Research Article
Glutamine Synthetase and Glutamate Dehydrogenase in Broccoli: Changes in Activities During Postharvest Senescence of Two Cultivars
Not Available
Toshiyuki Matsui
Not Available
Haruo Suzuki
Not Available
Yusuke Kosugi
Not Available
The edible portion of broccoli (Brassica oleracea L.) which is collectively called as head is harvested at its immature stage. Inevitably, after it has been detached from the mother plant, the head will actively undergo physiological processes for its continued growth. The supply disruption of necessary metabolites for the normal physiological processes and exposure to ambient temperature at harvest impose stress and consequently, senescence or quality deterioration. In addition to these stress-inducing factors, genetic makeup of the crop also controls senescence. King and Morris[1] reported that differences in storage quality such as the tendency to become yellow, pattern of ethylene production, respiratory activity and the decline in organic acids[2] have been observed between cultivars. This implies that genetic factors are involved in the process. Moreover, the temperature to which the product is exposed has also been found to affect deterioration of fresh products. Some studies showed that broccoli heads can only stand for 2-3 days when stored at 20°C[3] and 3-4 weeks if stored at 0°C[4]. Symptoms of deterioration include wilting, chlorosis in the floret sepals and in severe cases, browning and dehiscence of florets and abscission of off-flavors.
Quality degradation in perishable products such as broccoli[1] and asparagus[5] are accompanied by accumulation of ammonia and changes in organic compounds like amino acid. Considering the vital physiological implication of these compounds on the shelf life of perishable products, activity of enzymes responsible for their biosynthesis is helpful in the understanding of the deteriorative process. The important nitrogen-assimilating enzymes involved in this process include glutamine synthetase (GS, EC 6.3.1.2) and glutamate dehydrogenase (GDH, EC 1.4.1.2). It has been reported that GS catalyzes the first step in amino acid biosynthesis from ammonia[6]. It brings nitrogen into metabolism by condensing ammonia with glutamate, with the aid of ATP, to yield glutamine. The product, glutamine, is a source of nitrogen in the biosynthesis of many metabolites, including amino acids, nucleotides and amino sugars[7]. In cases where supply of ammonia is excessive, plant tissues undergo appropriate modification in their metabolism to prevent ammonia concentration from rising to a toxic level[8-10]. In this situation, GDH is induced while GS is repressed[4]. GDH is also responsible for the link between carbohydrate and amino acid metabolism[12] or it could further act in the deamination of glutamate connecting it to carbon metabolism rather than nitrogen metabolism[13].
An in-depth understanding of the deteriorative process of broccoli is required to improve its quality and shelf life. Hence, this study was conducted to describe the biochemical characteristics and the physiological significance of nitrogen-assimilating enzymes occurring in harvested broccoli. Specifically, we investigated the activities of GS and GDH enzymes and accumulations of ammonia and total amino acid in the floret and branchlet portions of two broccoli cultivars during postharvest senescence. Some physiological changes which have been reported to influence shelf life of broccoli are also discussed.
Plant material: Two broccoli cultivars (‘Hartland’ and ‘Sairin’) grown in winter were harvested from a farmer’s commercial field in Miki, Kagawa, Japan. Right after harvest, the heads were packed in a box with crushed ice and transported to the laboratory. Each head was trimmed, enclosed with a perforated plastic sheet and held at 20°C. At the end of each storage period, the florets were separated from the branchlets and immediately kept at -30°C until analysis.
Weight loss: Weight loss was determined by weighing the heads daily until the end of the five-day storage period. The decrease in weight was expressed as percentage of the sample weights as determined on a daily basis.
Color change/hue angle determination: Color change in three different portions of the florets were measured using a chromometer (Nippon Denshoku, Kogyo Co., Ltd.), equipped with an 8 mm measuring head and a C illuminant. The meter was calibrated using the manufacturer’s standard white plate. Color change was quantified in L, a and b color spaces. L refers to the lightness of the head and ranges from black=0 to white=100. Positive and negative values of a indicate red-purple and green colors, respectively, while the positive and negative values of b indicate yellow and blue colors, respectively[14]. Hue angle, h°=tan-1(b/a) when a>0 and b>0 or ho=180°+tan-1(b/a) when a<0 and b>0 was calculated from the a and b values[15]. The result was expressed as hue angle.
Respiration rate measurement: Each head was weighed and placed in a 6 L glass jar held at 20°C. Carbon dioxide production was measured daily from an intact head sealed in a glass jar for 1 h by taking 10 ml gas sample from the glass jar and injecting the sample to a TCD gas chromatograph equipped with a 1 m activated charcoal column at 70°C (GC-8 AIT, Shimadzu Co., Ltd.). The result was expressed as ml CO2 kg-1h-1.
Enzyme extraction and assay: Five grams fresh-weight sample from each portion of the broccoli head was added with 1% polyvinyl polypyrrolidone (PVPP) proportional to the sample weight, 1 g sea sand and 5 ml buffer solution. The mixture was homogenized using mortar and pestle. Extraction was performed as described by Hurst and Clark[16] in which the buffer contained 50 mM tris-HCl (pH 7.6), 10 mM MgSO4, 1 mM EDTA, 1 mM dithiothreitol (DDT), 12 mM 2-mercaptoethanol, 5 mM L-glutamate and 100 ml glycerol L-1. The homogenate was squeezed through four layers of cotton cloth. The residual tissues were re-extracted with an additional 5 ml of the same buffer and the filtrate was centrifuged at 11,000 rpm at 2°C for 10 min.
Enzyme activities were assayed in a total volume of 1 ml assay mixture. For determination of GS activity, 80 mM Na L-glutamate, 100 mM tricine-KOH buffer (pH 7.0), 6 mM NH2OH, 20 mM MgSO4.7H2O, 1 mM diethylenetriamine pentaacetic acid (DTPA), 8 mM ATP and 8 mM mercaptoethanol were used. After incubating at 35°C for 8 min, the reaction was stopped by adding 1 ml ferric reagent containing 0.37 M FeCl3, 0.67 M HCl and 0.2 M trichloroacetic acid (TCAA). Absorbance of the sample was recorded at 540 nm using a double beam spectrophotometer following the method of Lowry et al.[17] using bovine serum albumin as the standard. GDH activity was determined in both aminating and deaminating directions in a total volume of 1 ml assay mixture. For GDH-amination activity determination, a mixture of 20 mM α-ketoglutaric acid, 100 mM tris-HCl (pH 8), 200 mM NH4Cl, 1 mM CaCl2, 0.20 mM NAD(P)H, 20 μl distilled water and 200 μl enzyme solution was used. The GDH-deamination activity was assayed with a mixture containing 100 mM L-glutamate, 100 mM tris-HCl (pH 9.3), 1 mM NAD(P)+, 0.5 mM CaCl2, 240 μl distilled water and 200 μl enzyme solution. Both amination and deamination activities were monitored using a double beam spectrophotometer (Shimadzu model UV-150-02) at 350 nm to NADH oxidation or NAD+ reduction. One unit of GDH activity is defined as the oxidation or reduction of 1 μmol of coenzyme (NADPH/NADP, respectively) per min at 30°C.
Ammonia assay: Two grams of fresh-weight sample from each portion of the broccoli head was extracted with 10% TCAA at 0°C (ice bath) and centrifuged at 11,000 rpm at 2°C for 10 min. The ratio of tissue weight to TCAA solution was 1:10. Ammonia content was assayed using the procedure of Kun and Kearney[18] which required a 1 ml assay mixture containing 200 μl 0.5 M tris-buffer (pH 8), 100 μl 0.1 M 2-oxoglutarate solution (pH 7.4), 30 μl 8 mM β-NADH solution, 150 μl distilled water and 500 μl of neutral extract sample. The decrease of NADH, as determined by the change of extinction at 365 nm, was used as a measure of the reaction.
Extraction and assay of total amino acid: Two grams of fresh-weight sample from each portion was homogenized with 15 ml of 75% ethanol under ice-cold (0°C) condition and incubated in a water bath shaker at 45°C for 30 min. The solution was filtered into a flask and the residue was again added with 15 ml of 75% ethanol and placed in the water bath for another 30 min. The homogenate was centrifuged at 3,000 rpm for 10 min to separate the tissues from the solution. The supernatant was filtered through a funnel lined with filter paper (Toyo, No. 2) and evaporated under reduced pressure. The residue from the flask was dissolved in 10 ml deionized water and centrifuged at 11,000 rpm, at 2°C for 10 min.
Total amino acid was determined with a mixture containing 0.5 ml citrate buffer (0.2 M citric acid monohydrate adjusted to pH 5 with 1 N NaOH), 1.2 ml ninhydrin solution (containing 0.25 g ninhydrin dissolved in 0.5 ml 0.01 M KCN and 30 ml ethylene glycol monoethyl ether) and 1 ml extract (50x dilution). The mixture was incubated in boiling water for 15 min and cooled under running water for five min. Five ml 60% ethanol was added to the mixture before the measurement of sample absorbance at 570 nm.
Statistics: A randomized complete block design was adopted with three replications. The level of significance was calculated from the F-value of ANOVA. Linear correlation was used to evaluate the relationship between enzyme activities and ammonia and total amino acid accumulations.
Weight loss: The weight of the broccoli heads of both cultivars stored at 20°C was found to rapidly decline by about 5% of the initial weight during the first day of storage and a further gradual decrease in weight was observed as storage progressed (Fig. 1). Cumulatively, about 17% of the weight in both cultivars was lost after five days of storage. Weight loss as manifested by wilting of florets was the first sign of senescence in broccoli heads.
Color change/hue angle: Figure 2 shows the change in color of the two broccoli cultivars during storage at 20°C. The hue angle decreased with time in both cultivars. However, ‘Sairin’ showed a lower hue angle value than ‘Hartland’, which indicates its purple pigmentation. The decline in hue angle during storage was exhibited by a corresponding decrease in green color or yellowing of florets. All heads were substantially yellow at the end of the storage period. The degreening of florets was the most striking symptom of postharvest senescence in broccoli.
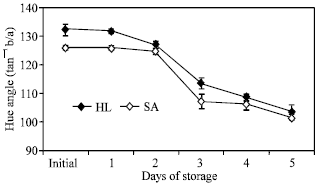
Respiration rate: The respiration rate abruptly decline on the first 24 h and 48 h of storage in ‘Sairin’ and ‘Hartland’, respectively and slightly increase until the end of the experimental period (Fig. 3). ‘Sairin’ exhibited higher CO2 production rate than ‘Hartland’.
Enzyme activities: A remarkable difference in enzyme activities between the two portions of both cultivars was observed with florets having a significantly higher initial GS activity (Fig. 4). Enzyme activity in the floret portion continuously decreased while in the branchlets, a transient increase was observed on the first day of storage. After the first day, no apparent change was observed in enzyme activity in the branchlets of both cultivars until the end of the storage period.
GDH-aminating and deaminating activities in the floret portion of ‘Hartland’ gradually increased until the fourth day and third day of storage, respectively (Fig. 5). However, GDH activity in the florets of ‘Sairin’ was not detected.
| Table 1: | Correlation coefficient (r) values computed from linear regression analyses between enzyme activities and ammonia and total amino acid accumulations in the floret and branchlet portions of two broccoli cultivars held at 20°C for 5 days |
 | |
 | |
| Fig. 1: | Percent daily weight loss of two broccoli cultivars (‘Hartland’ and ‘Sairin’) held at 20°C for 5 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. Legend: HL = ‘Hartland’; SA = ‘Sairin’ |
 | |
| Fig. 2: | Hue angle change of two broccoli cultivars (‘Hartland’ and ‘Sairin’) held at 20°C for 5 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. Legend as shown in Fig. 1 |
In the branchlet portion, GDH-aminating activity in ‘Hartland’ dropped on the first day of storage but remained at a practically constant level until the fourth day while a further decrease until the third day was observed in ‘Sairin’.
 | |
| Fig. 3: | Respiration rate of intact heads of two broccoli cultivars (‘Hartland’ and ‘Sairin’) held at 20°C for 5 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. Legend as shown in Fig. 1 |
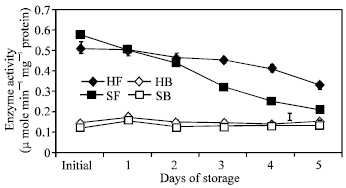 | |
| Fig. 4: | Changes in the activities of glutamine synthetase in the floret and branchlet portions of two broccoli cultivars (‘Hartland’ and ‘Sairin’) held at 20°C for 5 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. Legend: HF = ‘Hartland’ florets; HB = ‘Hartland’ branchlets; SF = ‘Sairin’ florets and SB = ‘Sairin’ branchlets |
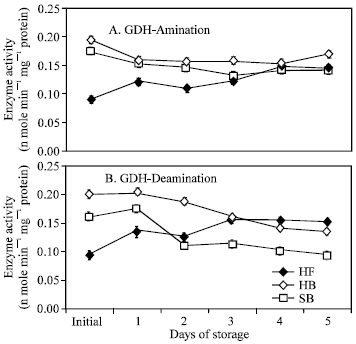 | |
| Fig. 5: | Changes in the activities of GDH-amination (A) and deamination (B) in the floret and branchlet portions of ‘Hartland’ and ‘Sairin’ held at 20°C for 5 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. Legend as shown in Fig. 4 |
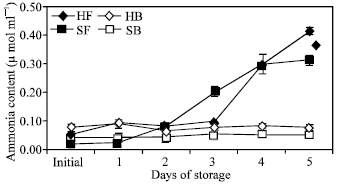 | |
| Fig. 6: | Ammonia content in the floret and branchlet portions of two broccoli cultivars (‘Hartland’ and ‘Sairin’) held at 20°C for 5 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. Legend as shown in Fig. 4 |
GDH-deaminating activity decreased continuously as storage progressed in both cultivars (Fig. 5). GDH activity was quantitatively higher in the branchlet than in the floret portion.
Ammonia accumulation: A large increase in ammonia content in the floret portion of both cultivars was found after the five-day storage period.
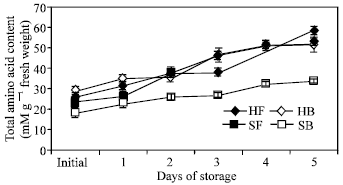 | |
| Fig. 7: | Total amino acid content in the floret and branchlet portions of two broccoli cultivars (‘Hartland’ and ‘Sairin’) held at 20°C for 5 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. Legend as shown in Fig. 4 |
It accounted for about 87 and 93% of the initial content in ‘Hartland’ and ‘Sairin’, respectively (Fig. 6). ‘Sairin’ accumulated ammonia in the florets two days earlier than ‘Hartland’. In the branchlet portion, the concentration of ammonia in ‘Hartland’ did not remarkably change except for a transient increase on the first day of storage. A slight increase was observed at the end of the storage period only in ‘Sairin’ cultivar.
Total amino acid accumulation: Total amino acid in the floret and branchlet portions of both cultivars increased significantly after the five-day storage period (Fig. 7). The floret portion of both cultivars showed higher amount of amino acid accumulated, which accounted for about 100 and 150% of the initial content in ‘Hartland’ and ‘Sairin’, respectively. In the branchlet portion, about 80% increase was observed in both cultivars.
Correlation between GS/GDH enzyme activity and ammonia and total amino acid accumulations: Table 1 shows the correlation coefficient (r) values between GS/GDH activities and ammonia and total amino acid accumulations in the floret and branchlet portions of the two broccoli cultivars. There was a highly significant negative correlation observed between GS activity and ammonia (r=-0.780) and total amino acid (r=-0.837) accumulations. GDH-amination activity in the floret portion of ‘Hartland’ showed a highly significant positive relationship with ammonia (r=0.715) and total amino acid (r=0.738) accumulations. GDH-deamination activity and total amino acid accumulation in the floret and branchlet portions of ‘Hartland’ showed significant positive (r=0.514) and highly significant negative (r=-0.716) correlations, respectively. In ‘Sairin’, GS activity in the florets showed a highly significant negative correlations with ammonia (r=-0.955) and total amino acid (r=-0.928) accumulations while in the branchlets only GDH-deamination activity showed a significant negative correlation with ammonia content (r=-0.582) and highly significant negative relationship with total amino acid accumulations (r=-0.813).
Exposure of broccoli heads to ambient temperature after harvest imposes significant physiological and biochemical changes that resulted to rapid senescence of the commodity. One of the earliest prominent signs of quality deterioration was the abrupt loss of weight during the first day of storage (Fig. 1). This was exhibited by the wilting of the florets and loss of turgidity in the branchlets, which may have been triggered by high water vapor pressure deficit associated with high temperature storage. Water stress could signal further deteriorative process in the tissues, which could subsequently lead to the death of cells. Following dehydration, decrease in greenness of florets was observed in both cultivars with “Sairin’ having lower hue angle value (Fig. 2). The loss of green color which could be regulated by ethylene[19,20] progressed with time, though it was not visually evident until the third day of storage. The lower hue angle value observed in ‘Sairin’ could be attributed to its purple pigmentation. Another important physiological change noted was the abrupt decline in respiration rate during the first 24 and 48 h of storage of ‘Sairin’ and ‘Hartland’, respectively and a subsequent increase thereafter (Fig. 3). This observation was confirmed in broccoli stored at 20°C[1] or at 16°C[32] and in asparagus spears stored at 325°C[5] after harvest which showed a declined in CO2 production during the first 24 and 48 h of storage, respectively. The higher initial respiration rate in both cultivars could be a response to stress imposed by harvest and trimming. Moreover, both cultivars exhibited a parallel respiration patterns except that ‘Sairin’ showed higher rate than ‘Hartland’. This difference in CO2 production could be one of the contributing factors to the shorter shelf life of ‘Sairin’. A higher respiration rate would mean rapid breakdown of respiratory substrate resulting in early substrate depletion accompanied by stress and eventual death of tissues.
Changes in the activities of GS and GDH were remarkable during postharvest senescence of broccoli. GS activity decreased in the florets while in the branchlets, a transient increase was observed only during the first 24 h of storage and no apparent change occurred until the end of the storage period of both cultivars. The decrease in GS activity in the florets could be a consequence of authophagic processes[21] induced by carbohydrate deprivation and/or ammonia accumulation and end product control[22]. It has been reported that towards the end of the postharvest life of broccoli, glucose and fructose levels decline and declines in amino acid and protein levels are accompanied by a large increase in ammonia content[23]. Deprivation of carbon source, therefore, could possibly have a negative effect on GS activity. This is further supported by observations that the embryonic axis of lupine seedlings deprived of reserves of metabolites[24] and commodities that are typically harvested immature at a time prior to cessation of physiological growth[25] are unable to maintain metabolic homeostasis. Hence, the decreased GS activity could be partly associated to the deprivation of the broccoli head of continued supply of necessary metabolites after it was detached from the mother plant. Furthermore, high concentration of ammonia was reported to repress GS activity[11]. At the end of the storage period, ammonia content in the florets increased to about 87 and 93% of the initial content in ‘Hartland’ and ‘Sairin’, respectively (Fig. 6). One typical characteristic of senescing tissues is the production of ammonia as a consequence of breakdown of chlorophyll, essential photosynthetic proteins and amino acids[23]. Thus, ammonia accumulation in broccoli could be the result of chlorophyll degradation, as indicated by the decrease in hue angle value (Fig. 2) and protein breakdown as indicated by the increase in total amino acid (Fig. 7). The higher initial GS activity in the floret portion could be attributed to the developmental state of the organ as they are comprised of immature and rapidly developing tissues. In asparagus, the same trend was observed in enzyme activity[5] which is expected to be higher in the tip of the spear since this is the region of the highest protein content and cell division[26,27].
In contrast, GDH showed increasing activity particularly in the floret portion of ‘Hartland’. Both aminating and deaminating activities gradually increased until the fourth day and third day of storage, respectively, implying a dual role of GDH during senescence. It has been reported that ammonia produced during proteolysis could be responsible for the observed increase in the aminating activity of GDH to detoxify the cell while the considerable increase in deaminating activity suggests that the enzyme also operates in the direction of energy generation[8,28] by supplying the cell with carbon skeletons[12]. A major notable difference observed between the two cultivars was that GDH activity was not detected in the florets of ‘Sairin’. Johansson and Gest[29] reported that a purple photosynthetic bacterium, R. capsulate, lacks GDH and normally uses the GS/GOGAT sequence of reactions for the assimilation of ammonia. Another probable explanation could be that the level of GDH was below the detection limit of the assay procedure adopted in this study. A separate in-depth experiment would provide more conclusive evidence for these suggestions.
In the branchlet portion, a significantly higher initial GDH-aminating and deaminating activities were observed in both cultivars but were not maintained throughout the experimental period. GDH-aminating activity in ‘Hartland’ dropped on the first day of storage but remained practically constant until the fourth day of storage whereas a further decrease was observed in ‘Sairin’ until the third day of storage. However, GDH-deaminating activity continuously decreased until the end of the storage period in both cultivars. The higher initial enzyme activity in this portion could be attributed to the age and tissue composition of the organ. In Zea roots, the enzyme is much more active in mature regions than in apical regions[30]. Branchlets are composed of more mature and harder tissues which may be resistant to degradation within a limited storage period and thus no substantial amount of ammonia was produced from proteolysis to trigger its activity. Ammonia was found to accumulate mainly in the floret rather than in the branchlet portion (Fig. 6). This result is in agreement with the report of King and Morris[1] which indicated that ammonia largely accumulated in the floral section during late senescence of broccoli. The trend of GDH activity in the branchlet portion further suggests that no considerable amount of ammonia was accumulated in this portion at the earlier stage of storage. Ammonia content in the branchlet portion of ‘Hartland’ was almost unchanged relative to its initial concentration except for a slight transient increase during the first 24 h of storage while only a slight increase was observed in the branchlet portion of ‘Sairin’ at the end of the storage period (Fig. 6). It is likely that GS in the branchlets could maintain its activity since the concentration of ammonia might not have reached repressive levels.
The inverse relationship of changes in the activities of GS and GDH in the floret portion of ‘Hartland’ could be attributed to ammonia accumulation which possibly regulates the activities of these enzymes. A highly significant negative correlation was observed between GS activity and ammonia and total amino acid accumulations in the floret portion of both cultivars while a highly significant positive relationship was evident between GDH-aminating activity and ammonia and total amino acid contents in the florets of ‘Hartland’. The complementing functions of these two nitrogen-assimilating enzymes do not only provide the necessary glutamate for biosynthesis but may also prevent excessive accumulation of free ammonia in the cell[22] thereby detoxifying the cell from ammonia by means of incorporation into amino acid[31].
This study has demonstrated that both cultivars show parallel patterns in weight loss, color change and respiration rate. However, ‘Sairin’ exhibited higher total weight loss and rate of respiration and lower hue angle value. The varying levels of enzyme activities in different portions of both cultivars could be attributed to diversity of tissues present in the excised head and may be due to varietal traits. The inverse relationship between the changes in the activities of GS and GDH might be influenced by ammonia accumulation in the tissues due to breakdown of chlorophyll and proteins. The shorter shelf life of ‘Sairin’ could be attributed to higher respiration rate which may have resulted in early depletion of respiratory substrate and the deficiency of GDH activity in the florets might have led to ammonia toxicity. Further studies at the molecular level are suggested for an in-depth understanding of the deteriorative process controlling changes in gene expression during postharvest senescence.
The authors would like to thank the Government of Japan through its Ministry of Education, Science and Technology for the financial support.