Research Article
Intelligence Quotient in Children with Epilepsy
Departments of Neuropsychiatry,Al-Azhar Faculty of Medicine, Egypt
Hani Al-Khaligy
Departments of Pediatrics, Al-Azhar Faculty of Medicine, Egypt
Cognition and intelligence are usually used as a synonymous terms but cognition is defined as the mental process of knowing from the environment, that includes aspects of awareness, perception, memory, reasoning and judgment (Park et al., 2013). On the other hand, intelligence is only one aspect of cognition and is evaluated by a standardized intelligence test (Sparrow and Davis, 2000).
Epilepsy is a disorder of the brain, caused by abnormal electrical activity in neurons and is closely associated with cognitive comorbidities which had major concerns in children with epilepsy (Berg, 2011). It is a chronic neurologic disorder and commonly affects children and adolescents. The estimated prevalence in Europe is about 4.5-5 per 1000 (Forsgren et al., 2005). Under the age of 15, the vast majority of epilepsies are idiopathic, without any identifiable cause, other than a genetic predisposition (Dragoumi et al., 2013).
Childhood epilepsy is associated with frequent social and learning disabilities. Intellectual disability is a major contributor to the psychosocial comorbidity in childhood epilepsy. However, the majority of studies treating this topic lack well-defined syndrome classification. They have included children defined by seizure type and in some studies have not distinguished between children with generalized and focal epilepsy (Nolan et al., 2003). These studies have been further limited by focusing on school-aged children (Bailet and Turk, 2000), children within the normal range of intellectual function (Kolk et al., 2001). Furthermore, studies evaluated the relationship of epilepsy syndrome to intelligence were largely confined to children with generalized idiopathic epilepsy (Pavone et al., 2001), demonstrating lower intellectual performance in these children compared to controls. In addition, studies of focal epilepsy has been confined to the group as a whole or restricted to the study of small numbers of children with specific lobar syndromes (Bulteau et al., 2000; Lassonde et al., 2000).
The aim of the study is to evaluate intelligence in epileptic children and to determine the factors associated with intellectual impairment.
The present study was carried out at both out-patients clinic of pediatric and Neuropsychiatry, Al-Azhar University hospital (New Damietta), during the period from June 2012 through September 2013. The studied subjects were classified in to the following groups: Group 1 (patient group): It consisted of sixty (60) epileptic children, who regularly visit our clinics for follow up, or ask medical advice for any other condition. Group 2 (control group): It consisted of sixty (60) normal healthy, age and sex matched children, free from medical and/or psychiatric problem.
Exclusion criteria: Children whose predominant seizure type was not recorded and children with progressive neurological disorders were not included in the study.
The clinical assessment of each child included the following demographic and clinical variables: age of epilepsy onset, duration of active epilepsy, seizure frequency and current medications. Medical history and neurological examination were obtained in all patients at the time of inclusion in the study.
Epilepsy classification was based on clinical data, seizure semiology, interictal and ictal EEG using International League Against Epilepsy (ILAE) criteria (ILAE, 1989). The EEG was recorded and reviewed for evidence of generalized or partial epileptiform discharges. Localization of the ictal EEG was based on the region of onset of epileptiform activity. Classification according to the ILAE was determined by concordance between at least two of the four parameters (clinical data, semiology, interictal EEG, ictal EEG) and the absence of discordance. Epilepsy syndromes were first classified as generalized or partial. Generalized epilepsies were subclassified into Generalized Idiopathic Epilepsy (GIE) or Generalized Symptomatic Epilepsy (GSE). Partial epilepsies were subclassified into Frontal Lobe Epilepsy (FLE), Temporal Lobe Epilepsy (TLE), Central Epilepsy (CE) or Occipital Epilepsy (OE).
For intellectual assessment, the IQ Wechsler Intelligence Scale for children, 4th edition-revised version was used and validated for use in epilepsy by Sherman et al. (2012). The test is divided into two sections with each section containing a number of subtests. The two broad sections of the test are the verbal scale and performance scale. Successful completion of any item on any of the verbal subtests requires a verbal response. On the performance subtests, the person must do been something in response to a question or task. When the entire test has administered, the assessor calculates what is called a composite score, a score that takes into account both sections. Because it is a test of intelligence, the test scores obtained are called IQ scores and the results stated as verbal scale IQ; performance scale IQ and full scale IQ (the composite score). The full scale score, according to the standard interpretation, indicates the level of a person’s intelligence. The level of intelligence was categorized as normal (intelligence quotient [IQ] = 80), borderline mental retardation (IQ 70-79), mild mental retardation (IQ 60-69) and moderate to-severe mental retardation (IQ<60).
Ethical considerations: Written permission to implement the study was obtained from Al-Azhar University hospital director and local ethical committee. Written consents were obtained from each patients and/or his or her caregiver before history taking, clinical examination and investigations after explaining the study and its aim to the parent.
Data management: The collected data were organized, tabulated and statistically analyzed, using Statistical Package for Social Science (SPSS) version 16 (SPSS Inc, Chicago, USA), running on IBM compatible computer with Microsoft ® Windows 7 Operating System. Mean, standard deviations were calculated for quantitative data and qualitative data were represented as relative frequency and percentage distribution. The student (t) test for comparison between two means, while Chi square (X2) or Fisher exact tests were used for testing significance of observed differences between studied patients for qualitative data. The level of significance was adopted at p<0.05%.
The general characteristics of the studied cases were depicted in Table 1. Male patients represented 55% of cases; age ranged from 5 to 15 years; the mean age at disease onset was 2.96 years; epilepsy was general in 30% and focal in 70% of cases; it was General Idiopathic Epilepsy (GIE) in 16.7%; General Specific Epilepsy (GSE) in 13.3%; Focal Central Epilepsy (FCE) in 10%, Frontal Lobe Epilepsy (FLE) in 16.7% and Temporal Lobe Epilepsy (TLE) in 20%. Majority of cases (45.0%) received one antiepileptic drugs, two drugs reported in 40% and three drugs in 15%; fits in the last 12 months were 0-1 in 46.7; 2-9 in 33.3% and 10 in 20.0%; total IQ score ranged from 55 to 97 (the mean was 77.96±13.26); it was normal in 33 cases (55.0%); borderline in 7 cases (11.7%); mild mental retardation in 9 cases (15.0%) and moderate to severe MR in 11 cases (18.3%).
Comparing study and control groups, we found that, there was no significant difference between both groups as regard to age or sex. On the other hand, there was significant decrease of IQ in study group when compared to control group (77.96±13 vs. 86.79±8.66, respectively) (Table 2).
Studying the relation between IQ and different clinical characteristics, it was found that, there was no significant difference between focal or general epilepsy; no difference between subtypes of epilepsy; no difference between different age groups; no difference in relation to number of antiepileptic drugs or number of fits in last year.
| Table 1: | Characteristics of the study group |
 | |
| n: Number, %: Percentage | |
| Table 2: | Comparison between study and control groups |
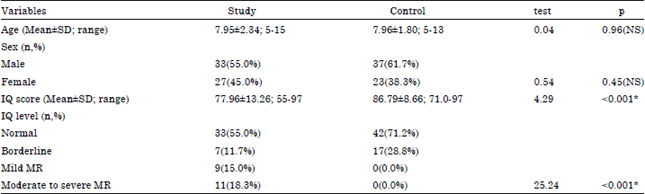 | |
| NB: Data represented as the arithmetic mean and standard deviation, SD: Test, refers to student, (t): Test or chi square test, p < 0.05 is significant, NS: No significant | |
On the other hand, females had significantly lower IQ in comparison to males (72.77±14.02 vs. 82.21±11.10, p-value = 0.005) (Table 3). Running multivariate regression analysis, only patient gender was associated with low IQ (β = 2.16, CI 0.17-0.87; p = 0.028).
| Table 3: | Comparison of IQ according to clinical characteristics |
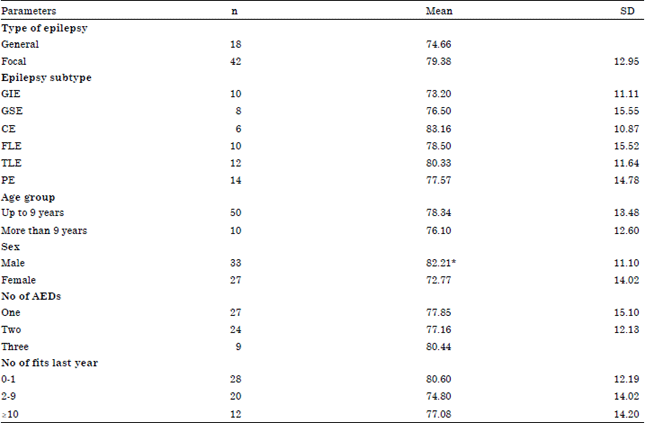 | |
| *Significant increase of IQ in male when compared to female (p value = 0.005), n: Number, SD: Standard deviation | |
The present study was designed to examine the levels of IQ in epileptic patients when compared to normal children and to know the relation with clinical factors. Results of the present study showed significant decrease of IQ in epileptic children when compared to normal children. In addition, female gender was associated with low IQ than males. The prevalence of MR in epileptic children in the present study was 33.3%. This incidence is slightly higher than that reported in different studies in previous literature; e.g., Murphy et al. (1995) reported that 30% of 10 year old children with epilepsy had mental retardation, while Camfield and Camfield (2007) reported that approximately 20% of children in a population-based epilepsy cohort had mental retardation (IQ<70). In addition, Berg et al. (2008) reported that 26.4% of patients with childhood epilepsy in a community-based cohort had subnormal cognitive function (IQ<80). The possible explanation for changes in incidence of MR in epileptic children can be attributed to different exposure to factors that influence intelligence such as education and rehabilitation systems, different sample sizes and different methods for IQ determination. On the other hand, Park et al. (2013) reported that, over 50% of the cohort had low intelligence (IQ<80). They explained this high prevalence of cognitive impairment by reluctance of parents to conduct IQ tests in all children with epilepsy and children with clinically suspected mental retardation were more likely to be included in their study.
Furthermore, it had been reported that, cognitive deficits are among the common neurobehavioral comorbidities of epilepsy (Hermann et al., 2008; Elger et al., 2004). Studies of a variety of epilepsies have reported intellectual ability to be below that considered normal for age (Nolan et al., 2003). While, the basic mechanisms underlying these deficits have been the subject of much speculation (Brooks-Kayal, 2011; Jensen, 2011), it is clear that the nervous system of children appears to be particularly vulnerable to the effects of intractable epilepsy.
It had been reported that, the proper understanding of risk factors negatively affect IQ in epileptic children is of utmost importance, as it will allow to interfere to correct correctable factors and treat it properly. In the present study, we found the female gender to be associated with lower IQ. These results are quietly different than that reported in previous studies, where duration of epilepsy, higher number of seizures in the preceding year and epilepsy classification were significant risk factors for low intelligence. Seizures are thought to damage the brain through anoxia, lactic acidosis and excessive excitatory neurotransmitters and this might underlie the associations between seizure burden and low intelligence (Meador et al., 2001).
In the present study, antiepileptic drug usage was not a significant risk factor for low intelligence in the multivariate analysis. However, in contradiction to these results, it was reported that, antiepileptic drugs decrease neuronal excitability, interfere with normal neuronal networks and induce cognitive deficits. Also, it was reported that, polypharmacy (more than one drug), higher target doses, higher serum level of AEDs and rapid titration are associated with adverse cognitive effects (Sankar and Holmes, 2004).
In the present study, age of the child or age of epilepsy onset were not found be risk factors for intellectual dysfunction. However, in previous literature, it was reported to be confounding risk factors especially the age of onset. It was reported that, the age at seizure onset is an important factor for cognitive function. Children with age of seizure onset <5 years showed significantly lower IQ regardless of epilepsy classification (Berg et al., 2008) and, among surgically treated patients with temporal lobe epilepsy, intellectual impairment was more frequent in patients with seizure onset before 1 year of age (Cormack et al., 2007). The possible explanation for this contradiction may be attributed to the fact that, in all studied cases of the present study, the age of onset was less than or equal to 5 years of age.
In the present study, epilepsy type or subtype were not found to affect IQ. According to Henkin et al. (2005), intellectual affects is not caused by seizure burden or treatments but are innate characteristics of IGE itself.
In short, the results of the present study confirmed the negative effect of epilepsy on intellectual function. On the other hand, it found that, only female gender was associated with lower IQ. The small number of cases may be a limiting factor in the present study. Thus, it is advisable to validate results of the present work in a large cohort.