Research Article
Impact of Intensive Leaching on Some Clay Minerals of the Soils from Al Hassa Oasis, Saudi Arabia
Department of Soil and Water, College of Agriculture and Food Sciences, king Faisal University, Al-Hassa, Kingdom of Saudi Arabia
Weathering of minerals could release considerable cations to soil solution, some of them are leached from soil (Rhoads, 1967; Al-Barrak, 2001). The remaining cations react to form the new minerals, e.g. Kaolinite, smectite and chlorite (Alperovitch and Rohades, 1975). Weathering and dissolution rate of different soluble minerals such as gypsum and to less extend calcium carbonate showed different pattern (Al-Barrak, 2001; Al-Hawas, 1998).
These differences mainly attributed to texture, quantities of soluble minerals, and type of clay minerals present in soil. The mechanism of various clay minerals on releasing different cations from their exchangeable sites as the case for all clay minerals or from their zeolotic channels and micropores as the case for palygroskite might affect the rate of minerals reaction. It has been speculated that the zeolitic channels in palygroskite may retained some cations such as Na+, Ca+2, K+, and Mg+2 (Al-Barrak, 2001; Al-Hawas, 1998; Elprince, 1985). Sodium ion is one of the cations that may cause the high pH, which subsequently influence the dissolution of calcite and gypsum (Al-Barrak, 2001). However, there has been no evidence provided regarding the releasing of Na+ and Mg+2 from palygroskite and transformation or neoformation of new minerals under intensive leaching.
The formation of silicate mineral and specifically palygroskite required high pH (7.7-9.0) and high Mg+2 source (Al-Hawas, 1998; Elprince 1985; Singer and Norish, 1974; Elprince et al., 1979). Yet the stability of palygroskite in the arid and semiarid environment has been speculated by Al-Rawi and Sys (1967) since palygroskite is stable in environment, where precipitation rate is up to 400 mm. On the other hand, (Paquet, 1983) reported that palygroskite is unstable if the precipitation is more than 300 mm. Few literature (Farran and Chentouf, 2000) has been reported that the palygroskite might be neoformed or transformed to another minerals during batch leaching under laboratory condition and palygroskite retained some amounts of Na+ in zeolitic channel. Therefore, studies regarding the ability of palygroskite in holding certain quantities of minerals (as reservoir for cation minerals) and transformation or neoformation of palygroskite worth investigation.
The aim of this study was to examine the neoformation or transformation of palygroskite, smectite, mica and koalinte during batch extraction with water. Identification and quantification of minerals in soil before and after extraction and the source of Na+ and Mg+2 during the dissolution were also studied.
The study was conducted in laboratory condition at King Faisal University in 2003-2004. Soils of sandy clay loam (Gypsiferous-calcareous, Coarse silty, carbon tic hyperthermic Cambic Gypsiorthids), silty loam (Calcareous Sandy siliceous hyperthermic Typic Torripsamments), sandy clay (Gypsifereous-calcareous palygroskitic hyperthermic Anthropic Torrifluvents) and sandy loam (Gypsifereous-calcareous Montominrtic hyperthermic Typic Torriorthents) in addition to pure palygroskite as control were collected from C horizon from Al-Hassa oasis, Saudi Arabia. These soils were selected according to the differences in the types of clay minerals and natural of gypsum and CaCO3. The soils were dried, crushed and sieved to fraction size < 2 mm. The textural classes, (pH), Electrical conductivity (EC), Cation Exchange Capacity (CEC) were determined Table 1.
Leaching process: Duplicate 2.5 g samples of air-dry soil and 50 mL of ultra pure distilled water were placed into closed centrifuge tubes. The contact time for the experiments was 1 h for twenty times, during which the soil suspensions were shaked at 20±1°C. The supernatants were separated by centrifugation and decantation. EC and pH, Na+ and Mg2+ concentrations were measured according to the methods of Page et al. (1982).
One gram of pure palygroskite mixture and clay mixture of each soil and 50 mL ultra distilled water were placed into close centrifuge tube. The contact time was 1 h, during that; soil suspension was under constant and reproducible shaking under room temperature. The remaining sub-sample of palygroskite and clay were analysed by X-ray diffraction.
After completing the extractions, the CaCO3 and gypsum contents were determined by method of Bower and Huss (1948) and the mineralogy of soils were determined by the methods of Bish (1994). The measured values were averaged on duplicated samples.
In the soil, the Reference Intensity ratio = (palygroskite/Quartz) were calculated before and after leaching (Table 4 and 5). The mixture of hydrofluoric acid (HF) and perochloric acid (HClO) and concentration of hydrochloric acid (HCl) for digestion of soils sample and pure palygroskite were carried out before and after leaching.
Native and clay minerals analysis: Before and after the dissolution, the samples were analyzed by X-ray diffraction. Separation of various fractions for mineralogical analysis was achieved by gravity sedimentation of dispersed soils without removing calcium carbonate for X-ray diffraction. Oriented Mg and K saturated clay samples were prepared on ceramic tiles at room temperature and scanned from 3 to 30° 2θ (Degrees two theta) with a computer controlled Phillips diffractometer using graphite diffracted monochromatic CuK∝ radiation. Separate Mg saturated samples were analysed by X-ray diffraction (XRD) at room temperature after glycerol solvation. K saturation samples were analysed by X-ray diffraction (XRD) at room temperature after heating the samples at 330°C for 2 h and at 550°C for 3 h.
QUANTITATIVE MINERALOGICAL ANALYSIS
Mixtures of standard minerals were prepared and used as external standards (Table 2). The method of calculation was according to Bish (1994). Quantification of minerals was palygorskite, smectite, kaolinite, calcite, chlorite, illite (mica) and quartz.
| Table 1: | Some morphological and chemical properties of profiles at the four sampling sites in Al-Hassa oasis |
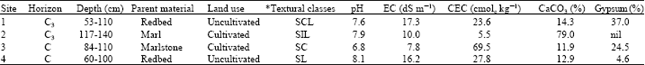 | |
| * SCL = Sandy Clay Loam, SIL = Silty Loam, SC = Sandy Clay and SL = Sandy Loam | |
| Table 2: | Synthesis of mixtures of minerals as external standards (g) |
 | |
The X-ray diffractogram of the soils indicated to the presence of quartz and calcite as the dominant minerals in the sand and silt fraction (Table 3) where palygorskite. Kaolinite was also identified in some site of the silt fraction (Table 3). The clay fractions showed that quartz, palygroskite and koalinite were the dominant minerals in most samples. Smectite and mica were identified in some site of clay fraction Table 3.
It seems that, mineralogical composition of silt and sand fraction almost similar in all profiles. Quartz was the most dominant minerals in sand fraction followed by calcite. Silt fraction however was dominated with calcite, gypsum and quartz. The abundance of these minerals might be due to the nature of parent material of the soil where (Al-Sayari and Zotl, 1978) pointed to that most of Al-Hassa soils were derived from marly and marly stone which are similar in their mineralogy.
Mineralogical composition of clay fraction was dominant with Quartz followed by palygroskite and kaolinite. Semectite, mica, calcite and chlorite were identified also. The analysis revealed that the presence of a large amounts of palygroskite and small quantity of smectite, illite (mica), quartz and kaolinite. Before leaching the Palygroskite was the dominant clay minerals in most of samples (range 31.49-94.11%) followed by smectite (0.5-85.40%), Chlorite (0.06-65.87%) and mica (1.15-10.56%). Quartz and kaolinite are less than 5% (Table 4 and 5).
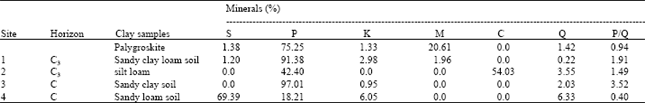
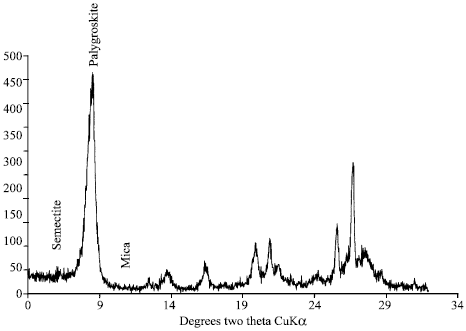
There was initially a large quantity of palygroskite in pure sample (95.58%), whereas after leaching become lesser (75.25%) and the kaolinite, smectite and mica appeared. Figure 1 and 2 show palygroskite might be transformed to smectite Semectite in palygroskite sample was absent before leaching and become 1.38% after leaching. This trend is in agreement with (Golden et al., 1985). It is expected that the pure palygroskite dissolution may be form kaolinite and smectite in repeated 1 h leaching and shaking at 20°C.
| Table 3: | Mineralogy composition of sand, silt and clay of the site sample of Al-Hassa oases soil |
 | |
| G = Gypsum, Cal = Calcite, P = Palygorskite, S = Smectite, K = Kaolinite, C = Chlorite, M = Mica and Q = Quartz, +: identified or detected , Plain: not detected | |
| Table 4: | The quantification of various minerals of clay fraction for the four soils and pure palygroskite before shaking. |
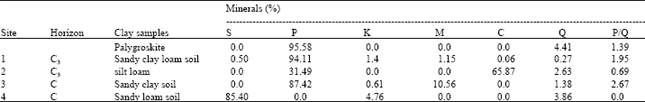 | |
| S = Smectite, P = Palygroskite, K = Kaolinite, C = Chlorite, M = Mica, Q = Quartz, Cal = Calcite | |
| Table 5: | The quantification of various minerals of clay fraction for the four soils and pure palygroskite after 1 h reciprocal shaking for twenty times |
 | |
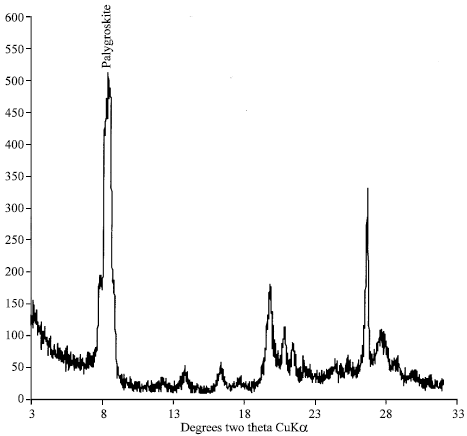 | |
| Fig. 1: | X-ray diffraction of pure palygroskite oriented before shaking |
 | |
| Fig. 2: | X-ray diffraction of palygroskite oriented at 1 h shaking |
This suggestion supported by the absence of smectite in the sample before leaching.
The effect of leaching on the pure palygroskite shows that palygroskite has decreased by 20% as showing by the P/Q ratio which showed decreasing from 1.39 to 0.94. However, new minerals (Smectite 1.38%, mica (illite) by 20.61% and kaolinite by 1.33%) have appeared which probably raise the possibility neoformation due to the release of different elements during leaching experiment (Fig. 2).
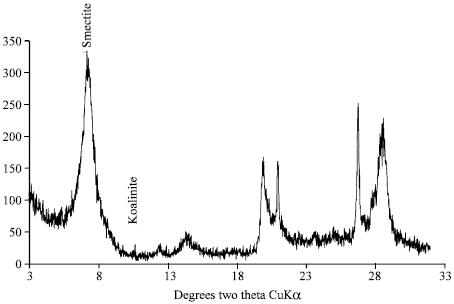 | |
| Fig. 3: | X-ray diffraction of clay fraction of sandy loam soil before shaking |
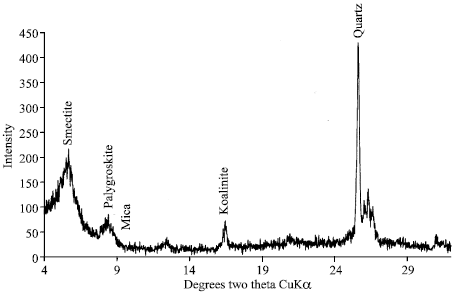 | |
| Fig. 4: | X-ray diffraction of clay oriented of sandy loam soil after 1 h shaking |
The effect of leaching on sandy clay loam soil shows that palygroskite has been decreased by 3% as shown by the P/Q ratio which showed decreasing from 1.95 to 1.91. However, other minerals has increased, which probably may be neoformation due to release different elements during leaching. These minerals precipitate as smectite 0.70%, Mica (illite) 0.81% and kaolinite 1.58%.
Results of quantification of the palygroskite of sandy clay soil shows that, palygroskite has increased by 10% as shown by the P/Q ratio which showed increasing from 2.67 to 3.52. This increase probably due to releasing of element under high pH and Mg+2 during leaching. However, the mica has disappeared probably due to the intensive leaching which might be asses to form palygroskite .
The effect of leaching on silt loam soil shows that palygroskite mineral has been increased by 11% as shown by the P/Q ratio which indicate increasing from 0.69 to 1.49 (Table 4 and 5). This increase probably due to releasing of Mg+2 elements during leaching from Mg-calcite or from zeolitic channels (Elprince, 1985). Which is supported by results from treated pure palygroskite (Table 6).
Effects of leaching on sandy loam soil show that smectite has been decreased by 16% as shown by the P/Q ratio which indicates decreasing from 1.8 to 1.20. The palygroskite and koalinite were not found in sandy loam soil. However, new minerals palygroskite (18.21%) and koalinite (1.34%) had appeared which probably due to the release of different elements during leaching (Fig. 3 and 4).
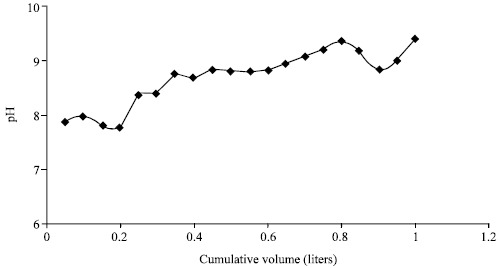 | |
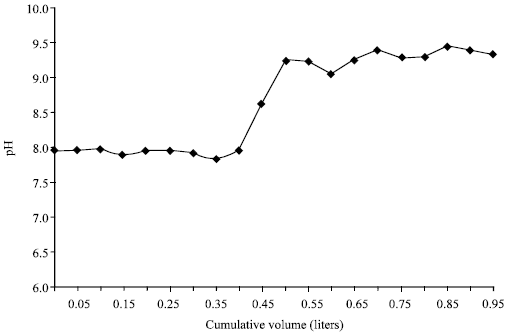
| Fig. 5: | Cumulative water volume against pH at 1 h shaking for sandy loam gypsifereous-calcareous soil |
 | |
| Fig. 6: | Cumulative water volume against pH at 1 h shaking for sandy clay loam gypsifereous-calcareous soil |
| Table 6: | Cumulative amounts of Na+ and Mg+2 released from pure palygroskite at 1 h shaking, Na to Mg ratio and exchangeable and retained Na |
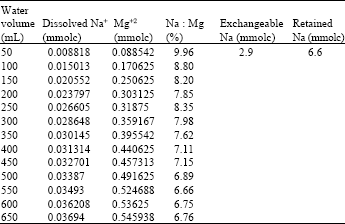 | |
The Zeolitic channels of Palygroskite seemed to be the source for Na+. The probable source of Mg+2 is the dissolution of palygroskite resulted from hydrolysis of Na-palygroskite. Exchangeable Na+ measuring and acid digestion for pure palygroskite offer a support for zeolitic channel of palygroskite. The result of extract and acid digested of palygroskite showed that total 10.1 mmol L-1 of Na, dissolved Na was 0.6 mmol L-1, 2.9 mmol L-1 of Exchangable Na and the remaining value 6.6 mmol L-1 retained by palygroskite (retained 6.6 mmol L-1 of Na+ by treated with NaCl at CEC of palygroskite determination).These results were also supported by treated pure palygroskite (Table 6).
After shaking ceased the pH in short contact time experiment increased probably due to hydrolysis of Na-palygroskite for the three soil sandy loam and sandy clay loam soil in Fig. 5 and 6.
The X-ray diffraction for clay fraction of Al-Hassa Oasis showed that the presence of clay minerals i.e. palygroskite, illite, smectite, chlorite, kaolinite. Quartz was the dominant minerals in non-clay minerals. Palygroskite is the most abundant clay mineral. The presence of these minerals could be ascribed to the nature of the parent material. Quantification after 1 h leaching indicated that, all minerals increased in quantities except of sandy loam soil, where smectite decreased during leaching. Smectite may be transformed to mica or kaolinite minerals. Pure palygroskite leached with distilled water might be transformed to mica, smectite and kaolinite under 20°C. However, Smectite, kaolinite and mica precipitated at expense of pure palygroskite. Pure palygroskite is probably source of Mg+2 resulted from dissolution of palygroskite. The zeolotic channels of palygroskite might be the source for Na+. Finally, the intensive irrigation in the field is likely to be conductive to the dissolution and neoformation of palygroskite, smectite, and mice.