ABSTRACT
Combustion synthesis is a technique that material synthesis is accomplished by the propagation of a combustion front across a powder compact. Heterogeneous distribution of reactants is common when powders are mixed but the conventional modeling treatments for combustion synthesis thus far have only considered uniform systems. The occurrence of the composition heterogeneity may result in the local variation of the parameters, including density, heat capacity and thermal conductivity. Such a variation of the thermochemical/physical parameters further changes the reaction temperature and propagation velocity; and affects the global properties. Thus, it is important to investigate the influence of composition heterogeneity on the combustion synthesized products. This study addresses the composition heterogeneity effect on Ni + Al combustion synthesis numerically. The correlation of the composition heterogeneity with the combustion temperature and propagation velocity is also studied. In addition, the heterogeneous effect caused by each individual parameter is investigated.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2005.64.69
URL: https://scialert.net/abstract/?doi=jas.2005.64.69
INTRODUCTION
Combustion/Micropyretic Synthesis[1-13] is a technique that material synthesis is accomplished by the propagation of a combustion front across a sample. The heat to propagate a combustion front is obtained from the heat of formation of the synthesized compound. The unreacted portion in front of the combustion front is pre-heated by the released energy, which initializes further reaction. A continuous cycle of reaction, heat transfer and reaction initiation is reflected in the propagation of a combustion front. The advantages of combustion synthesis include the rapid net shape processing and clean products. In addition, the combustion synthesized products have been reported to possess better mechanical and physical properties.
Several numerical and analytical models of combustion synthesis of a composite system have been well developed[2-4,6-9]. Lakshmikantha and Sekhar firstly explored the numerical model that includes the consideration of melting of each constituent of the reactants and products and the considerations involving dilution and porosity[3,4]. The analytical modeling of the propagation of the combustion front in solid-solid reaction systems is also reported[4]. The analytical model gives good results when comparing the experimentally determined numbers and the numerically calculated values. These numerical and analytical analyses provide the knowledge of the reaction sequence during combustion synthesis reactions.
Heterogeneous distribution of reactants is common when powders are mixed but the conventional modeling treatments for combustion synthesis thus far have only considered uniform systems. The composition heterogeneity may result in the local variations of the parameters of the reactants. The composition heterogeneity may further change the processing corresponding parameters during combustion synthesis, including reaction temperature and propagation velocity; and affect the global properties. To acquire the homogeneous products and establish the correlation of composition heterogeneity with the various combustion parameters, it is important to study the influence of the composition heterogeneity on combustion synthesis. This study is aimed at such understanding. In this study, the correlation of the composition heterogeneity with the combustion temperature and propagation velocity is studied. In addition, the heterogeneous effect caused by each individual parameter is also investigated.
During the passage of a combustion front in the reaction, the energy equation for transient heat conduction, including the source term, containing heat release due to the exothermic combustion reaction is given as[3,4,7]:
| (1) |
Each symbol in the equation is explained in the nomenclature section. The reaction rate, Φ(T,η), in Eq.(1) is given as:
| (2) |
In this study, a numerical calculation for Eq. (1) was carried out with the assumption of the first order kinetics. In the Eq. (1), the energy required for heating the synthesized product from the initial temperature to the adiabatic combustion temperature is shown on the left-hand side. The terms on the right-hand side are the conduction heat transfer term, the surface heat loss parameter and the heat release due to the exothermic combustion reaction, respectively.
The middle-difference approximation and an enthalpy-temperature method coupled with Guass-Seidel iteration procedure are used to solve the equations of the combustion synthesis problems. A one-dimensional sample of 1 cm long is divided into 1201 nodes (regions) in the computational simulation. The choice of using 1 cm sample length is only for computational purpose and the simulation results are applicable to practical experimental conditions. Firstly, the proper initial and boundary conditions are used to initialize the temperatures and enthalpies at all nodes. The initial condition for ignition at the first point of the sample could be both by starting the solution with a predetermined initial temperature as well as by the incorporation of a surface heat flux. A predetermined initial temperature supposes a corresponding heat flux at the surface. Whenever an initial condition is used, the starting temperature is the adiabatic temperature[5,12]. In this study, the initial conditions in the simulation are taken as follows. (1) At the ignition node, at time t ≥ 0, the temperature is taken to be the adiabatic combustion temperature and reacted fraction is equal to one. (2) At the other nodes, at time t = 0, the temperatures are taken to be the same as the substrate temperature. The detailed numerical procedures have been introduced in the previous studies[6,7,9] and the results generated from this numerical model have given a good agreement with the experimental values[8].
Composition heterogeneity is assigned randomly by computer algorism and thus determines the distribution of the reactant composition. In this study, the Ni+Al combustion reaction with 20 at. % Ni Al diluent is chosen to illustrate the effect of composition heterogeneity. Depending on the chosen heterogeneity for each specimen, the molar ratio of Ni to Al for each node is calculated from the random numbers (fR(j)) and the assigned heterogeneity (Hetero). The sequence of the random numbers (-0.5~+0.5) generated from the computation is repeatedly used in the specimens with different heterogeneities to compare the magnitude of heterogeneity effect. The composition of each reactant before the beginning of the reaction can be expressed as follows:
| (3) |
| (4) |
where, XniAlo = 20 at.%, -0.5≤fR (j) ≤ +0.5, j = 1,2,......,1201 and Xi is the molar fraction of reactant I. In order to assure the sum of the compositions for all 1201 nodes equal to the stoichiometric values, the calculated Ni and Al composition of each node is adjusted so that the average value of each composition is equal to the original homogeneous value, i.e.,
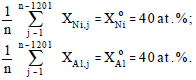 |
Table 1 shows the variations in reactant compositions for the different heterogeneities in this study. It is noted from Table 1 that a composition with 5% heterogeneity corresponds to 2% variation in composition, i.e., the Ni composition ranges from 39% to 41%. After the molar fractions of reactants are determined, the volume fraction of each reactant (Vreactant) is correspondingly calculated and the other parameters can be further determined at node j:
| (5) |
| (6) |
| (7) |
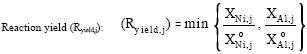 | (8) |
| Table 1: | The variation in Ni and Al reactant composition at different variations in composition for the Ni+Al combustion reaction with 20 at. % diluent |
 | |
In this study, the surface heat loss is taken as zero in the numerical calculation. Using Eqs. (2-7), the energy equation on nth time step at node j can be written as:
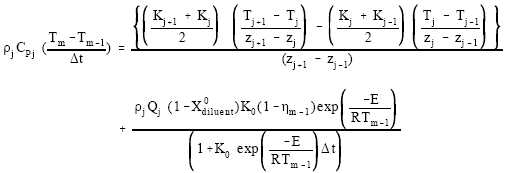 | (9) |
| Table 2: | The composition heterogeneous effects caused by individual parameter on the propagation velocity for the Ni + Al combustion reactions with 2 % variation in composition |
 | |
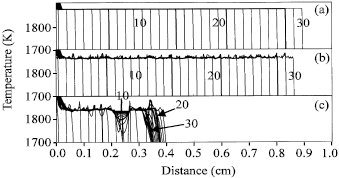 | |
| Fig. 1: | A plot of the combustion front temperature at various times along the NiAl specimen with 20 at.% diluent. The interval time between two consecutive time steps (profiles) is 0.0005 s. The figure only shows the first thirty time steps of the propagation sequence. The variations in reactant composition in (a), (b) and (c) are 0, 2 and 6%, respectively |
Using Eq. (9), the temperature, fraction reacted and enthalpy on nth time step at node j can be thus determined by the Guass Sidel iteration procedure.
The criterion used to ascertain whether the fraction reacted and enthalpies at each time level converge or not, is determined from the relative error criterion. Once the convergence criterion for every node is met, the enthalpy and fraction reacted of the last iteration in a time step are considered to be the corresponding final values. The calculations are normally performed from 500 to 2000 times, depending upon the calculated thermal parameters; to make all 1201 sets (nodes) meet the criterion for each time step.
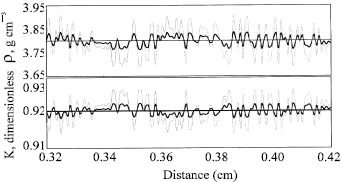 | |
| Fig. 2: | The plots of (a) density (ρ) and (b) thermal conductivity (K) along the NiAl reactants. The horizontal line denotes the values for the homogeneous specimens. The bold and gray curves denote the specimens with 2 and 6% variations in composition, respectively |
At least six hundred time steps are calculated to get the combustion front propagate one-centimeter length specimen completely. In this study, the higher pre-exponential factor (Ko) value, 8x108 1 s-1, is used to be capably of illustrating the variation of the propagation velocity for the NiAl synthesized product.
Figure 1 shows the temperature profiles of combustion fronts at various times along a NiAl specimen for 0, 2 and 6% variations in composition, respectively.
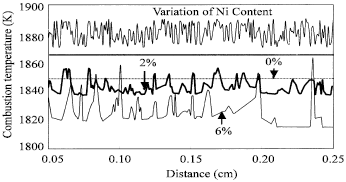 | |
| Fig. 3: | A plot of the combustion temperatures at different positions for the specimens with 0, 2 and 6% variations in composition, respectively. Also shown in the upper part of the figure is the variation of Ni composition |
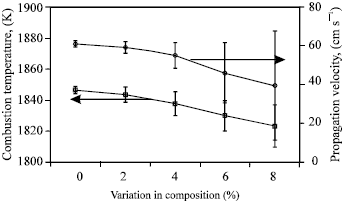 | |
| Fig. 4: | Plots of the average value and the standard deviation of the combustion temperature and propagation velocity for the NiAl stoichiometric combustion front with the different variations in composition |
The combustion front propagates at steady state for the homogeneous specimen (0% variation in composition), as shown in Fig. 1a. The combustion temperature (i.e., the highest reaction temperature) and the propagation velocity of a combustion front are noted to remain as constants during propagation. When non-homogeneous specimens are ignited, it is found that the temperature and the instantaneous propagation velocity are altered with the distance (Fig. 1b and c). The magnitudes of variations for temperature profiles are also increased with the increase in the variation in composition. The average combustion temperature is found to slightly decrease from 1846 K (for the homogeneous specimen) to 1844 K (for the specimen with 2% variation in composition). The average propagation velocity is also decreased from 61 to 59 cm s-1. When the variation in composition is further increased to 6%, the average values of the propagation velocity and the combustion temperature at the initial stage both are dramatically decreased to 46 cm s-1 and 1829 K, respectively. In addition, the propagation front is noted to stop half way (Fig. 1c).
Figure 2 shows the variations of the density and the thermal conductivity along the specimens with different variations in composition. The horizontal lines shown in Fig. 2 denote the values for the homogeneous specimens. The bold and gray curves denote the values for the specimens with 2 and 6% variations in composition, respectively. Since the compositions at each node are not changed in the homogeneous reaction, the thermal conductivity, density and exothermic heat of reaction are correspondingly kept as constant at all nodes. Thus, the combustion front is noted to propagate in a stable manner, as shown in Fig. 1a. As the heterogeneous specimen is ignited, the composition variation causes the molar ratio of Ni to Al to derivate from the stoichiometric ratio 1 : 1. This will lead to the reduction of the exothermic heat of the reaction in each reaction zone (node). In addition, Fig. 2 illustrates that the different reactant compositions alter the thermal conductivity and the density of each node. These variations result in the fluctuation in the temperature (Fig. 1b). As the variation in composition is further increased to a higher value, 6%, the temperature fluctuation is noted to significantly increase and combustion front even stops half way (Fig. 1c). Figure 2 also show that the magnitudes of variations of the density and thermal conductivity are increased as the variation in composition is increased. The larger variation of the temperature further results in the large-scale non-homogeneous microstructures and mechanical properties of the synthesized products.
To carefully investigate the variation of the temperature with the distance, the combustion temperature along the specimens with different variations in composition are calculated and plotted in Fig. 3. Figure 3 shows that the magnitude of the temperature variation is increased and the oscillatory frequency is decreased as the variation in composition is increased. Also shown in the figure is the variation of Ni content. It is noted that the combustion temperature does not strongly correlate with the variation of Ni content in time. However, as the variation of Ni content is accumulated to a certain level, the combustion temperature is noted to change periodically.
Figure 4 shows the plots of the combustion temperature and the propagation velocity for the NiAl stoichiometric combustion front with the different variations in composition. The figure clearly illustrates that the average combustion temperature and propagation velocity are decreased as the variation in composition is increased. The standard deviation (magnitude of variation) is also noted to increase with the heterogeneity. Generally a decrease in the propagation velocity and combustion temperature slows down the combustion reaction. It is inferred that the occurrence of the variation in composition correspondingly decreases the reactivity of the combustion reaction. Similar phenomena of the decreased propagation velocity and the reduced reactivity of the combustion reaction have also been reported when the diluent is present in combustion reactions[3,8]. Therefore, these observations suggest that the effects caused by the composition heterogeneity have the similar effects as adding diluents on combustion synthesis.
Each parameter is also assumed as independent of composition heterogeneity to illustrate the magnitude of the heterogeneous effects caused by each parameter on the propagation velocity. It is noted that as the exothermic heat of reaction is assumed as constant during the heterogeneous combustion synthesis, the calculated propagation velocities are enhanced up to 2.7% (Table 2). In addition, the propagation velocities are, respectively increased in 2.0 and 0.7% when the heat capacity and reactant density are taken as constant values. Therefore, it can be concluded that the reduction in the exothermic heat and the variation of heat capacity caused by the variation in composition are the major factors to reduce the propagation velocity.
This study addresses the composition heterogeneity effect on Ni + Al combustion synthesis numerically. It is found that the variation of reactant content directly influences the exothermic heat of the reaction and the reactant parameters locally, such as thermal conductivity, heat capacity and density. The combustion temperature and the propagation velocity of the combustion front are further altered. The calculated results show that the combustion temperature does not strongly correlate with the variation of Ni content in time. It is found that as the variation of Ni content is accumulated to a certain level, the combustion temperature is changed periodically. The combustion temperature of the Ni-Al combustion reaction is slightly decreased as the variation in composition is increased to 2%. A further increase in the variation in composition to 6%, the combustion temperature and propagation velocity are dramatically decreased and the combustion front even stops half way. It is also illustrated that an increase in the variation in composition increases the combustion temperature/propagation velocity variation. The calculated results show that the effects caused by the Ni composition heterogeneity are similar to that when adding diluents on combustion synthesis. In addition, the results also show that the reduction of the exothermic heat and the variation of heat capacity caused by the variation in composition are the major factors reducing the propagation velocity during Ni+Al heterogeneous combustion reaction.
ACKNOWLEDGMENT
The support from the National Science Council in Taiwan (Project No. NSC 93-2216-E-228-001) is acknowledged.
Nomenclature
| Cp | heat capacity of product (general form), kJ kg-1 K-1 |
| E | activation energy, kJ kg-1 |
| Hetero | composition heterogeneity, % |
| Ko | pre-exponential constant, (s-1 for zero order reaction) |
| Q | heat of reaction, kJ kg-1 |
| P | porosity, % |
| R | gas constant, kJ kg-1 K-1 |
| T | temperature, K |
| To | initial temperature, K |
| V | volume fraction, % |
| X | molar fraction, % |
| d | diameter of the specimen, m |
| fR(j) | random number at node j |
| h | surface heat transfer coefficient, J m-2 K-1 s-1 |
| t | time, s |
| z | dimensional coordinate, m |
| ρ | density, kg m-3 |
| κ | thermal conductivity (general form), kJ m-1 K-1 s |
| η | fraction reacted |
| Φ(Τ,_η)_ | reaction rate, 1 s-1 |
| superscript | |
| o | original |
| subscripts | |
| m | time step |
| j | node position |
REFERENCES
- Lin, C.N. and S.L. Chung, 2001. Combustion synthesis method for synthesis of aluminum nitride powder using aluminum containers. J. Mater. Res., 16: 3518-3525.
CrossRefDirect Link - Subramanian, V., M.G. Lakshmikantha and J.A. Sekhar, 1996. Modeling of sequential reactions during micropyretic synthesis. Metallurgical Mater. Trans. A, 27: 961-972.
Direct Link - Lakshmikantha, M.G. and J.A. Sekhar, 1993. An investigation of the effect of porosity and diluents on micropyretic synthesis. Metallurgical Mater. Trans. A, 24: 617-628.
CrossRefDirect Link - Lakshmikantha, M.G. and J.A. Sekhar, 1994. Analytical modeling of the propagation of a thermal reaction front in condensed systems. J. Am. Ceramic Soc., 77: 202-210.
Direct Link - Lakshmikantha, M.G., A. Bhattacharya and J.A. Sekhar, 1993. Numerical modeling of solidifaction combustion synthesis. Metallurgical Mater. Trans. A, 23: 23-34.
CrossRefDirect Link - Li, H.P., 2003. Numerical study of the second ignition for combustion synthesizing Ni-Al compounds. Metallurgical Mater. Trans. A, 34: 1969-1978.
CrossRefDirect Link - Li, H.P., 2003. The numerical simulation of the heterogeneous composition effect on the combustion synthesis of TiB2 compound. Acta Materialia, 51: 3213-3224.
Direct Link - Li, H.P., 2002. Investigation of propagation modes and temperature/velocity variation on unstable combustion synthesis. J. Mater. Res., 17: 3213-3221.
CrossRefDirect Link - Makino, A., 2001. Fundamental aspects of the heterogeneous flame in theself-propagating high-temperature synthesis (SHS) process. Prog. Energy Combust. Sci., 27: 1-74.
Direct Link - Merzhanov, A.G. and B.I. Khaikin, 1998. Theory of combustion waves in homogeneous media. Prog. Energy Combust. Sci., 14: 1-98.
CrossRefDirect Link - Munir, Z.A., 2000. Modeling and experimental studies on the effect of thermophysical properties on field-activated combustion synthesis reactions. Pure Applied Chem., 72: 2177-2186.
CrossRefDirect Link








