ABSTRACT
Pinus kesiya Royle ex. Gord (Khasi pine) is an economically important early successional pine species predominant in the North-East- India. Some genotypes have tremendous biomass potential and oleoresin prospects. This study was aimed at developing a protocol for the genetic transformation of P. kesiya embryogenic tissue. This was achieved via the introduction of a bar-GUS cassette under the control of the ubiquitin promoter, through biolistic transfer. Expression of positive histochemical GUS activity (41%) in the bombarded embryogenic tissue was observed. PCR analysis of bar transgenes (46%) transformation efficiency indicated successful genetic modifications of P. kesiya embryogenic tissue by the pAHC25 plasmid. This is the first successful report of genetic transformation in P. kesiya indicating that Khasi pine is amenable to gene transfer.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/biotech.2007.86.92
URL: https://scialert.net/abstract/?doi=biotech.2007.86.92
INTRODUCTION
Recombinant DNA technology is a powerful tool for the introduction of foreign genes conferring traits such as virus, insect and herbicide resistance. The development of tissue culture and transformation systems for tree species including conifers has advanced significantly in the past decade (Le et al., 2001). Since the first transgenic tree in conifers was obtained by Huang et al. (1991), stable transformation has been developed for many forest tree species for example Picea glauca, P. mariana and Larix laricina using particle bombardment method (Ellis et al., 1993; Charest et al., 1996; Klimaszewska et al., 1997). However, transformation of conifers has traditionally been a slow and tedious process (Sederoff, 1995). The advent of particle transfer resulted in biolistics being commonly used for gene transfer in conifers and it is routinely used in many laboratories, although a low transformation frequency abounds. Biolistic transformations, in contrast to Agrobacterium (Malabadi and Nataraja, 2003), have resulted in fragmented or multicopy integration events of the transgene (Meyer, 1995; Walter et al., 1998; Nigro et al., 2004), which may lead to transgene silencing (Kumpatla et al., 1997).
Few conifers have yielded regenerated plants after transformation by microprojectile bombardment. These include Picea abies (Walter et al., 1999), Pinus radiata (Walter et al., 1998), Betula pendula (Valijakka et al., 2000) and Pinus patula (Nigro et al., 2004). Extensive literature exists on transformation studies, many of which used biolistic method such as P. banksiana (McAfee et al., 1993), P. pinea (Sul and Kurban, 1998), P. sylvestris (Aronen et al., 1996), P. taeda (Tang et al., 2001) and P. roxburghii (Parasharami et al., 2006). To our knowledge, there are no published reports for P. kesiya in the literature. Thus, the aims of this study were to develop a biolistic regime for P. kesiya embryogenic tissue through the introduction of the pAHC25 plasmid which contains the selectable herbicide resistance bar gene and the uidA reporter gene. An efficient and reliable transformation method (Malabadi and Nataraja, 2003) is also an important tool for the future applications in commercial forestry. Candidate genes can be identified and characterized and used for in molecular breeding. We report for the first time the successful delivery and expression of both the uidA and bar genes into P. kesiya embryogenic tissue.
MATERIALS AND METHODS
Plant material: Pinus kesiya (Royle ex. Gord) seeds of a genotype PK405, PK04 and PK214 of open pollinated trees were collected from the Forest Department, East Khasi Hills, Shillong, Meghalaya, India. Seeds were surface cleansed with 1% Citramide for 2 min and washed thoroughly with sterilized distilled water for three times. Seeds were further treated with sodium hypochlorite solution (4-5% available chlorine) for 2 min, rinsed 5 times with sterile double distilled water and treated with 6% hydrogen peroxide for 24 h. Prior to dissection of embryos, seeds were surface decontaminated sequentially with 0.1% HgCl2 for 2 min, immersed in 70% ethanol for 3 min and finally rinsed thoroughly five times with sterile distilled water (Malabadi et al., 2002, 2003).
Culture medium and initiation of embryogenic tissue: Triacontanol (CH3 (CH2)28 CH2OH) was obtained from Sigma (St Louis, USA). Stock solution of TRIA was prepared by dissolving 1 mg of RTIA in 0.75 mL of CHCl3 containing 1 drop of Tween 20 and this stock solution was gradually diluted with distilled water to the final volume of 200 mL (Tantos et al., 1999).
Embryogenic tissue was established according to our previous protocols (Malabadi et al., 2005). Mature zygotic embryos (Fig. 1A) of 3 genotypes (PK405, PK04 and PK214) were cultured individually on half strength inorganic salts MSG basal medium (Becwar et al., 1990) containing 2.0 g L-1 Gellan gum (Sigma), 90 mM maltose (Hi-media, Mumbai), 1 g L-1 L-glutamine, 1 g L-1 casein hydrosylate, 0.5 g L-1 meso-inositol, 0.2 g L-1 p-aminobenzoic acid and 0.1 g L-1 folic acid. The medium was supplemented with 10 μg L-1 TRIA (Malabadi et al., 2005). The cultures were raised in 25x145 mm glass culture tubes (Borosil) containing 15 mL of the medium and maintained in dark for 4-6 weeks at 25±3°C. Nutrientmedium without TRIA served as a control. The pH of the medium was adjusted to 5.8 with NaOH or HCl before Gellan gum was added. The medium was then sterilized by autoclaving at 121°C and 1.08 kg cm-2 for 15 min. L-glutamine, p-aminobenzoic acid and TRIA were filter sterilized and added to the media after it had cooled to below 50°C.
All the cultures were examined for the presence of embryonal suspensor masses by morphological and cytological observations of callus. The cultures showing white mucilaginous embryogenic tissue were identified and subcultured on the initiation medium (Fig. 1B) for further two weeks for the better development of embryonal suspensor masses. The half-strength inorganic salts MSG basal medium (Becwar et al., 1990) supplemented with 9.0 μM 2, 4-D and 10 μg L-1 (Initiation medium) (Malabadi et al., 2005) was used for this purpose.
Maintenance of embryogenic tissue: The white mucilaginous embryogenic tissue developed on the above initiation medium (I) was subcultured on maintenance medium (II). The half-strength (inorganic salts) MSG basal medium containing 130 mM maltose, 4 g L-1 Gellan gum and supplemented with 2 μM 2, 4-D and 2 μg L-1 (maintenance medium) (Malabadi et al., 2005) was used for this purpose. On the maintenance medium, the embryogenic tissue containing embryonal suspensormasses was maintained for 3 weeks with two subcultures. All the cultures were maintained in dark and microscopic observation of cultures was conducted to ensure the development of pro-embryo.
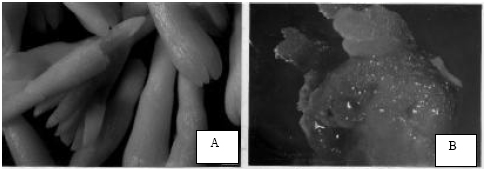 | |
| Fig. 1: | Recovery of transgenic plants of genotype PK04 of Pinus kesiya after bombardment of embryogenic tissue raised on MSG basal medium supplemented with TRIA. A- Group of mature zygotic embryos dissected from seed (scale bar 10 mm = 0.95 mm). B- Re-growth of bombarded white mucilaginous embryogenic callus on initiation medium (scale bar 10 = 8 mm). C- Development of advanced cotyledonary somatic embryos on maturation medium after bombardment (10 = 9.3 mm). D- Somatic seedlings ready for transfer |
Establishment of suspensor cultures: The embryogenic tissue was initiated into suspension culture, which was also used for bulking up of tissue before bombardment Embryogenic suspension cultures were initiated by inoculating approximately 200 mg of friable tissue into 50 mL liquid MSG medium. The flasks were stoppered with a sterile cotton wool bung and covered with aluminum foil. The cultures were placed on a rotary shaker at 120 rpm in the dark at 25°C for 8 days, or until pro-embryos were observed.
Maturation of somatic embryos: After the final phase of selection, filter paper discs supporting bombarded material were transferred to maturation medium to induce cotyledonary embryo development. The half strength (inorganic salts) MSG basal medium supplemented with 180 mM Maltose, 60 μM ABA and 8 g L-1 Gellan gum (maturation medium III) was tested for this purpose (Malabadi et al., 2005). All the cultures were again maintained in the dark for 8 to 12 weeks. Somatic embryos further matured to exhibit a hypcotyl and cotyledonary development.
Gene construct for transformation: The plasmid construct pAHC25 (Christensen and Quail, 1996) was used in this transformation study. This vector consisted of both the selectable marker, bar, which encodes for phosphinothricin acetyltransferase (De Block et al., 1987) and the GUS reporter gene encoding β-glucuronidase (Jefferson et al., 1987) each fused between the Zea ubiquitin promoter and the nos terminator. An eukaryotic intron sequence has also been inserted between the bar gene and its promoter, ensuring that bialaphos resistance and β-glucuronidase activity can only be expressed by transgenic plant material and not by residual bacterial contaminants. The Ubi-Bar chimaeric gene provides selection for transformants resistant to BASTA® herbicide (De Block et al., 1987).
Treatment of embryogenic tissue: Using the embryogenic tissue derived from suspension culture, embryonal suspensor masses, were filtered in 1.5 mL aliquots onto Whaman No. 1 filter paper supports and placed onto MSG solid medium supplemented with 90 mM maltose as an osmoticum (Malabadi et al., 2003) or no maltose (untreated) and both treatments left on the laminar flow bench overnight. The target tissues (liquid medium-derived cultures) were bombarded after 0, 5, 10 and 14 days growth on solid medium and subcultured onto selection medium the following day. A stepwise selection regime was implemented; consisting of the inclusion of 1 mg L-1 followed by 3 mg L-1 BASTA ® herbicide, a bioactive ingredient (glufosinate ammonium) in the medium at each subculture. BASTA is a water-soluble and contains an active ingredient of glufosinate ammonium at 200 g L-1.
DNA coating of microparticles: One hundred mg of 1.5 μm tungsten microparticles (ELAK Ltd, Hungary) were sterilized by overnight incubation in 2 mL 70% ethanol (v/v). The particles were briefly spun down at 2400 x g. The ethanol was removed and the micropaticles were washed twice with 2 mL sterile dH2O. The sterile particles were stored in sterile 50% glycerol (v/v) solution at -20°C. Macro-particles were stored in 100% ethanol overnight, placed onto an autoclaved Petri dish and left to air dry. Plasmid DNA was isolated as described by Li et al. (1995) and then coated onto the tungsten particles using the Perl et al. (1992) method to obtain a concentration of 4 μg DNA mg-1 tungsten particles.
Particle bombardment: All the experiments were performed using a gene gun (Gene booster, Germany) with a nitrogen-driven biolistic delivery system. The filtered tissue was bombarded with 10 μL of DNA-coated particles at 40 bar gas pressure per shot and -0.40 bar vacuum in the Genebooster chamber. The micro carrier travel distance was 70 mm from the stopping plate to the target tissue.
GUS assay: Random samples of bombarded material were histochemically stained with 0.3% 5-bromo-4-chloro-3-indolyl β-glucuronide (X-glcA) (w/v) buffer (X-glcA, Sigma), 5 mM K-ferrocyanide, 5mM K-ferricyanide, 0.005% Triton X-100 (v/v), 100 mM Na-phosphate buffer (0.5 M NaH2PO4. 2H2O, pH 7), dissolved in methanol) (Jefferson, 1987) at pH 7.0 for 6 h to overnight at 37°C and then viewed under a photomicroscope.
DNA extraction: Genomic P. kesiya DNA for PCR amplification was extracted after bombarded material had undergone selection at 3 mg L-1 BASTA® boactive ingredient (approximately 3 weeks after particle bombardment) by grinding 0.1 g embryogenic tissue with liquid nitrogen to a fine powder, using a pestle and mortar. The cellular powder was transferred, to sterile 1.5 mL microfuge tubes in which 500 μL urea extraction buffer (7 M urea crystals, 5 M NaCl, 1M Tris/Cl, pH 8.0), 0.5 M EDTA, 20% Sarkosyl (v/v) (British Drug House (BDH), England) was placed and then vortexed for 10 sec (Nigro et al., 2004). A ratio of 1:1 phenol to chloroform was added to the cell extract and shaken on a tabletop shaker at 120 rpm for 1 h at room temperature. After centrifugation (15 min at 15, 000 x g), the supernatant was then transferred to fresh microfuge tubes. The nucleic acids were precipitated and an equal volume, ice-cold isopropanol, mixed well by inversion and placed at -20°C for 15 min to precipitate the DNA. Nucleic acids were collected by 15 min centrifugation at 15,000 x g and subsequently purified using 70% ethanol, washed with 100% ethanol prior to air-drying for 3-5 min on a laminar flow bench (Nigro et al., 2004). Isolated genomic DNA was stored in 20 μL ultra water at -20°C until further use.
PCR-mediated gene detection: The PCR primer set 5’-GGTGGGAAGCGCGTTACAAG-3’ and 5’-GTTTACGCGTTGCTTCCGCCA-3’ was used to amplify the uidA gene and yielded a fragment of 1.2 kb after electrophoresis on 0.8% agarose (w/v). A GUS amplification cocktail, consisted of a 50 μL reaction with 100 ng genomic template DNA, 1.25 units of Taq DNA polymerase (Roche biochemicals), 0.5 μM of each primer, 10 mM of each dNTP: dATP, dTTP, dCTP and dGTP and 5 μL PCR buffer (Roche biochemicals). To enhance the efficiency of the PCR, 10% dimethyl sulphoxide (DMSO) (v/v) was also included in the reaction mixture (Nigro et al., 2004). The PCR contents were mixed well and all samples were overlaid with an equal volume of paraffin oil prior to undergoing 36 amplification cycles (Hybaid Thermal Reactor, Hybaid Ltd., England). The PCR was initiated with a denaturation step of 94°C for 1 min at the beginning of the cycling regime. This was then followed by 35 cycles each comprising of a 94°C denaturing temperature (30 sec), a 60°C annealing step (30 sec) and a 72°C extension step (45 sec). The final stage employed the same denaturation and annealing conditions as described above but the last primer extension step was increased to a 5 min (Nigro et al., 2004).
The bar gene was successfully amplified as described by Vickers et al. (1996) and Nigro et al. (2004) using Expand TM High Fidelity Taq DNA polymerase (Roche biochemical’s) with the bar primer 5’-ATATCCGAGCGCCTCGTGCATGCG-3’ (Roche products) designed for use with pAHC25 construct by Wan and Lemaux (1994) and yielded a 0.34 kb fragment (Fig. 2) if template was present. The bar gene products were analysed on a 1.5% agarose (w/v) gel (0.04 M Tris-Acetate, 0.002 M EDTA, pH 8.5) in TAE buffer after PCR.
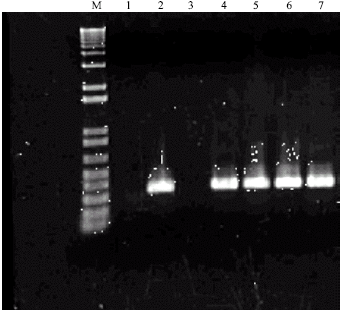 | |
| Fig. 2: | PCR mediated amplification for bar gene products described by Vickers et al. (1996) and Nigro et al. (2004). The DNA contents of lane are: Molecular weight marker M (Roche Biochemicals). Lane 1 = Genomic sample of unbombarded Pinus kesiya. Lanes 2, 4, 5, 6, 7 = Genomic samples of bombarded Pinus kesiya of genotype PK04 showing the integration of bar gene at 340 bp (3.4 kb). Lane 3 = No genomic DNA included in the reaction |
| Table 1: | Recovery of transgenic somatic seedlings following the bombardment of embryogenic tissue cultured on MSG basal medium supplemented with 3 mg L-1 of BASTA in three genotypes of Pinus kesiya |
 | |
| * Mean±SE followed by the same letter were not significantly different at p≤0.05 | |
Statistical analysis: In above experiments each culture tube received a single zygotic embryo. Each replicate contained 50 cultures and one set of experiment is made up of two replicates (total 100 cultures for one experiment) for each genotype of Pinus kesiya. All the experiments were repeated three times (300 cultures for one set of experiment for 3 genotypes). Data presented in the Table 1 was analyzed for significance using ANOVA and the differences contrasted using Duncan’s multiple range test. All the statistical analysis was performed at the 5% level using SPSS statistical software package.
RESULTS AND DISCUSSION
In this study, the growth of bombarded embryogenic tissue of all the three genotypes of P. kesiya was inhibited at 4 mg L-1 of glufosinate ammonium (BASTA). On the other hand lower concentrations (1, 2 and 3 mg L-1) of BASTA showed the growth of bombarded embryogenic tissue (data not shown). However, in order to reduce the toxicity to regenerating or recovering bombarded embrogenic tissue, the entire selection medium was incorporated with 3 mg L-1 of BASTA active ingredient. The white mucilaginous bombarded embryogenic tissue was subcultured on the maintenance medium for the further development of embryonal suspensor masses. The pro-embryos developed on the maintenance medium could not grow further, until they were transferred on a medium with enhanced maltose, ABA and Gellan gum respectively. The half strength (inorganic salts) MSG basal medium supplemented with 180 mM Maltose, 60 μM ABA and 8 g L-1 Gellan gum (maturation medium III) was tested for this purpose (Malabadi et al., 2005). The bombarded embryogenic tissue developed somatic embryos on maturation medium after a period of 10 to 12 weeks (Fig. 1C). The percentage of somatic embryogenesis was not similar in all the three genotypes of Pinus kesiya (Table 1). Highest percentage of somatic embryogenesis (13%) was recorded in a genotype PK04, with a total number of 11 somatic seedlings recovered per gram fresh weight of bombarded embryogenic tissue (Fig. 1 B and C) (Table 1). On the other hand in the rest of the two genotypes (PK405 and PK214), the bombarded embryogenic tissue showed decreased maturation potential and tissue becoming highly mucilaginous was observed after several months in culture, irrespective of particle transfer. However, genotype PK405 showed decreased percentage (2% in PK405) of somatic embryogenesis, with a total number of 7 somatic seedlings recovered per gram fresh weight of bombarded embryogenic tissue (Table 1). Furthermore, the bombarded embryogenic tissue of genotype PK214 failed to produce somatic embryos and resulted in the browning and ultimate death of the tissue on the maturation medium (Table 1). After 9 to 12 weeks of maturation, the advanced cotyledonary somatic embryos were picked up for the germination. After 6 weeks on germination medium (Fig. 1D), the plantlets were recovered and hardened.
Bombarded embryogenic tissue samples exhibited a range of expression strength of the β-glucuronidase enzyme, although higher magnification revealed that the embryonal heads had expressed the transient GUS activity and had turned a turquoise-blue color (data not shown). These results indicated that embryogenic tissue of this genotype was amenable to genetic transformation and the GUS reporter gene could be incorporated and expressed in the P. kesiya genome. The smaller bar amplicon was resolved at 0.34 kb (Fig. 2) using the PCR regime described by Vickers et al. (1996) and Nigro et al. (2004). Of the 100 samples tested, 46 contained positive bar amplicons resulting in higher transformation efficiency (46%) (Fig. 2) than GUS (41%). Perhaps the smaller gene was easier to incorporate into the genome and was expressed at high rate during selection. This indicated that co-integration of both the reporter GUS gene and the herbicide resistant bar gene did not always occur.
In conclusion, this study has successfully established a biolistic gene transfer regime. Transformation was confirmed by PCR and histochemical tests for GUS and bio-tests for continued BASTA resistance. Furthermore, a comprehensive examination into the use of Agrobacterium as a vessel for gene transfer is in progress in our laboratories. This would extend the scope of genetic transformation of P. kesiya as with other investigations in a variety of tree species (Holland et al., 1997; Tang et al., 2001; Nigro et al., 2004, Parasharmi et al., 2006). This transformation protocol for the first time provides a platform towards the commercial exploitation of transgenic P. kesiya, as well as the use of transgenic studies for the advancement of our fundamental understanding of the molecular physiology of coniferous trees.
ACKNOWLEDGMENTS
We are grateful to the Head, Department of Botany for providing all the facilities for this study. Rinu Thomas, Nancy and Savitha are warmly acknowledged for every help during the experiments.
REFERENCES
- Becwar, M.R., R. Nagmani and S.R. Wann, 1990. Initiation of embryogenic cultures and somatic embryo development in Loblolly pine (Pinus taeda). Can. J. Res., 20: 810-817.
Direct Link - Charest, P.J., Y. Devantier and D. Lachance, 1996. Stable genetic transformation of Picea mariana (black Spruce) via particle bombardment. In Vitro Cell Dev. Biol. Plant, 32: 91-99.
Direct Link - Christensen, A.H. and P.H. Quail, 1996. Ubiquitin promoter-based vectors for high-level expression of selectable and/or screenable marker genes in monocotyledonous plants. Transgenic Res., 5: 213-218.
CrossRefPubMedDirect Link - De Block, M., J. Botterman, M. Vanderwiele, J. Dockx and C. Thoen et al., 1987. Engineering herbicide resistance in plants by expression of a detoxifying gene. EMBO J., 6: 2513-2518.
Direct Link - Ellis, D.D., D.E. McCabe, S. McInnis, R. Ramachandran and D.R. Russell et al., 1993. Stable transformation of Picea glauca by particle acceleration. Biotechnol., 11: 84-89.
Direct Link - Holland, L., J.E. Gemmell, J.A. Charity and C. Walter, 1997. Foreign gene transfer into Pinus radiata cotyledons by Agrobacterium tumefaciens. New Zealand J. For. Sci., 27: 289-304.
Direct Link - Jefferson, R.A., 1987. Assaying chimeric genes in plants: The GUS gene fusion system. Plant Mol. Biol. Rep., 5: 387-405.
CrossRefDirect Link - Jefferson, R.A., T.A. Kavanagh and M.W. Bevan, 1987. GUS fusions: Beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J., 6: 3901-3907.
Direct Link - Klimaszewska, K., Y. Devantier, D. Lachance, M.A. Lelu and P.J. Charest, 1997. Larix laricina (tamarack): Somatic embryogenesis and genetic transformation. Can. J. For. Res., 27: 538-550.
CrossRef - Kumpatla, S.P., W. Teng, W.G. Bucholz and T.C. Hall, 1997. Epigenetic transcriptional silencing and 5-azacytidine-mediated reactivation of a complex transgene in rice. Plant Physiol., 115: 361-373.
Direct Link - Le, V.Q., J. Belles-Isles, M. Dusabenyagasani and F.M. Tremblay, 2001. An improved procedure for production of white spruce (Picea glauca) transgenic plants using Agrobacterium tumefaciens. J. Exp. Bot., 52: 2089-2095.
Direct Link - Li, X.Q., R.C. Stahl and G. Brown, 1995. Rapid micropreps and minipreps of Ti plasmids and binary vectors from Agrobacterium tumefaciens. Trans. Res., 4: 349-351.
CrossRef - Malabadi, R.B., H. Choudhury and P. Tandon, 2004. Initiation, maintenance and maturation of somatic embryo from thin apical dome section in Pinus kesiya (Royle ex. Gord) promoted by partial desiccation and Gellan gum. Sci. Hortic., 102: 449-459.
Direct Link - McAfee, B.J., E.E. White, L.E. Pelcher and M.S. Lapp, 1993. Root induction in pine (Pinus) and larch (Larix) sp., using Agrobacterium rhizogenes. Plant Cell Tissue Organ Cult., 34: 53-62.
CrossRef - Nigro, S.A., N.P. Makunga, N.B. Jones and J. van Staden, 2004. A biolistic approach towards producing transgenic Pinus patula embryonal suspensor masses. Plant Growth Regul., 44: 187-197.
Direct Link - Parasharami, V.A., V.B. Naik, S. von Arnold, R.S. Nadgauda and D.H. Clapham, 2006. Stable transformation of mature zygotic embryos and regeneration of transgenic plants of chir pine (Pinus roxburghii sarg.). Plant Cell Rep., 24: 708-714.
Direct Link - Perl, A., H. Kless, A. Blumenthal, G. Galili and E. Galun, 1992. Improvement of plant regeneration and GUS expression in scutellar wheat calli by optimization of culture conditions and DNA-microprojectile delivery procedures. Mol. Gene Genet., 235: 279-284.
CrossRefDirect Link - Sul, I.W. and S.S. Kurban, 1998. Influence of bombardment with BA-coated microprojectiles on shoot organogenesis from Phlox paniculata L. and Pinus pinea L. tissues. In Vitro Cell Dev. Biol. Plant, 34: 300-302.
Direct Link - Tang, W., R. Sederoff and R. Whetten, 2001. Regeneration of transgenic loblolly pine (Pinus taeda) from zygotic embryos transformed with Agrobacterium tumefaciens. Plant Cell Rep., 22: 1-15.
Direct Link - Tantos, A., A. Meszaros and J. Kissimon, 1999. The effect of triacontanol on micropropagation of balm, Melissa officinalis L. Plant Cell Rep., 19: 88-91.
Direct Link - Vickers, J.E., G.C. Graham and R.J. Henry, 1996. A protocol for the efficient screening of putatively transformed plants for bar, the selectable marker gene, using the polymerase chain reaction. Plant Mol. Biol. Rep., 4: 363-368.
CrossRef - Walter, C., L.J. Grace, A. Wagner, D.W.R. White and A.R. Walden et al., 1998. Stable transformation and regeneration of transgenic plants of Pinus radiata D. Plant Cell Rep., 17: 460-468.
Direct Link - Walter, C., L.J. Grace, S.S. Donaldson, J. Moody and E. Gemmell et al., 1999. An efficient biolistic transformation protocol for Picea abies embryogenic tissue and regeneration of transgenic plants. Can. J. For. Res., 29: 1539-1546.
CrossRef









