ABSTRACT
The wall flora (especially plants growing on the Walls of the walls of the historical buildings) of Sanliurfa has been presented in this study. It was undertaken with the aim to determine the taxa growing on the walls of historical building of Sanliurfa and make contribution to the flora of Sanliurfa. This study flora and vegetation of stony walls (wall tops, vertical wall surfaces) in South-easth Turkey. In total, 113 species of vascular plants were recordedreleves. Flora of walls is composed of a high number of accidental species. Only four species (Hyoscyamus niger, Hordeum spontaneum, Capparis ovata and Brassica alba) were frequently recorded on walls. Differences in species` features (life strategy, life form, dispersal) and ecological requirements of plants (light, moisture) were analysed between vertical wall surfaces and wall tops. Their structure, species composition, ecology and distribution are briefly discussed.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.153.162
URL: https://scialert.net/abstract/?doi=ajps.2006.153.162
INTRODUCTION
The region of Thrace has attracted the attention of many botanists rich flora produced as a result of its varied climatic and geographical features. The total number of Pteridophytes and Spermatophytes from the area until now[1]. Walls represent a specific environment, which is partly similar to rocks and rock fissures[2]. Nevertheless, their artificial origin, location in the urban and rural landscape and technology of wall building influence a range of plant species, which are able to colonize this habitat The following habitat attributes principally differentiate walls from the rocks: (i)Walls consist of building materials, which are usually piled up using various binding materials of lower durability and chemical composition different from the building material. Disintegration of the binding materials is responsible for accumulation of fine-grain rubble in crevices and thus provides substrate with variable content of nutrients that generally allow early succession of vegetation. The succession can be accelerated if soil itself is used as a binding or covering material. (ii) By being repeatedly cleaned and fettled, walls are in essence a temporary habitat. Frequent disturbances of wall vegetation thus contribute to a high variation in species composition and exclude many species typical of rocks and rock fissures. (iii)Walls are usually isolated objects of small dimensions. Therefore, wall microclimate is more strongly influenced by fluctuation of climatic factors (precipitation, temperature and irradiation) than in the case of rocks. (iv) Walls consist of limited number of microhabitats. Crevices resemble each other and sidewalls are of uniform slope and microtopography. In contrast, rocks are of apparent microhabitat diversity. (v) Since walls are situated within urban and rural landscape, composition of wall flora is strongly influenced by the mass-effect from the surrounding ruderal and seminatural vegetation types. Considering vertical division, walls usually consist of three different zones: (i) the base, (ii) the vertical wall surface with joints (fissures) and (iii) the wall top. Species composition of basal zone consists of plants growing on vertical surface and species of nearby vegetation. This is caused by favourable environmental conditions of the basal zone (more moisture and nutrients). The second zone is best developed on old walls of monuments and buildings in historical town centres, disintegrating castle fortification, shady walls in gardens, etc. Development of plant communities mostly depends on the level of disintegration of mortar, concrete or any other type of binding material. Many isolated walls were removed from the villages. Rebuilding of town walls increased during the last decade as a consequence of increased interest in conservation of historical monuments. This consequently stimulated my interest to collect data. The present study deals with the wall flora and vegetation of lower and middle altitudes of in south east Turkey Şanlıurfa).
According to the Flora of Turkey[3], some foreigner researchers; among them Davis[3] and Zohary[4], have visited and collected plant specimens from Şanliurfa In addition, according to Zohary, some other foreign researchers who have visited the region and gathered plant species from Şanliurfa, are Mc Neil, Handel-Mazetti, Haradjian, Haussknecht, Huber-Morath., Brown, Alava, Fenzl and Aucher. In the region, the only vegetational research known was the study focused on the Şanliurfa[4]. Much attention was paid to wall vegetation in Turkey[5-7].
MATERIALS AND METHODS
Sanliurfa is situated in the south-east of Turkey, 808 km away from Ankara. It has a surface area of 18.584 km sq. and a population of 1.303.589 (Fig. 1). This study is based on collection and wall’s field observations carried out in 2003 and 2004.
The altitude of the area changes between 350-1200 m. The climate is cold and rainy in winter, hot and dry in summer. The central part of South-east Turkey belongs to a warm region (mean annual average above 16°C) with a lower annual rainfall (below 600 mm). Higher elevations belong to a moderately warm region (mean annual average below 15°C) with a higher annual rainfall (above 600 mm)[8]. The economy of the province currently depends upon export demand. Its cultivable land is used mostly to grow cereals. Wheat is the main crop followed by barley and lentil. There is also chickpea farming and pistachio culture. Its industrial crops are cotton are seasame. Şanliurfa has characteristic Irano-Turanian flora with some Mediterranean elements also. The plant taxa on the walls of the historical buildings in Şanliurfa served as the material of this study. At total 245 plant specimens were collected during the years 2003 and 2004. The range of sampling stands included all types of walls (i.e. isolated walls in courtyard, fortification, city walls, walls of disintegrated buildings, monuments, etc.). The study was restricted to the wall tops and the wall joints, whereas the flora and vegetation of the wall bases (i.e. vertical surface up to 30 cm above ground) were not recorded. Localities were selected by occasional walking through rural landscape and by visits of towns with preserved historical centres, castles and chateaux. Data on flora and vegetation were collected only on walls fulfilling the following arbitrary rules: occurrence of at least two species of vascular plants and minimal cover of vegetation > 15% (wall top) or > 5%(vertical surface). Following these rules underestimated variability of species found on the walls, but it was necessary to avoid prohibitively time-consuming sampling, as well as problems with classification of early successional stages or of unique samplings. In total, 225 relevés had been collected (incl. those extracted from the published papers), but 113 relevés were excluded from the subsequent analyses due to their unique plant composition. The classical Zürich-Montpellier approach[9] and International Code of Phytosociologial Nomenclature[10] were used. Where it was not possible to classify stands using the Zürich-Montpellier approach, the deductive approach[11] was used (d.c. = derived community; b.c. = basal community).
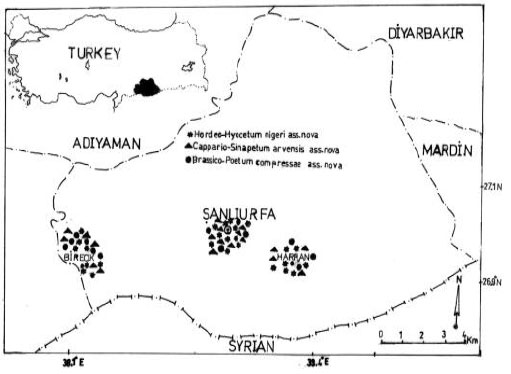 | |
| Fig. 1: | Map of the territory studied. Dashed curve specifies the territory, which was intensively studied. Distribution of the respective communities according to the releve/ date is given |
| Appendix 1: | Header data for Table 2 |
 | |
Nomenclature of taxa follows Davis. Life strategies were classified according to Grirne[12], life forms, dispersal strategies and origin status of vascular plants were taken from[13,14]. A key to locality numbers representing the wall flora of Şanliurfa is given Table 2. and Appendix 1 header date for Table 2. This study was Welch t-test according to Hill[15], (t: 4.1 df : 112 p < 0.001).
RESULTS AND DISCUSSION
Overview of flora: On walls, there is a high number of vascular plant species occurring with a very low frequency. Whereas 113 species were recorded in total, 180 of them (i.e. 87%) attained frequencies below 20% and 78 species (i.e. 38%) were found only in one relevé. The most common species-Taraxacum sect. Ruderalia and Poa compressa-occurred on 74 and 54% of studied walls, respectively. They were found on both vertical surfaces and wall tops. Totally, 36 families were recorded on studied walls; the most common were Poaceae Compositae (21.23% of total flora), Compositae (15.9%), Brassicaceae (13.27%), Solanaceae (6.19 %) and Lamiaceae (5.3%). The most frequent life strategies were C (30.4%), CSR (25.4%) and CR (18.8%); the most common life forms were hemicryptophytes (51%) and therophytes (26%). Native species comprised 77.2%, archaeophytes 13.0% and neophytes 9.8% of the total species number. Anemochory (40.0%) and autochory (18.2%) were the most common dispersal strategies of plants growing on walls. The flora on the walls consisted predominately of heliophilous to shade-tolerant plants, which indicate mesic to warm habitats and semi-dry to freshly moist soils. Reaction showed no clear pattern with more frequent species indicating neutral to basic soils. According to nitrogen requirements, the flora on walls indicated nutrient poor to moderately rich habitat (Table 1).
Nevertheless, high coefficient of variation indicates bimodal distribution of indicator values for nitrogen, i.e. high frequency of plants requiring either high or very low concentration of nutrients.
Comparison of wall tops and vertical surfaces: Considering that a lower number of relevés was recorded on vertical surfaces, the number of species recorded on wall tops did not exceed that on vertical surfaces (vascular plants:112 and 117 species, respectively). Nevertheless, species richness per relevé was significantly higher on wall tops (wall tops: 13.1±6.3; vertical surfaces: 8.8±4.6, Welch t-test, t = 4.1, df = 112, p < 0.001). Plants occurring on both vertical surfaces and wall tops represented only 32% of the total species numbers. The wall tops exhibited a lower number of families in comparison with the vertical surfaces (31 and 40 families, respectively). Species possessing CR, R, SR and S strategies were more common on wall tops, while those with C, CS and CSR strategies prevailed on vertical surfaces (Fig. 2). Therophytes were more common on wall tops, hemicryptophytes and phanerophytes on vertical surfaces. The two habitats did not differ in the proportions of alien and native species. Anemochorous, autochorous and endozoochorous plants were more frequent on vertical surfaces, while epizoochorous plants prevailed on wall tops (Fig. 2).
The wall tops did not differ from the vertical surfaces in mean indicator values for temperature, reaction and nitrogen (based on vascular plants data). Significant differences between wall tops and vertical surfaces were found for light and moisture (Table 1). Higher shade tolerance of species growing on vertical surfaces compared to those occurring on wall tops was recorded. Similarly, more species with high moisture indicator values were found on vertical surfaces (Table 1). Contrarily to the vascular plants, there were no differences between the wall tops and vertical surfaces in proportions of particular life strategies (Fig. 2).
Communities on wall tops and vertical surface Wall vegetation
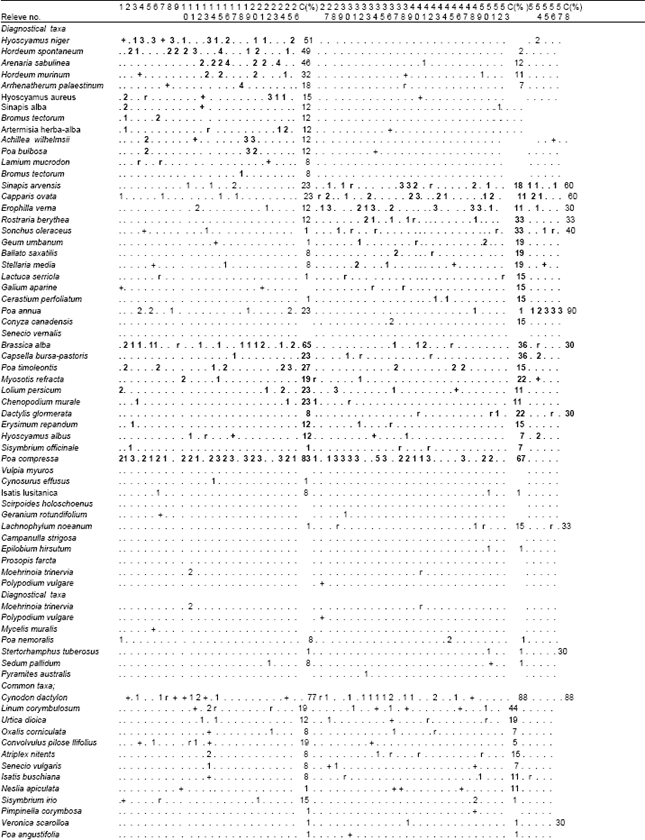
Hyoscyamus niger, and Hordeum spontaneum (Hordeo-Hyocetum nigeri ass.nova):This species-rich (mean 16.4, range 4-27) thermophilous type (Table 2, rels. 1-26) is differentiated by frequent occurrence of succulents and species of semi-dry habitats. Bromus tectorum is less dominant and other grasses frequently accompany it. The herb layer has a cover from 35 to 80% (mean 61%). The group of typical species includes annuals to short perennials, some perennial species (e.g. Hyoscyamus aureus, Artemisia herba-alba and Lamium mucronatum) and annual ruderal species. The community inhabits tops of disintegrating fortification of castles and bulwarks, plus tops of old isolated thicker walls in the villages (building material: clay slates, rarely granite and gneiss). The sites are strictly sun-exposed. Mostly shallow soils are of A-C type and contain a varying amount of skeleton and nutrients. The soil usually contains a high amount of carbonates since clay slate represents the prevailing building material. Broken relief, with zones of bare ground and crevices, characterizes the structure of the wall top. The habitats are only rarely mechanically disturbed by humans, but usually are heavily eroded. The community is distributed in the hilly parts of south-east Turkey Şanlıurfa) up to 600 m a.s.l. (Fig. 1).
Sinapis arvensis and Capparis ovata (Cappario-Sinapetum arvensis ass.nova).
| Table 1: | Mean indicator value (± 1 LDS) of selected ecological factors based on vascular plants[23] growing on studied walls. Differences between wall tops and vertical wall surfaces were tested using two sample t-test. CV : -coefficient of variation (%); ns. -non-significant |
 | |
| Table 2: | Wall vegetation in Şanliurfa; Hyoscyamus niger and Hordeum spontaneum (Hordeo-Hyocetum nigeri ass.nova) rels.1-26), Sinapis arvensis and Capparis ovata (Cappario-Sinapetum arvensis ass.nova) rels.1-58 , Poa compressa and Brasica alba (Brassico-poetum compressae ass.nova) rels. 27-58 |
 | |
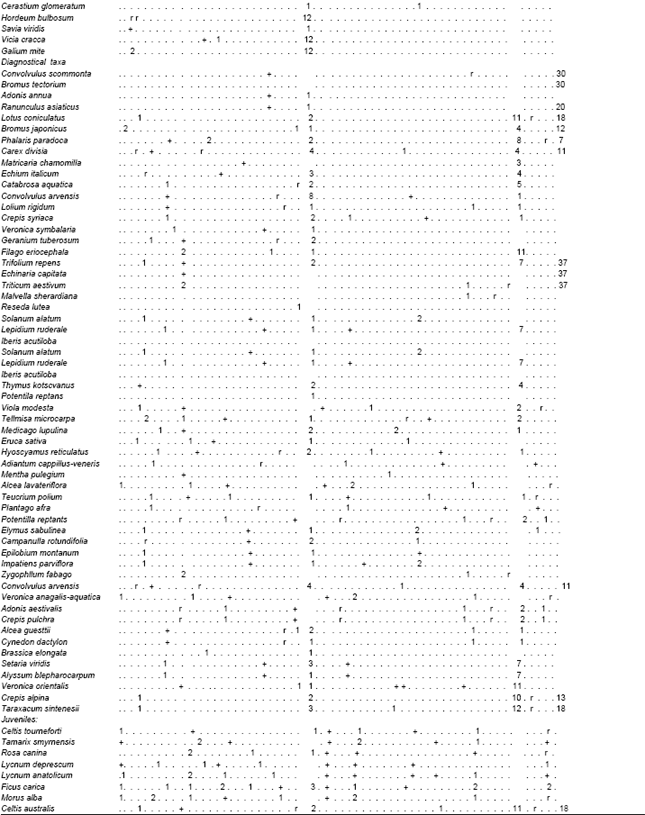 | |
| The number of example areas which are after from 10. area were showed below by below . For example 10. example area was showed like this 1 : 10 The bold characteristics which are tables show the plant association | |
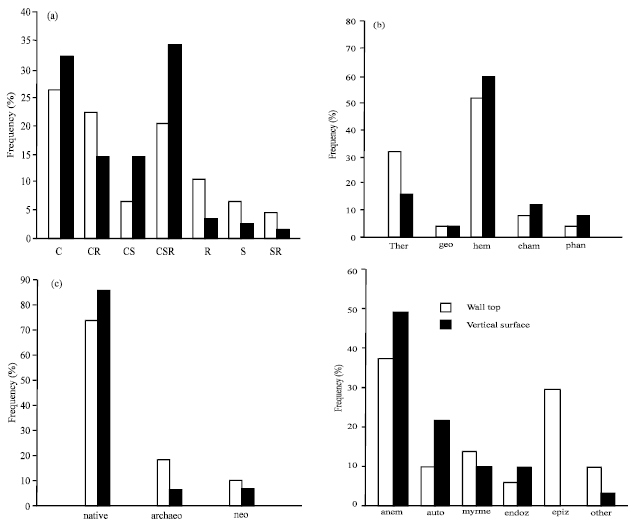 | |
| Fig. 2: | Frequency distribution of flora characteristics on wall tops and vertical wall surfaces. (a) Life strategies according to (22). χ2= 18.8, df = 6, p = 0.005; (b) Life forms: ther – therophytes, geo-geophytes, hem-hemicryptophytes, cham-chamaephytes, phan-phanerophytes. χ2 = 17.6, df = 4, p = 0.002; (c) Origin status: archaeo-archaeophytes, neo-neophytes χ2= 3.8, df = 2, p = 0.15; (d) Dispersal strategies of vascular plants: anem-anemochory, auto-autochory, myrme-myrmecochory, endoz-endozoochory, epiz-epizoochory, χ2 = 14.9, df = 6, p = 0.02 |
The community (Table 2, rels. 27-53) is characterized by dense stands of dominant Sinapis arvensis (cover of: 25–90%, mean 62%) with lower species richness (mean 13.3, range 7-25) growing on usually thick soil layer (ca. 5-15 cm) with dark brown A-horizon. Besides Erophilla verna subsp. verna, many stands are dominated by Capparis ovata ssp. Phalestina Erosion is less important due to dense sward on stabilized soil layer; as a result, there are low frequencies of R-strategists (i.e. weeds). On the other hand, stands are differentiated by the presence of C and CR-strategists (e.g., .Ballato saxatilis and Lactuca serriola) The moss layer with dominant Galium aparine has a lower cover with a mean of 29% (range 0-80%). The most typical stands of the community are the tops of wall fences in villages and old wall fences around cemeteries (building material: clay slates, rarely granite, bricks and schist), which were sometimes initially covered by soil layer. The soil contains lower amount of skeleton and higher amount of nutrients. The sites are either sun-exposed or shaded by surrounding trees. Widely distributed in the hilly part of Şanliurfa up to 550 m a.s.l. (Fig. 1).
Poa compressa and Brasica alba (Brassico-poetum compressae ass.nova): The species-poor community (mean 6.5, range 2-15) comprises stands dominated by Poa compressa (Table 2, rels. 1-58). Most of the diagnostic species of the communities mentioned above are lacking. It is typical of tops of isolated walls with thick and undisturbed soil layer. Poa compressa develops compact dense sward (cover of : 35-80%, mean 61%; cover of : 0-90%, mean 12%) that competitively displaces other species in the course of succession. Widely distributed in the hilly parts of Şanliurfa (Fig. 1).
Shrub vegetation dominated by Ficus carica var. carica was found only rarely on the tops of isolated walls (Table 2, rels. 1-58). Tamarix smyrnensis composes 1-3 m tall, dense stands with a high cover (mean 65%, range 40-100%) and low species-richness (mean 8.7, range 3-17). Due to strong shading from the lilac, herb and moss layers are only sporadically developed (cover of: mean 25%, range 1-70%; : mean 22%, range 0-40%) and consist of a number of accidental species. The community inhabits wall tops with very thick soils (ca. 10-30 cm) in later stages of soil accumulation. The stands originated by plantation of lilac or, less frequently, by its seeding or intergrowth from shrubs adjoining the walls. It was found only rarely on old walls in lower parts of Wall vegetation in Şanliurfa (Table 2).
Frequency analysis showed that the most common species onwalls could be classified into two groups: (i) plants specific to wall habitats or habitats with similar ecological conditions, i.e., rock debris swards and their semiruderal derivates (e.g., Poa compressa, Hyocyamus sp. div., Hordeum murinum) and rock fissures (e.g., Capparis ovata div., Brassica alba); (ii) common species with a wide ecological amplitude, which colonize the walls due to mass-effect from the surroundings (e.g., Taraxacum sintenesi). High number of accidental species that reach only low covers on walls indicate the influence of surrounding vegetation on species composition on the walls[3,16,17], as well as strong limitation by wall environment and/or competitive exclusion in a small-scale habitat. Although frequencies of respective dispersal strategies of plants occurring on wall tops do not differ from general pattern found in Central-European flora[18], vertical inclination apparently favours the establishment and spread of anemochorous, autochorous and endozoochorous species. Aside from historical and topographical aspects, vertical surfaces are more frequently repaired as against wall tops in the studied territory. Regular disturbances thus discriminate especially in favour of establishment of the anemochorous plants. Surprisingly, the proportion of myrmecochorous plants on vertical surfaces does not exceed that on wall tops. It is caused by the absence or very rare occurrence of many myrmecochorous species that are common in wall fissures in east-meditteranean parts of Europe (e.g., Capsella bursa-pastoris, Poa sp. div.) in the studied territory. In addition, most native myrmecochorous plants, which are dispersed by ants both on wall tops and vertical surfaces, successfully grow on both habitats. The highest proportion of species of Compositae family found on studied walls is related to its high species number in Central Europe and the remarkable success of this family in terms of dispersal and establishment[19]. The proportions of neophytes and archaeophytes found on the studied walls were similar to those found in the Czech settlements Duchoslav. There are several reasons for this difference: (i) Although the difference was non-significant, this study found archaeophytes and neophytes more frequently onwall tops than on vertical surfaces. Since most of the earlier research studied flora and vegetation on vertical surfaces only, they underestimated proportion of alien species. (ii) Proportion of alien species on walls is the largest in where wall vegetation has not such a special look as in other areas of area. The pattern of life forms recorded on the studied walls is the same as that found onwalls in other parts of Europe[20]. Higher proportion of therophytes on wall tops contrasted with a lower proportion on vertical surfaces. It is in agreement with higher proportion of R, CR and SR-strategists growing on wall tops, as opposed to the vertical surfaces. Especially winter-spring annuals form a considerable part of vegetation of wall tops Duchoslav, indicating a significant role of seasonal periodicity and disturbance. In contrast, a high frequency of hemicryptophytes and phanerophytes on vertical surfaces coincides with competition for limited space under relatively stabilized soil conditions. Such stands are typical of sites with uneven surface and thin A-C soils containing a higher amount of fine-grained soil and organic matter. Competition with vascular plants is not as strong in these stands as in stands dominated by Poa compressa later in succession. These stands exhibit both very low species richness and low cover of mosses. Indication of ecological conditions based on vascular plants clearly showed that wall tops and vertical surfaces differ in light and moisture:wall tops were drier and gained more incident radiation than vertical surfaces. In summary, ecological differentiation and contrasting topography of habitats are the factors which may explain the low number of species occurring both on wall tops and vertical surfaces. Moreover, physiological limits of early plant development exist: horizontal growth of radicle of young plants prevents many species from successful establishment and regular occurrence in horizontal fissures[21]. The phenomenon of wall tops is fully developed only on old isolated walls in the countryside. This is due to different construction and functionality of walls in villages and towns. Most of town walls lacked developed vegetation, owing to the use of concrete and compact and hard stones (e.g., granite) as a building material covered by protruding copings. Classified the top structure of isolated walls in South-east Anatolia into three types according to the prevailing ecological process: (i) erosion-accumulative, (ii) accumulative and (iii) erosion-type. Similar situation was recorded in the study area. The erosion-accumulative and erosion types have higher species richness with a large number of succulents and archaeophytes and lower cover of grasses. On the other hand, lower species richness and dominance by Poa sp. div. is typical of the accumulative type. It is evident that classification of vegetation on wall tops[22] directly reflects the environmental heterogeneity mentioned above. Nevertheless, floristic differences within and among the relevé material concerning wall top vegetation from former Turkey point to a high variability in species composition. Local stands are under influence of both different species pools and different meso- and macroclimate conditions of respective regions. Vegetation on wall tops consists of species which indicate many different syntaxonomic units of various ranks.
The shrub species with dominance of Ficus carica was frequently found on walls in south-east Turkey Şanlıurfa) but its low species richness and floristic heterogeneity does not allow its exact classification. Celtis teurnefortii is an indicator species of tops of old wall fences with very thick soil horizon. Stands to some extent similar to the mentioned type are reported from warm parts of the South-east Anatolia.
ACKNOWLEDGMENTS
I would like to thank the Research Fund of Harran Universty (HUBAK) for its financial support (Project No. 403), The comments and suggestion of my friend Dr. Esat Çetin and Research assistant Lokman Varişlı (Harran Universty) are also much appreciated.
Appendix 2: Lokalities of releves in Table 2
| 1: | The Urfa castle (on the walls); |
| 2: | The Ulu Mosque (on the walls); |
| 3: | The ˜izmalı Bridge (on the Walls) ; |
| 4: | The Eyübiye Mosque (on the garden walls); |
| 5: | The old Bath (on the dome); |
| 6: | The Fırfirlı Church ( on the wals); |
| 7: | The Fırfirlı Churce, ( on the wals); |
| 8: | Old Karakoyun Bridge (on the walls); |
| 9: | Şıh Mesut Mosque (old Mosque) (on the dome); |
| 10: | Yıldız Bath (on the dome); |
| 11: | Damat Süleyman Paşa Mosque (on the garden walls); |
| 12: | Vertical surface of the Urfa Castle; |
| 13: | Vertical surface of the Hasan Paşa Mosque; |
| 14: | Vertical surface of the Germüş Churce; |
| 15: | Vertical surface of the Rızvaniye Moslem theological School; |
| 16: | Bediüzzaman Graveyard (on the garden walls); |
| 17: | Prophet İbrahim tomb (on the dome); |
| 18: | Germüş Churce (on the walls); |
| 19: | Old Dergah Mosque (on the walls); |
| 20: | Germüş Churce (on the walls); |
| 21: | Old Haşimiye Bazaar (on the walls); |
| 22: | Şehbenderiye fountain (on the walls); |
| 23: | Hekimdede fountain (on the walls); |
| 24: | Emencelizade fountain (on the walls); |
| 25: | The Millet Bridge (on the walls); |
| 26: | Birecik Urfakapısı City Walls (on the walls); |
| 27: | The Birecik Castle (on the walls); |
| 28: | Birecik Meçan Kapısı City Walls (on the walls); |
| 29: | Birecik Ulu Mosque (on the walls); |
| 30: | Birecik City Walls (on the walls); |
| 31: | Birecik Long Bazaar (on the walls); |
| 32: | Birecik Old Culture house (on the walls); |
| 33: | Birecik Atar Bazaar (on the walls); |
| 34: | Birecik Çulcu Bazaar (on the walls); |
| 35: | Birecik Turşu Şığ Müslüm house (on the walls); |
| 36: | Vertical surface of the Harran Castle. fortification; |
| 37: | Birecik Belediye ınn (on the walls); |
| 38: | The Harran Castle (on the walls); |
| 39: | Harran City Walls (on the walls); |
| 40: | Vertical surface of the Harran Castle. fortification; |
| 41: | Harran Old Houses (on the walls); |
| 42: | Vertical surface of the mam Bakır Tomb fortification; |
| 43: | İmam Bakır Mosque (on the walls); |
| 44: | Harran Halep KapısıWalls (on the walls); |
| 45: | Harran Rakka KapısıWalls (on the walls); |
| 46: | Harran Ulu Mosque (on the walls); |
| 47: | Harran Musul Kapısı(on the walls); |
| 48: | Harran Rakka Kapısı(on the walls); |
| 49: | The Urfa castle (on the walls); |
| 50: | Şıh Mesut Mosque (on the Walls); |
| 51: | Prophet Ibrahim tomb (on the dome); |
| 52: | Beykapısı City Walls; |
| 53: | The Birecik Castle (on the walls); |
| 54: | Vertical surface of the Birecik City Wall; |
| 55: | Harran vertical surface of the castle fortification ; |
| 56: | Şanliurfa, Vertical surface of BalıklıGöl Walls; |
| 57: | Vertical surface of the Harran City Wall; |
| 58: | Vertical surface of the Harran Castle. |
REFERENCES
- Borgegard, S.O., 1990. Vegetation development in abandoned gravel pits: Effects of surrounding vegetation substrate and regionality. J. Veg. Sci., 1: 675-682.
CrossRef








