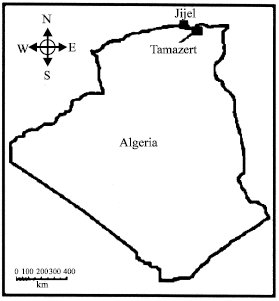 | |
| Fig. 2: | Geographical situation of Tamazert |
Geologically, the deposit of kaolin of Tamazert of El-Milia is a layer that is constituted by a hydrothermal change of Feldspars rich in potassium.
PETROGRAPHIC DESCRIPTION
The micaschisteous formation: These formations are the schist rocks have well marked, the phyletic levels that materializes the surfaces of schistsites is visible to our eyes. (Touahri, 1998)
These rocks show on the surface in the Southwest of layer (El-Khmis and Azarrar) has the blackish color in surface and rich in phyletic minerals, these rocks show sometimes the folds within these formations come in of the quartzite.
The gneissetic formation: The most important basset of this formation appear to the level of the place called; Limarbet Salah and all along the road that led to Ouled Boufaha to the south of The basset, these are the massive rocks, with dark color, these gneiss don't show the foliation, seen their weak proportion of mica.
They are constituted by the middle grains sizes. By part; these gneiss contain some eyes Quartzo-Feldespathic constituted by mono crystals-align of feldspar in the same way to the plan of foliation.
MATERIALS AND METHODS
In this study we have take two methods to analyses our two simples; White kaolin and Iron kaolin, this analysis has been realized in the laboratories of the company of Kaolin Company of kaolin Soalka, El-Milia-2001, Algeria.
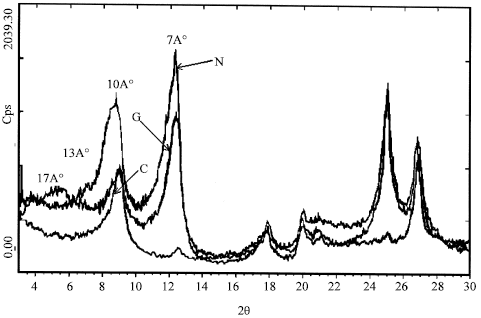 | |
| Fig. 3: | X diagram of kaolin powder (WK). N: X initially diagram. G: X diagram with the ethylene glycol. C: X diagram after heating at 500°C |
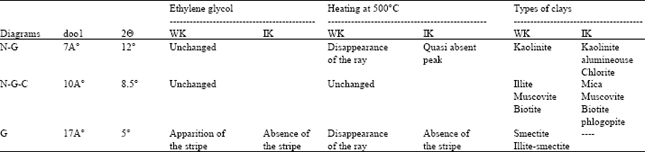
| Table 1: | Characterization by X-Ray Diffraction of kaolins WK and IK |
 | |
Analyses by X-rays diffraction (XRD): The diagrams of X-Ray diffraction have been obtained with a PHILIPS Diffractometer, with a carry sample and a source delivering a radiance of K stripe of the copper. The analysis by X-ray Diffraction (XRD) were realized on the fine powder of the raw samples of iron kaolin (IK) and white kaolin (WK) and on the powders obtained by grinding of the following fractions: > 100; 100-80; 80-40; 40-25; 25-10; >10 μm taken from the suspensions of the two samples (IK) and (WK).
Results and Discussion about (XRD): The X Diffractogram powders (Fig. 3) present a set of three diagrams corresponding at each physico-chemical conditioning appropriate the (WK) sample. In the origin position we gets the diagram noted N, then after absorption of a quantity given of ethylene glycol, we get the X diagram notes G, followed from the X diagram, we get also the X diagram noted G, followed from the X diagram noted C achieves after the heating at 500°C during 3 h. We distinguishes to the total 4 mains peaks situated respectively on the angles of diffraction 2θ about 8,5°, 12°, 25° and 26,75 ° on the N diagram.
The Table 1 summarizes the different observable modifications, which are due to the conditioning of the kaolin samples, established on the 3 types of. N, G and C Diffractograms.
The interpretation of the different effects, chemical and thermal on the samples of kaolin (WK) and IK) succeeded to the following commentaries:
The existence of a peak in 2θ equal to 12°A, corresponding to a mineralogical phase, of structure lamellar leaflets noted on the N and G diagrams and the disappearance of this same peak on the C diagram, after heating at 500°C, translate the typical behavior of the kaolinite. Indeed, the distance d001 between leaflets, take according to the normal of the leaflets plan, estimated to 7°A stays constant for the two samples took at the nature condition (N), or when they are put to the contact of a solution of ethylene glycol. Therefore, when the sample has heating in 500°C, we find the total disappearance of the stripe at 7°A in the case of the White Kaolin sample (WK), then, that it subsists a fragment of this very weak intensity peak for the Iron Kaolin sample (IK), attribute to the existence of weak proportion of chlorites in this case.
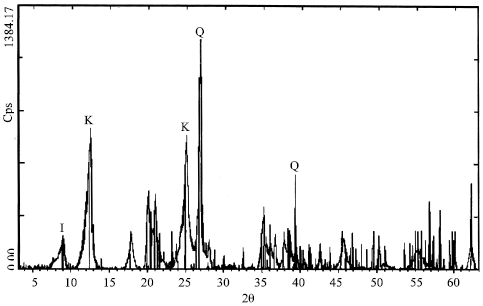 | |
| Fig. 4: | X diffractogram of the fraction < 10 μm of the White Kaolin (WK) Q: Quartz, K: Kaolinite, I: Illite |
It demonstrates that the crystalline phase of the clay becomes suddenly amorphous by the apparition of a disorder in the leaflets; the formation equilibrates of the amorphous phase is expressed by the following equation:
The process is irreversible; N, G and C, the cooling of the sample won't enter the reappearance of the peak at 7°A.
The peak in 8,5°A on the N, G and C diagrams is the typical behavior of the Illite and the Muscovite and the Biotite is more present in the sample of IK.
In the present case, the width of the peak let suppose the presence of Illite than of Mica. The mineralogical phase is characterized by a structure with leaflets of distance reticular valued d001 at 10°A; belonging to the family of Mica, Illite and Muscovite.
The peak in 5°A that appears on the G diagram and disappears on the C diagram, with the huskiness of the peak of the Illite (10°A) on the N diagram, is explained by the behavior of Smectite or Illite-Semectite (Fig. 4) in weak quantity, existing especially in the White Kaolin sample (WK).
This peak corresponding to the crystalline structure proper to the clays of type inflating and expansive characterized of this fact, by a distance inter-leaflet very important estimated to 17°A. We will remark the persistence of a weak peak in 7°A on the C diagram that confirms the presence of the Smectite.
The absence of the peak in 17°A however is remarked on the X diffractogram of powder, not represented here in this case of White Kaolin sample.
According to these results of the analysis by X-ray Diffraction (XRD), the mineralogical compositions of the two kaolin samples (WK and IK) are approximately the same and more especially for the fine grains. The presence of the kaolinite is discovered in the two samples (WK and (IK) in the fractions inferior to 100 μm.
The content of kaolinite is practically the same; the presence of quartz signaled in the totality of the fractions with important proportions, which decrease toward the fine. The Albite that is also a silicate coming at the origin of hyper-acidic rocks existing a lot more in the fine fractions (25-40 μm) and (10-25 μm), white the Anorthoclase.
These same minerals also discovered in the iron kaolin sample, with the absence of the Albite in all fractions inferior to 100 μm. Contrarily to this, we remark the presence of the Anorthoclase in the fractions inferior to 80 μms.
The Muscovite and Quartz generally has been retorted in all grains, whatever their dimension, for the two types of kaolin samples.
Otherwise, the Goethite is the mineralogical phase that more characterizes in the Iron kaolin sample. This last contains in the fine grains, the Oxide of Iron in the hydrated state FeO (OH) with an elevated content (Fig. 5) in the very fine fraction < 10 μm.
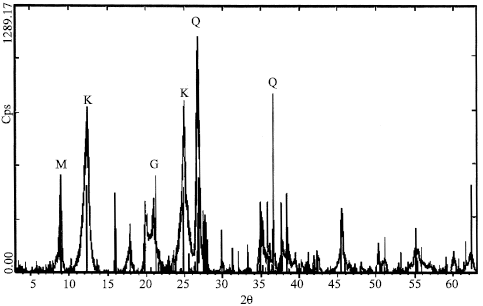 | |
| Fig. 5: | X Diffractogram of the fraction <0.1 mm of the Kaolin (IK) Q: Quartz, K: Kaolinite, G: Goethitem, M: Muscovite |
CHEMICAL ANALYSIS BY X-RAY FLUORESCENCE (XRF)
Chemical analysis by X-ray Fluorescence of the different slices of the Iron kaolin (IK) and white kaolin (WK) has been achieved with a PHILIPS spectrophotometer, for this reason we taken a quantity about 1.2 g of dry sample and we mixed with 6 g of fondant and 7 μL of the solution of Lithium Bromide in a pot in glass. The sample has putted in a crucible with 1050°C. The product of fusion is poured automatically in a second crucible in Platinum, then we let the sample get cold to be a pearl; this last is submitted to the analysis of X-Ray Fluorescence.
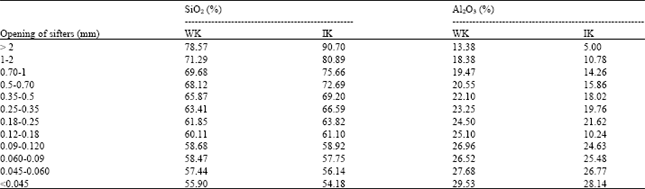
Result and discussion about X-rays (XRF): The results are represented in the Table 2 and 3, respectively, for the WK and the IK, According to this results, we can see the variations (Fig. 6) of the contents of the principal Oxides, such as the Silica (SiO2) and the Alumina (Al2O3) of the tow samples. The corresponding representative tables shows an increase of the contents of silica, in the same time a reduction of the Alumina contents according to the size of grains.
We remark that these variations in (Table 3) are more important for the iron kaolin (IK); they are explained by the presence of more elevated contents in Silica. But the increase of these contents is less important for the WK.
The variations of the contents in SiO2 according to the contents in Al2O3 of the sample of IK present a reduction of the contents in silica when there is an increase of the contents in Alumina and the graph of variation (Fig. 6) present a linearity as the report of proportionality global SiO2/Al2O2 is constant and has estimated about 1.5817.
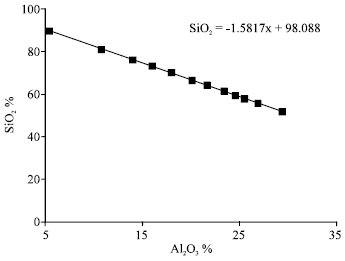 | |
| Fig. 6: | Graph of correlation between the variations of the contents in SiO2 and in Al2O3 of the IK |
These results are in agreement with the enrichment of the fractions of the stronger dimension of particles, by mineralogical phases of more elevated contents in Silica and the weak content in Alumina, as Quartz and Muscovite. On the other hand, we note enrichment very important for the fine mineralogical phases, containing some contents less raise in Silica and correlatively of the contents more raised in Alumina, as Kaolinite, or various clays, especially characterized by very distinct physico-chemical properties of those Kaolinites.
Except, the Silica and Alumina, of other minor elements as the Hematite (Fe2O3), or the Goethite (FeO (OH)), the Oxide of Potassium (K2O) and (LOI), as well as others various elements have the state of traces, exist in the two Kaolin types, however with variable contents.
| Table 2: | Chemical composition by X fluorescence of the fine fractions of the WK |
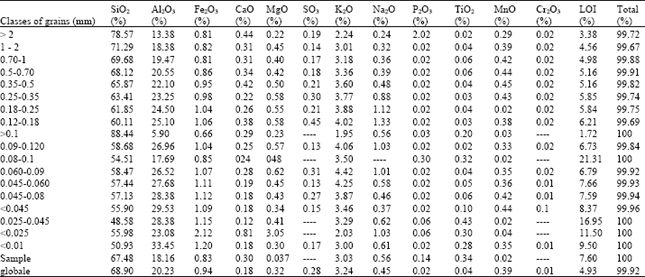 | |
| Table 3: | Contents of SiO2 and Al2O3 in the grains slices of the kaolin (IK) and (WK) |
 | |
| Table 4: | Chemical composition by X-Ray fluorescence of the fine fractions of the Iron Kaolin (IK) |
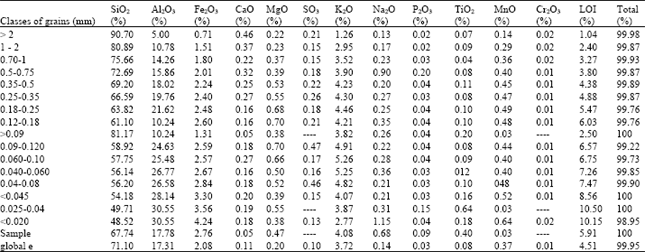 | |
The most often, are the Oxides of Titanium (TiO2) under its varied crystalline shapes, as the Anatase and the Rutile and the Oxides of Chromium (Cr2O3) and the Oxide of Manganese (MnO), that are present with very weak contents in the tow samples.
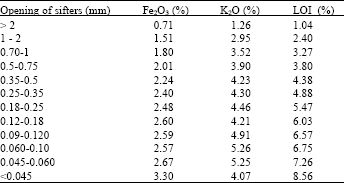
| Table 5: | Contents in Fe2O3, K2O and loss in fire of the iron kaolin (IK) |
 | |
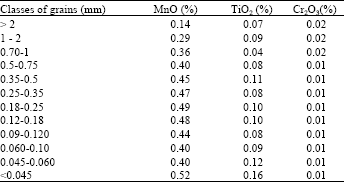
| Table 6: | Contents in TiO2, Cr2O3 and MnO of iron kaolin (IK) |
 | |
These minerals, in spite of their weak concentrations, these minerals, they are susceptible to generate some rheologics properties and physico-chemical that have a direct influence on the quality of the Kaolinite.
Otherwise, we have represented in (Table 5) the variations of the contents of Hematite, in Oxide of Potassium and in Loss on Ignition (LOI), in the different size of grains. For most graph of variation, this one show that the contents in Oxides and the Loss on ignition decrease when the dimensions of the particles increase between 0 and 2 mm.
These results has an agreement with the previous analyses by X-ray Diffraction particularly concerning the contents in Oxides of Iron (Fe2O3), which increase more in the fine fractions, with a maximal value in Fe2O3; about 3,30% in Iron kaolin.
The variations of the contents in Cr2O3 in (Table 6) present that variations are very weak and stay constants in the Iron kaolin sample (IK) and the Oxide of Titanium vary extensively and are less negligible compared to the preceding values. The contents in Oxides of Manganese are less weak compared to those of Oxides of Chromium and Titanium. The maximal value is about 0.52% corresponds to the fine grains (< 0.045 mm). The existence of content in Oxide of Manganese elevates the magnetic susceptibility and could make more efficient of treatment of the Iron kaolin with the magnetic separation technique.
CONCLUSIONS
The existence of some elements as the Goethite and Hematite, as well as the Anatase and the Rutile, generate the disorder in the chemical composition of the kaolinite.
These modifications of structural order are for consequence of change of the quality raw materials of the kaolin extracted from the deposit of Tamazert.
The research by X-rays Diffraction, show that the analyzed samples are constituted mainly of Kaolin with similar rates, of Mica (muscovite-Illite), as well as of Quartz in heavy proportion in the two types of samples (IK and WK).
According to these results of the analysis by X-Ray diffraction, the mineralogical compositions of the two kaolin samples (WK and IK) are approximately the same and more especially for the fine fractions. The presence of the Kaolinite is discovered for the two samples in the fractions inferior to 100 μm.
The content of Quartz increases in the fractions of dimension elevated (2-0.1 mm) that are the richest in Feldspar. The Loss on Ignition (LOI) and the concentration in Fe2O3 is the most elevated in the very fine fraction (< 0.1 mm).
We remark that variations are more important for the IK; they are explained by the presence of more elevated content in Silica. But the increase of these contents is less important for the WK.
The existence of content in oxide of manganese elevated magnetic susceptibility could make more efficient of treatment of the iron kaolin by a magnetic separation proceeds.
Finally result of the present study may be being able to give to the company some useful results and also can help some researchers how to treat this kaolin.
ACKNOWLEDGMENTS
I thank all responsible of the company of kaolin SOALKA. Algeria), I thank also, A. Bouzenzana and A. Amirech (Institute of mines, university of Tebessa Algeria), Sani A.M. Bachir, (China university of geosciences, Wuhan. China) for the helpful discussions during this study.
REFERENCES
- Ambikadevi, V.R. and M. Lalithambika, 2000. Effect of organic acids on ferric iron removal from iron-stained kaolinite. Applied Clay Sci., 16: 133-145.
Direct Link








