ABSTRACT
Background and Objective: Sclerotium rolfsii is a soil-borne pathogen that has a very wide host range, including chilli plants. This fungus causes damping-off and stems rot diseases. The objective of the research was to evaluate the effect of different storage times of Trichoderma harzianum alginate pellet with rice bran on its viability and its potential to control damping-off disease caused by S. rolfsii on chilli. Materials and Methods: The design used in this research was a Completely Random Design with seven treatments and four replications. The treatments were different storage times of alginate pellet T. harzianum (0, 2, 4, 6, 8, 10 and 12 weeks). Observation parameters were viability of alginate pellet, percentage of pre-emergence damping-off, percentage of post-emergence damping-off and number of T. harzianum propagules in soil. Results: The results showed that the different storage times of alginate pellet T. harzianum until 12 weeks did not affect its viability and inhibited the pre and post-emergence damping-off diseases. Also, the numbers of propagules of T. harzianum in the soil after 30 days of seeding at different treatments were still high and did not differ among them. Conclusion: The storage of T. harzianum alginate pellet for up to 12 weeks was still stable and able to suppress the development of S. rolfsii damping-off on chilli plant.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2022.203.207
URL: https://scialert.net/abstract/?doi=ajps.2022.203.207
INTRODUCTION
The fungus Sclerotium rolfsii is a soil-borne pathogen that has a very wide host range, including chilli plants1. This fungus attacks chilli plants ranging from the seedling phase, known as a damping-off disease to adult plants known as stem rot disease which causes death in a relatively short time. This fungus is difficult to control because it forms the sclerotium, a viable structure that can survive years in the soil without a host plant or in extreme environmental conditions1,2.
The control efforts carried out are generally more emphasized on a chemical control using fungicides. The continuous use of fungicides can cause various negative impacts such as the death of non-target organisms, pathogen resistance and the emergence of residual effects. Therefore there is a need to find an environmentally friendly control such as the use of Trichoderma harzianum as biocontrol agents. The fungus T. harzianum is an antagonistic fungus with various antagonistic mechanisms such as parasitism, competition and antibiosis3 and it has been tested for its ability to suppress several soil-borne pathogens, including S. rolfsii 4.
The introduction of the biological agent T. harzianum requires basic food to adapt to new environments. Besides that, the formulation is one of the main keys that determine the success of a biocontrol agent in controlling plant pathogens. One of the objectives of the biocontrol agent formulation is to stabilize microorganisms during production, distribution and storage5. Many research results showed that T. harzianum pellets can suppress the attack of various pathogenic fungi such as Fusarium oxysporum f. sp. lycopersici causing wilt disease on tomatoes6 and Helminthosporium oryzae causing brown spots on rice7.
This study aimed to determine the ability of T. harzianum alginate pellets with rice bran in suppressing S. rolfsii attacks on chilli plants and the effect of T. harzianum pellet storage time on S. rolfsii attacks.
MATERIALS AND METHODS
Research site: This research was carried out in the Phytopathology Laboratory and Green House, Agriculture Faculty, Andalas University from July to December, 2019. This study consisted of 2 stages, namely:
- Effect of the storage time of T. harzianum pellets on its viability
- Effect of the storage time of T. harzianum alginate pellets on S. rolfsii attacks on chilli plant
Effect of the storage time of alginate pellet formulation of T. harzianum on its viability: The design used in this experiment was a Completely Randomized Design (CRD) with 7 treatments and 3 replications. The treatments consisted of several different storage times of alginate pellet formulation of Trichoderma harzianum with rice bran namely A (0 weeks), B (2 weeks), C (4 weeks), D (6 weeks), E (8 weeks), F (10 weeks) and G (12 weeks).
Preparation of alginate pellet formulation of T. harzianum: The rice bran was mashed with a coffee grinder and the suspension was made by mixing 63 g of rice bran with 450 mL of sterile distilled water. Meanwhile, alginate suspension was prepared by blending 9 g of alginate with 450 mL of sterile distilled water for 1 min. Both suspensions were sterilized by using an autoclave for 20 min at 121°C, mixed and added 150 mL T. harzianum suspension (106 conidia mL–1). The mixture was shaken for 5 min. Furthermore, this suspension mixture was added dropwise to 0.2 M CaCl2. After 20 min the pellets are collected on 1mm pore size plastic mesh and washed with running water and dried for 24 hrs at room temperature8.
Viability of T. harzianum on alginate pellets: Twenty-five pellets were arranged on Petri dishes (9 mm) containing PDA and each treatment consisted of 3 Petri plates and then incubated for 1 week. Observation of the viability of T. harzianum in alginate pellets was started on the second to the eighth day after incubation.
Effect of storage time of alginate pellet formulation of T. harzianum on S. rolfsii attack: The design used in this study was a Completely Randomized Design (CRD) with 8 treatments and 3 replications. The treatments consisted of several different storage times for T. harzianum pellets, namely A (without pellet), B (without storage), C (2 weeks), D (4 weeks), E (6 weeks), F (8 weeks), G (10 weeks) and H (12 weeks).
Isolation and identification of S. rolfsii: S. rolfsii isolate was isolated from infected chilli plants. Isolation was carried out using a single spore method. After obtaining a pure culture it was identified using the literature by Watanabe9.
Propagation of S. rolfsii: S. rolfsii were cultured massively in Corn Meal Sand (CMS) medium for 10 days by placing the medium in 100 g of heat-resistant plastic bags and giving the bag a plastic pipe ring and gagging it with cotton. Then it was sterilized in an autoclave for 20 min at a temperature of 121°C for 2 consecutive days. After sterilization, 1 cm2 of S. rolfsii (7 days) on PDA medium was placed in plastic bags.
Seeding of chilli seeds and Infestation of S. rolfsii: The seeding medium used was a mixture of soil and chicken manure (1:1 v/v), which has been sterilized by the Tyndallization method. The medium was placed in a seedbed (15×15×45 cm) as much as 4 kg per seedbed. Then seeds were seeded as much as 75 seed/seedbed.
Infestation of S. rolfsii was carried out 2 weeks before seeding by mixing 30 g of S. rolfsii in a CMS substrate with 1 kg of soil.
Introduction of T. harzianum alginate pellet: The introduction of T. harzianum alginate pellets from various storage periods (according to treatment) was carried out one week before seeding as much as 5 g kg–1 soil by mixing T. harzianum pellets with soil.
Observation parameters
Percentage of infected seeds before emerged to the soil surface (pre-emergence damping-off): This observation was carried out 30 days after seeding. The percentage inhibition of pre-emergence damping-off was calculated using the following formula by Kumar et al.10:
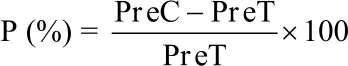 |
where, P is the Percentage inhibition, PreC is the Percentage of pre-emergence damping-off in control and PreT is the Percentage of pre-emergence damping-off in treatments.
Percentage of infected seeds after emerged to the soil surface (post-emergence damping-off): This observation was carried out 30 days after seeding. The percentage inhibition of post-emergence damping-off was calculated using the following Eq:
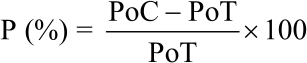 |
where, P is the Percentage inhibition, PoC is the Percentage of pre-emergence damping-off in control and PoT is the Percentage of pre-emergence damping-off in treatments.
Amount of T. harzianum propagules in the soil: The amount of T. harzianum propagules in the soil was counted 30 days after seeding.
RESULTS
T. harzianum viability on alginate pellets: Different storage times of alginate pellet formulation of T. harzianum with rice bran did not affect on viability of T. harzianum. The viability of T. harzianum in all treatments of T. harzianum pellet storage up to 12 weeks were 100% as showed in Table 1.
T. harzianum effect on alginate pellets on pre-emergence damping-off disease: Application of T. harzianum alginate pellets at all different storage times significantly reduced the number of infected seeds before they emerge to the soil surface (pre-emergence damping- off). Percentage of infected seeds on control (without T. harzianum alginate pellet) was 13.66% and significantly different from other treatments. Statistically, there was no effect of pellet storage time on the number of infected seeds before they emerge to the soil surface. Percentage inhibition of pre-emergence damping-off ranged from 70.71-92.69% in Table 2.
T. harzianum effect on alginate pellets on post-emergence damping-off disease: The result of this study also indicated that application of T. harzianum alginate pellets at different storage times significantly reduced the number of infected seeds after they emerged to the soil surface (post-emergence damping- off).
| Table 1: | Viability of T. harzianum alginate pellet on medium PDA |
| Treatments (storage time of pellet) | Viability T. harzianum (%) |
| Control (without storage) | 100 |
| 2 weeks | 100 |
| 4 weeks | 100 |
| 6 weeks | 100 |
| 8 weeks | 100 |
| 10 weeks | 100 |
| 12 weeks | 100 |
| Table 2: | Percentage of infected seeds before emerged to the soil surface (pre-emergence damping off) |
| Treatments (storage time of pellet) | Pre-emergence damping-off (%) | Inhibition (%) |
| Without pellet | 13.66a | - |
| Without storage time | 3.33b | 75.62 |
| 2 weeks | 2.00b | 85.36 |
| 4 weeks | 1.00b | 92.69 |
| 6 weeks | 3.33b | 75.62 |
| 8 weeks | 3.66b | 72.20 |
| 10 weeks | 4.00b | 70.71 |
| 12 weeks | 3.00b | 78.04 |
| Numbers within the same column followed by the same letters are not significantly different (p = 0.05) | ||
| Table 3: | Percentage of infected seeds after emerged to the soil surface (post-emergence damping-off) |
| Treatments (storage time of pellet) | Post-emergence damping-off (%) | Inhibition (%) |
| Without pellet | 24.66a | - |
| Without storage time | 0.66b | 97.32 |
| 2 weeks | 2.00b | 89.21 |
| 4 weeks | 6.00b | 76.56 |
| 8 weeks | 1.66b | 93.27 |
| 10 weeks | 2.66b | 89.21 |
| 12 weeks | 2.33b | 90.55 |
| 12 weeks | 3.33b | 86.50 |
| Numbers within the same column followed by the same letters are not significantly different (p = 0.05) | ||
| Table 4: | Number of T. harzianum propagules in the soil 30 days after seeding |
| Treatments (storage | Number of T. harzianum |
| time of pellet) | propagules/gram soil |
| Without pellet | 7.20*102a |
| Without storage time | 1.33*106b |
| 2 weeks | 0.85*106b |
| 4 weeks | 0.80*106b |
| 6 weeks | 0.52*106b |
| 8 weeks | 1.05*106b |
| 10 weeks | 0.90*106b |
| 12 weeks | 0.40*106b |
| Numbers within the same column followed by the same letters are not significantly different (p = 0.05) | |
Percentage of post-emergence damping-off on control was 24.66% while those of other treatments were ranged from 1-4%. Statistically, there was no effect of pellet storage times on the number of infected seeds after they emerged from the soil surface. Percentage inhibition of post-emergence damping-off was ranged from 76.56-97.32 in Table 3.
Number of T. harzianum propagules in the soil: Application of T. harzianum alginate pellet was able to increase the number of T. harzianum propagules in the soil observed 30 days after application. The difference in pellet storage times did not show a statistically significant effect on the number of propagules formed in Table 4.
DISCUSSION
The results of this study showed that the storage time of alginate pellet of T. harzianum up to 12 weeks did not affect the viability of T. harzianum as shown in Table 1. This happened because T. harzianum was immobilized in certain carriers, alginate and rice bran that made its shelf life increase. This is following what was suggested by Harman11 that Trichoderma culture should be entrapment in certain carriers and should be prepared as formulations for easy application and storage. The results of research by Bhai4 also showed that the conidial and mycelial stages in encapsulated form remained viable for more than six years at room temperature.
Application of alginate pellet of T. harzianum at different storage times could suppress the pre and post-emergence damping-off diseases however the different storage times of pellets did not show the different ability in suppressing the disease. The ability to suppress the diseases because T. harzianum is an antagonistic fungus with various antagonistic mechanisms such as parasitism, competition and antibiosis12. Trichoderma strains can produce extracellular enzymes and antifungal antibiotics and they can also be competitors to plant pathogenic fungi, stimulating plant development and inducing resistance13. Mayurama et al.8 also stated that the T. harzianum encapsulation showed an improvement in the chitinolytic and cellulosic activity. Research results by Kucuk and Kivanc13 showed that alginate encapsulation of Trichoderma successfully improved chitinase and -1, 3-glucanase production in vitro for a prolonged time even at higher temperature (40°C). Attitalla and Salleh14 also stated that the immobilized Trichoderma improved chitinase production compared with free spore suspension especially when chitin was used as a carbon source. Alginate encapsulated Trichoderma did not only prolong the metabolic activity of the entrapped organism but also it promoted the slow release of microbial spores into the medium for successful enzyme production15.
The ability of T. harzianum alginate pellets to suppress the attack of plant pathogenic fungi has been shown by many researchers such as Rhizoctonia solani on beet seed16 and clonal propagation of Eucalyptus17, Sclerotinia sclerotiorum on potato18, Fusarium oxysporum f. sp. lycopersici on tomatoes6 and Helminthosporium oryzae on rice14. The result of research by Kucuk and Kivanc13 also showed that encapsulated fungus was able to provide greater control of S. sclerotiorum in vitro test.
Differences in the duration of storage of T. harzianum pellets did not affect the suppression of the S rolfsii attack on chilli. This happened because the T. harzianum pellets stored for up to 12 weeks were still stable as evidenced by their viabilities that were not different as shown in Table 1. This was also supported by the number of T. harzianum propagules in the soil 30 days after the introduction of the pellets did not show a significant difference among treatments.
CONCLUSION
The viability of the Trichoderma harzianum alginate pellet was stable until 12 weeks of storage. Application of T. harzianum alginate pellet at different storage times as much as 5 g kg–1 of soil reduced Pre-emergence damping-off and Post-emergence damping-off diseases by S. rolfsii on chilli plants with percentage inhibition above 70 and 76%, respectively. Storage of T. harzianum alginate pellets at different times did not affect their effectiveness in reducing S. rolfsii attacks on chilli plants. Also, the different storage times of T. harzianum alginate pellets did not affect the number of T. harzianum propagules in the soil 30 days after the introduction of the pellet.
SIGNIFICANCE STATEMENT
This study discovered the ability of Trichoderma harzianum alginate pellet with rice bran at different storage times up to 12 weeks in reducing damping-off on chilli that can be beneficial for alternative control damping-off. Storage of T. harzianum pellet up to 12 weeks did not affect its ability in reducing the damping off disease.
REFERENCES
- Agrios, G.N., 2005. Plant Pathology. 5th Edn., Elsevier Academic Press, United States, ISBN-13: 9780080473789, Pages: 952.
Direct Link - Cilliers, A.J., Z.A. Pretorius and P.S.Van Wyk, 2003. Integrated control of Sclerotium rolfsii on groundnut in South Africa. J. Phytopathol., 151: 249-258.
CrossRefDirect Link - Pal, K.K. and B.M.S. Gardener, 2006. Biological control of plant pathogens. Plant Health Instr.
CrossRefDirect Link - Bhai, R.S., 2020. Preservation and long-term storage of Trichoderma spp. by sodium alginate encapsulation. J. Plant. Crops, 48: 36-44.
CrossRefDirect Link - Leggett, M., J. Leland, K. Kellar and B. Epp, 2011. Formulation of microbial biocontrol agents-an industrial perspective. Can. J. Plant Pathol., 33: 101-107.
CrossRefDirect Link - Anuar, I.S.M., K.A.K. Sulong, H.A. Ghani and M.Z. Wahab, 2020. Alginate encapsulation of Trichoderma harzianum against brown spot disease on rice (Oryzae sativa) in vivo assays. Food Res., 4: 138-141.
CrossRefDirect Link - Maruyama, C.R., N. Bilesky-José, R. de Lima and L.F. Fraceto, 2020. Encapsulation of Trichoderma harzianum preserves enzymatic activity and enhances the potential for biological control. Front. Bioeng. Biotechnol., Vol. 8.
CrossRefDirect Link - Watanabe, T., 2010. Pictorial Atlas of Soil and Seed Fungi: Morphologies of Cultured Fungi and Key to Species. CRC Press, United States, ISBN-13: 9781138627109, Pages: 426.
Direct Link - Kumar, S., M. Thakur and A. Rani, 2014. Trichoderma: Mass production, formulation, quality control, delivery and its scope in commercialization in India for the management of plant diseases. Afr. J. Agric. Res., 9: 3838-3852.
CrossRefDirect Link - Harman, G.E., 2006. Overview of mechanisms and uses of Trichoderma spp. Phytopathology, 96: 190-194.
CrossRefDirect Link - Shaban, G.M. and H.M.A. El-Komy, 2001. Survival and proliferation of alginate encapsulated Trichoderma spp. in Egyptian soil in comparison with allyl alcohol soil fumigation. Mycopathologia, 151: 139-146.
CrossRefDirect Link - Kucuk, C. and M. Kivanc, 2005. Effect of formulation on the viability of biocontrol agent, Trichoderma harzianum conidia. Afr. J. Biotechnol., 4: 483-486.
Direct Link - Attitalla, I.H. and B. Salleh, 2010. Improvement of carboxymethyl cellulase and xylanase production by alginate immobilized Trichoderma harzianum. Biotechnology, 9: 529-532.
CrossRefDirect Link - Mafia, R.G., A.C. Alfenas, L.A. Maffia, G.M. Ventura and E.A. Sanfuentes, 2003. Encapsulation of Trichoderma inhamatum for the biological control of Rhizoctonia solani in clonal propagation of Eucalyptus. Fitopatol. Bras., 28: 101-105.
CrossRefDirect Link - Lewis, J.A. and G.C. Papavizas, 1987. Application of Trichoderma and Gliocladium in alginate pellets for control of Rhizoctonia damping-off. Plant Pathol., 36: 438-446.
CrossRefDirect Link - Bae, Y.S. and G.R. Knudsen, 2007. Effect of sclerotial distribution pattern of Sclerotinia sclerotiorum on biocontrol efficacy of Trichoderma harzianum. Appl. Soil Ecol., 35: 21-24.
CrossRefDirect Link - Vinale, F., G. Ambrosio, K. Abadi, F. Scala and R. Marra et al., 2004. Application of Trichoderma harzianum (T22) Trichoderma atroviride (P1) as plant growth promoters and their compatibility with copper oxychloride. J. Zhejiang Univ., 30: 425-425.
Direct Link








