ABSTRACT
Background: Heavy metals pollution of drinking water has become one of the most serious problems due to their sever toxicities to human body. Materials and Methods: In this study, the adsorption of Pb+2, Cd+2 and Cu+2 ions from contaminated water using dried brown alga Sargassum latifolium (SAP) and its activated carbon (SAC) was investigated. The impact of different parameters such as initial metal ions concentration, contact time, adsorbent dose and pH on the removal were evaluated. Results: The optimum adsorption was found to occur at contact time 60 min, pH value 5.0, adsorbent dose 8.0 g L–1 and initial concentration 80 mg L–1. The maximum removal capacity of SAC was 167 mg g–1 for Cd+2, 147 mg g–1 for Cu+2 and 141 mg g–1 for Pb+2. The SAC was found to be the highest efficient column capable to remove 100% of heavy metals from drinking water samples. Conclusion: Thus SAC column is considered as an efficient and cheap biotechnology for removing the heavy metals in drinking water treatment plants.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/biotech.2016.65.75
URL: https://scialert.net/abstract/?doi=biotech.2016.65.75
INTRODUCTION
Heavy metals pollution is the major environmental problem in surface and ground water of many countries, where the metal ions contents of contaminated water in several cases exceed the admissible sanitary standards1,2. Therefore, the US Environmental Protection Agency (USEPA), World Health Organization (WHO) and many government environmental protection agencies have set the Maximum Permissible Limits (MCLs) for the heavy metals in industrial effluent as well as drinking water3.
Cadmium, copper and lead are the most toxic pollutants found in surface water and groundwater as well as trade wastewater4. These toxic heavy metals are leading to surface waters through agriculture, domestic sewage and different industrials activities including metal plating, mining, tanneries, textile, dying, battery, electroplating, plastics and chemical5,6.
The traditional water treatment process including coagulation, sedimentation, sand filtration and chlorination are ineffective in removing heavy metals7,8. Consequently, the occurrence of these heavy metals in drinking water causes a serious threat to human health as kidney damage, human carcinogenesis, liver damage and nervous system disorder9-12. Therefore, it is urgent to treat heavy metals of drinking water prior to its discharge to the distribution systems.
Activated carbon is a widely and common technique used for metal absorption in water treatment processes6,13. However, the application of activated carbon is very restricted in growing countries because of its high cost14. Various approaches have been studied for the production of more effective and cheaper absorbent materials. Among several biological materials reported in the literature, brown algae are identified as a promising biomass for the development of new activated carbon due to their high performance in metal uptake and the abundance of algal biomass in the littoral zones of world oceans4,15.
The aim of this study is to estimate the performance of marine brown alga Sargassum latifolium for the removal of heavy metals and to identify the major parameters affecting its biosorption. Furthermore, this is the first time to study the removal efficiency of activated carbon based brown macroalga Sargassum latifolium for toxic Cd+2 , Pb+2 and Cu+2 ions of drinking water treatment plants.
MATERIALS AND METHODS
Preparation of algal materials
Preparation of Sargassum algal powder (SAP): The marine brown alga Sargassum latifolium (Fig. 1) was collected from the Red Sea at Hurghada coast (located 27 latitude and 33 longitude) in Egypt, during winter season (January, 2015).
 | |
| Fig. 1: | Collected marine macroalgal species Sargassum latifolium |
The alga was washed with tap water then distilled water to remove impurities. Washed alga was oven dried at 60°C to a constant weight. The dried algal materials were ground using a Retsch ZM200 titanium mill. The powdered materials were sieved to obtain 0.1-0.2 mm particles.
Preparation of Sargassum activated carbon (SAC): The dried algal material was carbonized by at 600°C for 3 h. Activated carbon was prepared by soaking the carbonized sample in potassium hydroxide for 48 h (sodium hydroxide: carbonized sample in ratio of 3:1). The sample was calcined at 800°C for 3 h. After cooling, the produced activated samples were washed several times with distilled water, then dried at 110°C16.
Metal ion solutions: Stock solutions (1000 mg L–1) of tested heavy metal were separately prepared by dissolving CuSO4·5H2O, Cd(NO3)2.4H2O and Pb(NO3)2 (analytical grade) in deionized water. The working solution was prepared by diluting stock solution to appropriate volumes. The heavy metals concentrations in the solutions were determined using Perkin-Elmer AAnalyst 700 flame atomic absorption spectrophotometer.
Characterization of adsorbents
Textural characterization: Total surface area (m2 g–1), pore radius (Å) and total pore volume (mL g–1) were performed via nitrogen adsorption at -196°C for SAP and SAC using Quantachrome NOVA2000, USA gas sorption analyzer17.
Scanning Electron Microscopy (SEM): Scanning electron micrographs of SAP and SAC were obtained using Quanta 250 FEG, USA. The sample was dried at 110°C for 4 h and gold coated.
FTIR spectroscopy: Fourier Transform Infrared Spectra (FTIR) for SAP and SAC were detected on a Mattson 5000 FTIR spectrometer, UK. For FTIR studies, pellet of algal material and KBr (1:10) was made to study the functional groups and binding sites present on the algal surface18.
Batch removal experiments
Determination of optimum pH: The algal materials were soaked in separate 250 mL flasks each containing 50 mL of metal ions. The parameters of initial metal ion concentration and biosorbent dosage were fixed at 10 mg L–1 and 0.1 g, respectively. Effects of pH were tested at pH value 2-8. The flasks were placed in a rotated shaker (120 rpm) at room temperature. After 120 min, the solution was filtered to analyze heavy metals left in the solution.
Determination of optimum biomass dosage: This part was performed to verify the effect of biomass weight on the adsorption process. Different weights of biomass (2, 4, 6, 8 and 10 g L–1) were mixed and shaken with a solution of 10 mg L–1 at pH 5 for 120 min.
Determination of optimum contact time: Constant weight of SAP and SAC (0.4 g) were added to 10 mg L–1 heavy metal solution. Contact times were fixed at t = 5, 10, 20, 60 and 120 min. The solutions were filtered after each contact time and analyzed by atomic absorption spectrophotometer.
Determination of optimum initial metal concentration: In order to assess the effect of initial metal concentration on the adsorption, different concentrations (5, 10, 20, 40, 60, 80 and 100 mg L–1) were examined at constant parameters; biomass dosage 8.0 g L–1, solution pH 5.0 and contact time 60 min.
Metal removal efficiency: Biosorption capacity of algal materials (qe) can be calculated in mg g–1 as follows in Eq. 1:
| (1) |
where, Ce is the equilibrium concentration (mg L–1), V is the volume of solution, Co is the initial metal concentration (mg L–1) and m is the mass of biosorbent (g). Percentage of metal removal can also be displayed as follows in Eq. 2:
| (2) |
Continuous removal experiments
Preparation of water samples: Raw and treated water samples were collected from different DWTPs situated on the river Nile and its branches in Egypt during April, 2015. Three water samples replicates were collected in polyethylene containers during working hours of the treatment stations. Upon arrival to the laboratory, water samples were filtered through millipore filters (0.45 μ) and then heavy metal ions were analyzed by atomic absorption spectrophotometer.
Preparation of algal column: A glass column with the dimension, 20 cm long and 1.2 cm wide (Fig. 2) was used to remove the most toxic heavy metals in water samples of Drinking Water Treatment Plants (DWTPs). A constant weight of different algal materials was placed in each column. The drinking water sample was pumped up flow at the bottom of each column. Treated water samples were filtered through a 0.45 μm millipore filter paper. The filtrate was subjected to metal analyze by atomic absorption spectrophotometer. Blanks were made using drinking water samples to determine their original heavy metal concentration before using them in the removing columns.
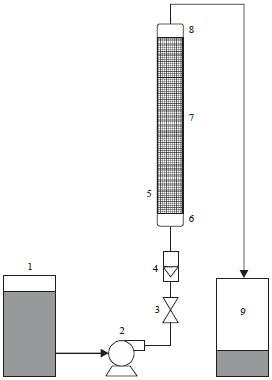 | |
| Fig. 2: | Experimental arrangement of the removal packed bed column, 1: Drinking water, 2: Peristaltic pump, 3: Valve, 4: Flow meter, 5: Column, 6: Bottom sieve, 7: Packed algal material, 8: Top sieve and 9: Algal treated water |
RESULTS AND DISCUSSION
Surface characterization of algal materials: Adsorption capacity of algal materials is directly affected by surface area and porosity. Table 1 showed the obtained data using nitrogen adsorption at -196°C. The surface area of SAC (1207 m2 g–1) about 16.2 times more than SAP surface area (74 m2 g–1) which is related to the effect of activating agent (NaOH) in creation of internal surface area due to the formed microspores. Total pore volume for SAC (0. 831 mL g–1) which is about 5.1 times than that calculated for SAP because the application of NaOH as activating agent create numerous small pores on the surface of SAP. These superior properties could provide more active sorption sites for adsorption of heavy metals19.
This result was also confirmed by the SEM micrograph for SAP and SAC (Fig. 3). The SEM image for SAP showed the absence of pores on the surface which reflects the small surface area for algal powder.
| Table 1: | Textural characterization of Sargassum latifolium powder (SAP) and its activated carbon (SAC) |
 | |
| Fig. 3(a-b): | SEM micrograph for Sargassum latifolium (a) Dried powder and (b) Activated carbon |
However, treatment with NaOH at 800°C appeared to increase the pore size of SAC by the removal of certain portions on algal surface biomass, leaving behind a wide distribution of pore size14,20.
Adsorption capacity of algal powder and its activated carbon not depends only on surface area but also on chemical surface function groups. The FTIR spectra for SAP and SAC (Fig. 4) displayed the active functional groups as nitro, carboxyl, hydroxyl, amino and other active groups which can bind with heavy metal ions21.
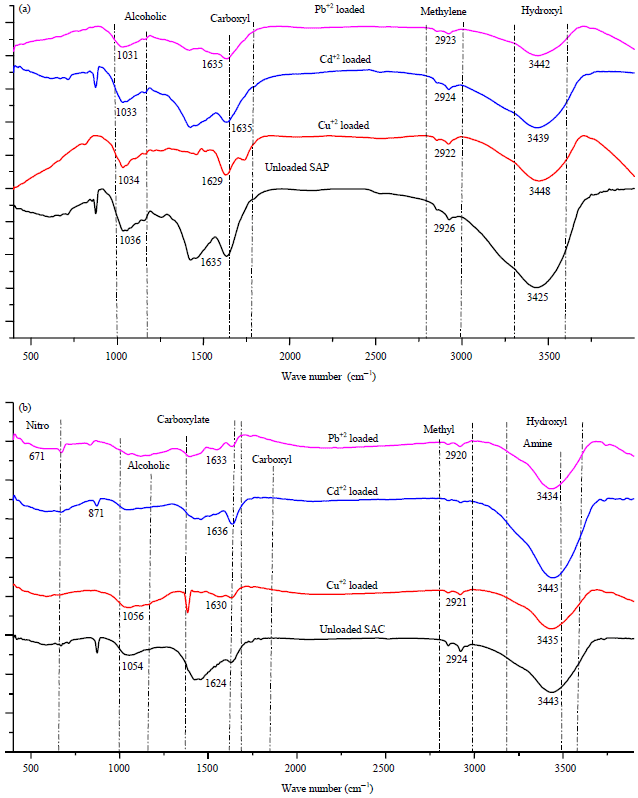 | |
| Fig. 4(a-b): | FTIR spectra for (a) Dried powder and (b) Activated carbon |
It was observed that SAC spectrum is rich with surface function groups compared with the original SAP which may be due to activation with NaOH. The peak shape of unloaded SAP and SAC was changed by a shift in peak wavelength after heavy metal loading.
Optimization of heavy metals adsorption
Solution pH: The solution pH is the most important factor affecting heavy metal adsorption capacity. It generally affects the chemical metal states and availability of active groups on algal surface. The algal adsorption capacity was examined at different pH values ranging from 2.0-8.0 as displayed in Fig. 5. The adsorption efficiency of different heavy metal ions was markedly increased with rising solution pH from 2.0-4.0. The maximum metal uptake, 85% for SAP and 98% for SAC was obtained at pH 5 and then gradually decreased as solution pH increased from 6-8. Therefore, a pH 5 was considered as an optimum pH value for further metal ion adsorption.
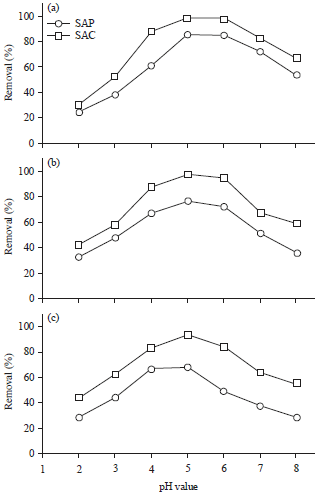 | |
| Fig. 5(a-c): | Effect of pH on (a) Cu+2, (b) Cd+2 and (c) Pb+2 removal efficiencies by Sargassum powder (SAP) and its activated carbon (SAC) |
At lower pH values (2.0-4.0), the metal biosorption efficiency of SAP and SAC is very poor, due to large quantity of H3O+ ions which compete with tested heavy metals at binding sites22,23. As solution pH increases, the electrostatic repulsions between the surface of algal masses and the positively charged metal ions are depressed, which increase the percentage of metal uptake17,24. The decrement in the metal uptake capacity at higher pH values (6.0-8.0) could be due to the repulsion between the negative charge of algal surface and negative charge of the anionic species in solution25,26.
Algal dosage: In this study, various quantities of SAP and SAC (2.0–10.0 g L–1) were contacted with a fixed initial metal concentration and solution pH at 10 mg L–1 and 5, respectively. The percentage of metal uptake versus the algal dosage (g L–1) is shown in Fig. 6. For adsorption of metal ions onto SAC increase adsorbent dosage between 2.0 and 8.0 g L–1 is accompanied by increase in percentage metal uptake from 59-90%, 46-99% and 42-96% for Pb+2, Cu+2 and Cd+2, respectively.
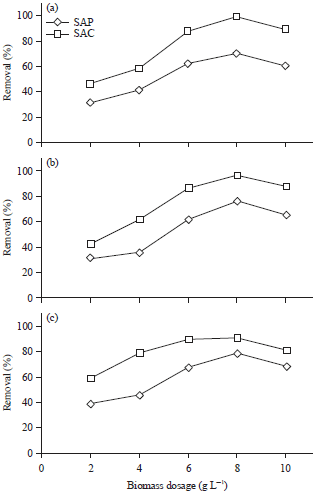 | |
| Fig. 6(a-c): | Effect of algal dosage on (a) Cu+2, (b) Cd+2 and (c) Pb+2 removal efficiencies by Sargassum powder (SAP) and its activated carbon (SAC) |
Considerable decrease in removal capacity was clearly noticed as algal dosage increased between 8.0 and 10.0 g L–1. Similar trend was noticed in case of SAP but with lower adsorption percentage due to the higher porosity and surface area of SAC compared to SAP. Based on these results, the algal dosage 8.0 g L–1 was chosen as an optimum value for metal uptake.
The maximum metal adsorption at a higher adsorbent dosage (8.0 g L–1) could be due to the higher number of free binding sites on SAP and SAC which enhance the percentage of metal ions uptake. High adsorbent amounts than 8.0 g L–1 are known to cause partial aggregation of biomass and a consequent reduction in intercellular distance, leading to the protection of binding sites from metal ions6,22.
Contact time: Metal ions biosorption is strongly affected by contact time. Figure 7 shows biosorption of tested metal ions by Sargassum powder (SAP) and its activated carbon (SAC) with different contact times (5-120 min). The adsorption efficiency increases with increase the contact time up to 60 min, followed by a relatively slow biosorption process.
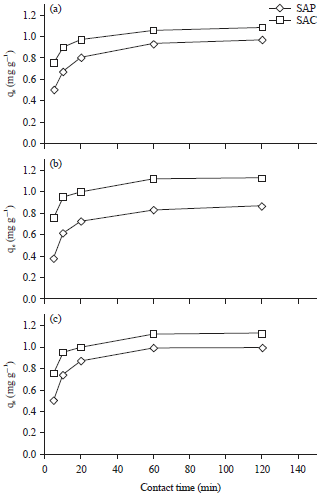 | |
| Fig. 7(a-c): | Effect of contact time on (a) Cu+2, (b) Cd+2 and (c) Pb+2 removal efficiencies by Sargassum powder (SAP) and its activated carbon (SAC) |
However, the majority of adsorption was achieved after 30 min and equilibrium time reached at 60 min. Therefore, 60 min was selected as optimum contact time for metal ion biosorption. This shorter contact time represents a clear preference for practical uses of SAP and SAC in a packed column experiments for removal of heavy metals from drinking water treatment plants.
Metal ion concentration: Metal ion concentration is strongly influences the metal uptake processes. Figure 8 showed that the adsorption efficiency of SAC for different metal ions increased at the lower metal ion concentrations and reached the maximum of 94% for Cu+2, 90% for Cd+2 and 91% for Pb+2 at 80 mg L–1. After that with increasing initial concentration, the SAC adsorption efficiency still unchanged. The same trend was spotted in case of SAP but with lower adsorption efficiency compared to SAC.
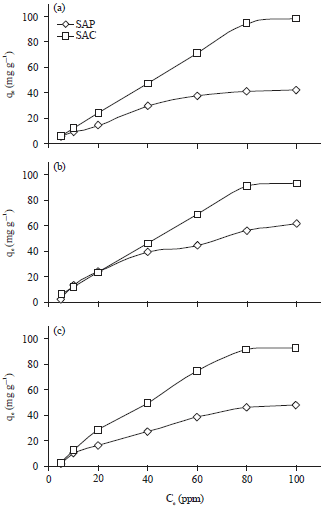 | |
| Fig. 8(a-c): | Effect of (a) Cu+2, (b) Cd+2 and (c) Pb+2 ions concentrations on removal efficiencies by Sargassum powder (SAP) and its activated carbon (SAC) |
This behavior was attributed to that all active sites on the algal surface were unoccupied resulting in high adsorption at the beginning. Thereafter with increasing initial metal ions, the removal efficiency was decreased because of a few binding sites were available on the surface of SAP and SAC20,27.
Langmuir isotherm adsorption model was used to describe the adsorption processes between metal ions and biomaterial28. From this model, the maximal algal adsorption capacity was determined according to the following Eq. 3:
| (3) |
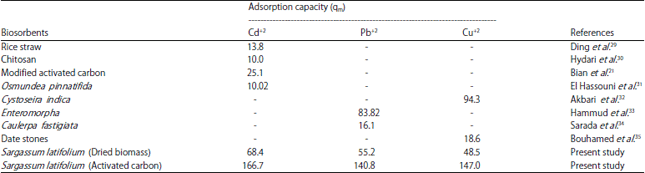
where, Ce (mg L–1) is the equilibrium concentration, qe (mg g–1) is the amount algal material at equilibrium, b (L mg–1) is Langmuir constant and qm (mg g–1) represents the maximum adsorption capacity for SAP and SAC. From the linear plot of Langmuir equation (Fig. 9), the maximum adsorption capacity is calculated in Table 2. Upon observation of the Table 2 (i) qm for SAC more than SAP which related to the higher surface area for SAC and the presence of microporosity as indicated by SEM image. (ii) Comparing qm values for activated carbon of S. latifolium with various sorbents reported in the literature, indicating the higher efficiency of activated carbon to remove different heavy metals than the majority of the biomasses given in the Table 2. This comparison demonstrated that activated carbon of macroalga S. latifolium is a potential biosorbent material for different heavy metals because of its high adsorption capacity.
Application of algal columns to remove heavy metals from drinking water
Heavy metals in drinking water samples: Levels of tested heavy metal ions in raw and treated water samples of different treatment plants on the river Nile and its branches exhibited distinct variations (Table 3). Concentration of heavy metals in raw water ranged between 0.001 and 0.081 mg L–1. Different DWTPs have been shown to be ineffective in eliminating different heavy metals from raw water samples. The results showed that the concentration tested heavy metal ions (Cd+2, Cu+2 and Pb+2) of surface and drinking water samples are higher than the permissible limits set by WHO36.
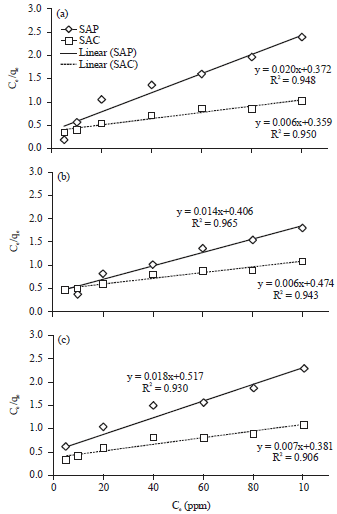 | |
| Fig. 9(a-c): | Langmuir adsorption isotherm of (a) Cu+2, (b) Cd+2 and (c) Pb+2 on Sargassum powder (SAP) and its activated carbon (SAC) |
| Table 2: | Comparison of adsorption capacity (qm) of brown alga Sargassum latifolium with that of different algal species and other biomasses |
 | |
| Table 3: | Concentration of toxic heavy metals (mg L–1) in water samples of different DWTPs |
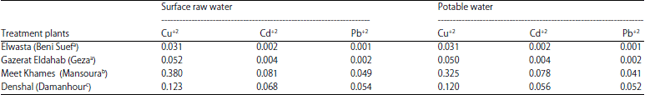 | |
| aTreatment plant on river Nile, bTreatment plant on Damietta branch and cTreatment plant on Rosetta branch | |
| Table 4: | Efficiency of algal columns to remove heavy metals (mg L–1) from drinking water samples |
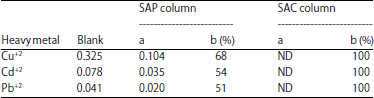 | |
Blank: Concentration of heavy metal in potable water of treatment plant, a: Concentration of heavy metal in water sample passed through algal column, b: Percentage of metal removal and ND: Not detected | |
The lack in coagulation and filtration system of the water treatment plants is reasons of heavy metal residual in potable water1,16.
Efficiency of algal columns to remove heavy metals from drinking water: The ability of SAP and SAC columns to remove different metal ions from drinking water samples varied strongly from one column to another (Table 4). The capacity of SAP column to remove Cd+2, Cu+2 and Pb+2 ions ranged between 51 and 68%. Whereas, a complete removal of all tested heavy metals (100%) was recorded in case of SAC column. The highest heavy metal adsorption of algal activated carbon could be due to the highest pore size and surface area of SAC which supplied more binding active sites for adsorption of heavy metals. Therefore, activated carbon of brown alga Sargassum latifolium will be the most suitable technology for treating contaminated water in the final drinking water treatment process before being discharged to the distribution system.
CONCLUSION
Sargassum latifolium powder (SAP) and its activated carbon (SAC) were prepared to treat toxic Cd+2, Cu+2 and Pb+2 ions. SEM and nitrogen adsorption indicated the presence of porosity on SAC more than SAP. The optimum removal conditions of SAC and SAP was found to occur at contact time 60 min, pH 5.0, adsorbent dose 8.0 g L–1 and initial metal concentration 80 mg L–1. Activated carbon of Sargassum was found to be more efficient than its algal powder in removing of tested heavy metals.
Although, SAC is a widely used and accepted method in water treatment, the present study is the first evidence to use Sargassum activated carbon in removing heavy metals of DWTPs. The results demonstrated that SAC column could be cheap and liable for elastic design in complete removal of heavy metals from drinking water samples. Consequently, study should continue to transfer this laboratory scale research to industrial scale throughout the characterization of SAC saturation over time as well as the effectiveness and the cost under conditions that could be experienced in DWTPs.
ACKNOWLEDGMENT
The facilities provided by the Faculty of Science, Damanhour University, Egypt are greatly acknowledged and appreciated.
REFERENCES
- Ab Razak, N.H., S.M. Praveena, A.Z. Aris and Z. Hashim, 2015. Drinking water studies: A review on heavy metal, application of biomarker and health risk assessment (a special focus in Malaysia). J. Epidemiol. Global Health, 5: 297-310.
CrossRefPubMedDirect Link - Shang, Y., X. Yu and M.E. Romero-Gonzalez, 2015. Screening of algae material as a filter for heavy metals in drinking water. Algal Res., 12: 258-261.
CrossRefDirect Link - Khan, K., Y. Lu, H. Khan, S. Zakir and Ihsanullah et al., 2013. Health risks associated with heavy metals in the drinking water of Swat, Northern Pakistan. J. Environ. Sci., 25: 2003-2013.
CrossRefDirect Link - He, J. and J.P. Chen, 2014. A comprehensive review on biosorption of heavy metals by algal biomass: Materials, performances, chemistry and modeling simulation tools. Bioresour. Technol., 160: 67-78.
CrossRefDirect Link - Aydin, H., Y. Bulut and C. Yerlikaya, 2008. Removal of copper (II) from aqueous solution by adsorption onto low-cost adsorbents. J. Environ. Manage., 87: 37-45.
CrossRefDirect Link - Deng, P.Y., W. Liu, B.Q. Zeng, Y.K. Qiu and L.S. Li, 2013. Sorption of heavy metals from aqueous solution by dehydrated powders of aquatic plants. Int. J. Environ. Sci. Technol., 10: 559-566.
CrossRefDirect Link - Huang, Y., D. Wu, X. Wang, W. Huang, D. Lawless and X. Feng, 2016. Removal of heavy metals from water using polyvinylamine by polymer-enhanced ultrafiltration and flocculation. Separat. Purific. Technol., 158: 124-136.
CrossRefDirect Link - Xu, L., W. Ouyang, Y. Qian, C. Su, J. Su and H. Chen, 2016. High-throughput profiling of antibiotic resistance genes in drinking water treatment plants and distribution systems. Environ. Pollut., 213: 119-126.
CrossRefDirect Link - Kim, Y.D., D.H. Yim, S.Y. Eom, S.I. Moon and C.H. Park et al., 2014. Differences in the susceptibility to cadmium-induced renal tubular damage and osteoporosis according to sex. Environ. Toxicol. Pharmacol., 38: 272-278.
CrossRefPubMedDirect Link - Park, Y.H., D. Kim, J. Dai and Z. Zhang, 2015. Human bronchial epithelial BEAS-2B cells, an appropriate in vitro model to study heavy metals induced carcinogenesis. Toxicol. Appl. Pharmacol., 287: 240-245.
CrossRefPubMedDirect Link - Mohamed, S.A., M.F. Elshal, T.A. Kumosani, A.O. Mal and Y.M. Ahmed et al., 2016. Heavy metal accumulation is associated with molecular and pathological perturbations in liver of Variola louti from the Jeddah Coast of Red Sea. Int. J. Environ. Res. Public. Health, Vol. 13, No. 3.
CrossRefDirect Link - Gilani, S.R., S.R. Zaidi, M. Batool, A.A. Bhatti, A.I. Durrani and Z.Mahmood, 2015. Report: Central Nervous System (CNS) toxicity caused by metal poisoning: Brain as a target organ. Pak. J. Pharm. Sci., 28: 1417-1423.
PubMedDirect Link - Esmaeili, A., B. Saremnia and M. Kalantari, 2015. Removal of mercury(II) from aqueous solutions by biosorption on the biomass of Sargassum glaucescens and Gracilaria corticata. Arab. J. Chem., 8: 506-511.
CrossRefDirect Link - Sharma, P., H. Kaur, M. Sharma and V. Sahore, 2011. A Review on applicability of naturally available adsorbents for the removal of hazardous dyes from aqueous waste. Environ. Monit. Assess., 183: 151-195.
CrossRefPubMedDirect Link - Kannan, R.R., M. Rajasimman, N. Rajamohan and B. Sivaprakash, 2010. Brown marine algae Turbinaria conoides as biosorbent for Malachite green removal: Equilibrium and kinetic modeling. Front. Environ. Sci. Engin. China, 4: 116-122.
CrossRefDirect Link - Ibrahim, W.M., E.H. Salim, Y.A. Azab and A.M. Ismail, 2015. Monitoring and removal of cyanobacterial toxins from drinking water by algal-activated carbon. Toxicol. Ind. Health.
CrossRefDirect Link - Hassan, A.F., A.M. Abdel-Mohsen and M.M.G. Fouda, 2014. Comparative study of calcium alginate, activated carbon and their composite beads on methylene blue adsorption. Carbohydr. Polym., 102: 192-198.
CrossRefDirect Link - Pezoti, O., A.L. Cazetta, K.C. Bedin, L.S. Souza and A.C. Martins et al., 2016. NaOH-activated carbon of high surface area produced from guava seeds as a high-efficiency adsorbent for amoxicillin removal: Kinetic, isotherm and thermodynamic studies. Chem. Eng. J., 288: 778-788.
CrossRefDirect Link - Hasfalina, C.M., R.Z. Maryam, C.A. Luqman and M. Rashid, 2012. Adsorption of copper (II) from aqueous medium in fixed-bed column by Kenaf fibres. APCBEE Procedia, 3: 255-263.
CrossRefDirect Link - Shin, W.S. and Y.K. Kim, 2014. Biosorption characteristics of heavy metals (Ni2+, Zn2+, Cd2+, Pb2+) from aqueous solution by Hizikia fusiformis. Environ. Earth Sci., 71: 4107-4114.
CrossRefDirect Link - Bian, Y., Z. Bian, J. Zhang, A. Ding, S. Liu, L. Zheng and H. Wang, 2015. Adsorption of cadmium ions from aqueous solutions by activated carbon with oxygen-containing functional groups. Chinese J. Chem. Eng., 23: 1705-1711.
CrossRefDirect Link - Jayakumar, R., M. Rajasimman and C. Karthikeyan, 2015. Sorption and desorption of hexavalent chromium using a novel brown marine algae Sargassum myriocystum. Korean J. Chem. Eng., 32: 2031-2046.
CrossRefDirect Link - Lee, S.H. and C.H. Park, 2012. Biosorption of heavy metal ions by brown seaweeds from Southern Coast of Korea. Biotechnol. Bioprocess Eng., 17: 853-861.
CrossRefDirect Link - Ibrahim, W.M., 2011. Biosorption of heavy metal ions from aqueous solution by red macroalgae. J. Hazard. Mater., 192: 1827-1835.
CrossRefPubMedDirect Link - Escudero, C., N. Fiol, I. Villaescusa and J.C. Bollinger, 2009. Arsenic removal by a waste metal (hydr)oxide entrapped into calcium alginate beads. J. Hazard. Mater., 164: 533-541.
CrossRefPubMedDirect Link - Esmaeili, A. and A.A. Beni, 2015. Biosorption of nickel and cobalt from plant effluent by Sargassum glaucescens nanoparticles at new membrane reactor. Int. J. Environ. Sci. Technol., 12: 2055-2064.
CrossRefDirect Link - Donmez, G. and Z. Aksu, 2002. Removal of chromium(VI) from saline wastewaters by Dunaliella species. Process Biochem., 38: 751-762.
Direct Link - Langmuir, I., 1916. The constitution and fundamental properties of solids and liquids. Part I. solids. J. Am. Chem. Soc., 38: 2221-2295.
CrossRefDirect Link - Ding, Y., D.B. Jing, H.L. Gong, L.B. Zhou and X.S. Yang, 2012. Biosorption of aquatic cadmium (II) by unmodified rice straw. Bioresour. Technol., 114: 20-25.
CrossRefPubMedDirect Link - Hydari, S., H. Sharififard, M. Nabavinia and M.R. Parvizi, 2012. A comparative investigation on removal performances of commercial activated carbon, chitosan biosorbent and chitosan/activated carbon composite for cadmium. Chem. Eng. J., 193-194: 276-282.
CrossRefDirect Link - El Hassouni, H., D. Abdellaoui, S. El Hani and R. Bengueddour, 2014. Biosorption of cadmium (II) and copper (II) from aqueous solution using red alga (Osmundea pinnatifida) biomass. J. Mater. Environ. Sci., 5: 967-974.
Direct Link - Akbari, M., A. Hallajisani, A.R. Keshtkar, H. Shahbeig and S.A. Ghorbanian, 2015. Equilibrium and kinetic study and modeling of Cu(II) and Co(II) synergistic biosorption from Cu(II)-Co(II) single and binary mixtures on brown algae C. indica. J. Environ. Chem. Eng., 3: 140-149.
CrossRefDirect Link - Hammud, H.H., A. El-Shaar, E. Khamis and E.S. Mansour, 2014. Adsorption studies of lead by Enteromorpha algae and its silicates bonded material. Adv. Chem.
CrossRefDirect Link - Sarada, B., M.K. Prasad, K.K. Kumar and C.V.R. Murthy, 2014. Potential use of Caulerpa fastigiata biomass for removal of lead: Kinetics, isotherms, thermodynamic and characterization studies. Environ. Sci. Pollut. Res., 21: 1314-1325.
CrossRefPubMedDirect Link - Bouhamed, F., Z. Elouear, J. Bouzid and B. Ouddane, 2015. Multi-component adsorption of copper, nickel and zinc from aqueous solutions onto activated carbon prepared from date stones. Environ. Sci. Pollut. Res. Int.
CrossRefDirect Link - WHO, 2011. Guidelines for Drinking-Water Quality. 4th Edn., World Health Organization, Geneva, Switzerland, ISBN-13: 9789241548151, Pages: 564.
Direct Link








