Review Article
A Review of the Thermodynamics and Kinetics of Oxyfuel Gas Cutting of Steel
Department of Materials Science and Engineering,Obafemi Awolowo University, Ile-Ife, Nigeria
Oxyfuel gas cutting is a relevant and widespread industrial process that uses fuel gases and oxygen to cut metals (Adedayo and Oyetoyan, 2010; Adedayo, 2009a, b). In a considerable proportion of the published work on the subject of oxygen cutting, the details of the oxidation reaction are overlooked or confused. For example, it is not uncommon for the oxidized material to be attributed with the physical characteristics of iron rather than iron oxide. The iron oxide produced as a result of oxidation by oxyfuel gas cutting is not clear-cut elucidated. De Garmo et al. (1999) believes Fe3O4 is formed. Masachuesset Institute of Technology (2002) identifies FeO, Fe2O3 and Fe3O4 as potential oxides that could be produced. Higgins (1993) reported FeO as the only oxide formed. The analysis of the oxidation reactions pertinent to gas cutting of steels has also been majorly ignored. The focus of most literature has been on the Fe content of the ferrous alloy, while the other contents are ignored. The oxidation process of iron and steel though similar in some respects, yet, in other aspects, show significant differences. Carbon has been known to have significant effects on properties and microstructures of ferrous alloys (Adedayo, 2010). A practical example of this is found during the construction of the steel skeleton for a flour mill in Nigeria in 2006. It was planned that the period of execution of the job would be 6 months, based on the understanding that mild steel (0.16 w% C) would be used for the steel skeleton. The cost of the project should be N15.23 million. However, the client purchased medium carbon steel (0.33 wt. % C). The result was that the rate of job execution was much slower than expected. The job would require about 14 months. Also more volumes of oxyfuel gases are required. The cost for labour, accommodation, feeding, transport and rented equipments had doubled. Generally, cost became very high and over N13 million have to be added to initial N15.23 million. Definitely, there was problem. The client believed this was a gimmick by the contractor to siphon fund. The client believed 0.33 wt % C steel should cut faster since it has a lower melting point.
In this review study, elucidation is made on the product of oxidation of steel at high temperatures. A description of the mechanism of interactions of the different phases involved in the reactions of slag forming process when steel is cut is provided. The roles of carbon and the activity of oxyfuel gases in this process are elucidated. Theoretical models of the thermodynamics and kinetics of cutting process pertinent to steels are also discussed. Similar theoretical models have been applied in laser-oxygen assisted cutting.
BACKGROUND AND FUNDAMENTALS OF OXYFUEL GAS CUTTING
French engineers Edmond Fouche and Charles Picard were the first to develop an oxygen-acetylene torch in 1903 (Wikipedia, 2010; Carlisle, 2004). Oxy-fuel is one of the oldest welding processes, though in recent years it has become less popular in the developed worlds due to the laser oxygen cutting innovation which is one of the mainstays of modern industry profiling of mild or carbon steel. However, it is still widely used for welding/cutting pipes and tubes, as well as repair work. It is also frequently well-suited and favored, for fabricating some types of metal-based artwork. In the developing worlds where incesant power outage is a common phenomenon, oxyfuel gas cutting has made arc-welding/cutting unlucrative. The low cost of oxyfuel gas cutting equipment is one of the main reasons for its use.
Oxy-fuel processes may use a variety of fuel gases, the most common being acetylene. Other gases that may be used are propylene, Liquified Petroleum Gas (LPG), propane, natural gas, hydrogen and MAPP gas. Many brands use different kinds of gases in their mixes. Sometimes a metal-cutting torch is colloquially called a gas-axe, smoke wrench, hot wrench, blue wrench or hot blue spanner (in Britain). Colloquially, many people mistakenly call a welding torch a blowtorch (Wikipedia, 2010). In the USA the word blowtorch is also used for what in Britain is called (uncommonly) a blowlamp.
By far the most common use for the oxy-fuel torch is in the cutting of steel and iron based alloys. Cutting is done with a modified torch. This torch is similar to a general purpose torch but adds several features. The first is a lever which allows the user to release a continuous jet of pure oxygen out of a set of nozzles at the tip of the torch. These nozzles are usually in the center of the tip and are separate from the nozzles used to generate the torch flame. The second addition to the cutting torch is a knob for adjusting oxygen flow through these nozzles. The basic principles of the process lie in rapid high-temperature oxidation of the cut metal. In gas cutting, the cutting torch is used to heat metal to kindling temperature when a stream of oxygen then trained on the metal combines with the metal which then flows out of the cut (kerf) as an oxide slag, The cut quality depends on the torch tip size, type and distance from the metal (AWS, 1997), the oxygen and preheat gas flow rates and the cutting speed. Also, the melting point of the cut metal relative to that of its oxide also plays an important role indetermining the cut quality and the cutting rate. For example, iron (melting point of 1536°C) can be flame cut while aluminum (melting point of 660°C) and magnesium (melting point of 650°C) can not (Masachuesset Institute of Technology, 2002). This is because melting point of iron oxide (1380°C) is lower compared to that of the metal. Aluminum oxide and magnesium oxide have melting points of 2000°C and 2800°C, respectively. However, titanium (Ti melting point = 1668°C and TiO2 melting point = 1750°C) gas cutting is possible.
METAL OXIDATION PROCESS
Generally, during hot rolling process, an oxide scale layer always develops at the metal surface. Oxidation of metal is not simply chemical combination of metal and oxygen.
| (1) |
But consists of two partial processes
| (2) |
| (3) |
| (4) |
Or
| (5) |
There has been considerable interest in the high temperature oxidation of metals-textbooks on this subject include Birks and Meier (1983) and Kofstad (1988). Oxidation of steel at temperatures higher than 843 K leads to three different iron oxide layers: wustite (FeO), magnetite (Fe3O4) and hematite (Fe2O3), in oxygen content increasing order, going from substrate to free surface as shown in Fig. 1.
Below 843 K, only the last two are thermodynamically stable (Suarez et al., 2006). According to the literature, the relative thickness fractions of these layers between 973 and 1473 K are 95% wustite, 4% magnetite and 1% hematite at equilibrium (Suarez et al., 2006; Poirier et al., 2006) although, this balance can vary from one case to another. Depending on temperature, time, atmosphere conditions and steel chemistry, energy barriers develop, which must be overcome for an oxide to grow (Suarez et al., 2006).
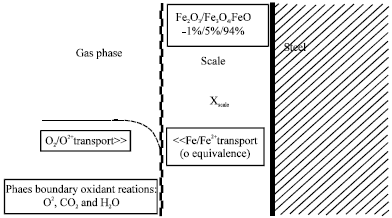 | |
| Fig. 1: | Mechanism of oxidation of steel (Poirier et al., 2006) |
 | |
| Fig. 2: | Polished cross sections of sparks given off during the cutting process (Powell et al., 2009; Ivarson et al., 1992). The light part of each section is iron, the darker material is FeO. (a) Shows a spark that consists of an FeO shell surrounding an iron sphere containing FeO particles. (b) Shows a smaller, more heavily oxidized particle which consists of an FeO sphere containing iron particles. Note; the particles have a very thin outer skin of Fe2O3 (not visible in these micrographs) |
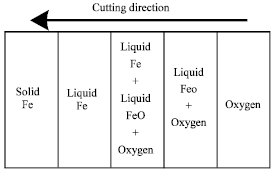 | |
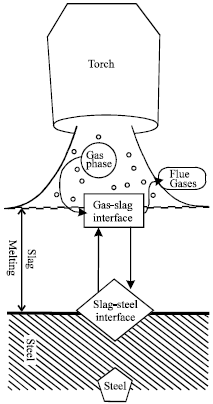
| Fig. 3: | A schematic cross section of the cutting zone (Powell et al., 2009) |
However, Powell et al. (2009) reported that during the rapid high-temperature oxidation cutting of ferrous alloys, as a result of the limited exposure time of the melt to the oxygen combination, not all of the iron in the melt is oxidized. The melt leaves the bottom of the cut zone as incandescent sparks which can be collected and sectioned to reveal their partially oxidized nature (Fig. 2a, b). In a typical sample from CO2 laser/oxygen cutting, Powell et al. (2009) showed that approximately half the iron in the cut zone was found to be oxidized to FeO (the proportion of iron oxidized will, of course, change significantly with process parameters). Figure 3 shows schematic cross section of the cutting zone. The level of formation of the other oxides of iron was found to be negligible (Ivarson et al., 1992). This experimental result is supported by the point that Fe2O3 and Fe3O4 are thermodynamically unstable at the temperatures associated with oxygen cutting. However, it was observed (Ivarson et al., 1992) that each spark was coated in a thin (~1 μm thick) surface layer of Fe2O3.
When steel is cut by oxyfuel process, the iron content of the steel is oxidized to iron oxide, while the carbon content to carbon monoxide. Generally, carbon dissolved in iron is mainly oxidized to carbon monoxide (Min and Fruehan, 1992).
 | |
| Fig. 4: | Pattern of interactions during flame cutting |
The kinetics of the process depends, among other things, on the composition of the cut metal and the activity of oxygen (Powell et al., 2009; Story and Fruehan, 2000). Kinetics of oxidation of iron in molecular oxygen has been studied over a wide temperature range (Zheludkevich et al., 2004). The influence of oxygen pre-dissociation on the activity of oxygen has also been reported. The rate of carbon oxidation, i.e., CO-formation, is found to be proportional to a steady state oxygen activity which is established on the carbon surface by the interplay of oxygen transfer reactions and carbon oxidation. This was proven in detail by Grabke (1999). Kinetics of oxidation reactions of metals at high temperatures, are generally assumed to be controlled by the rates of mass transport of reactants and products in the gas-slag interface, slag-metal interface and slag phases (Mori, 1988). The passage of oxygen from the gaseous phase through the slag to the metal is generally believed to be as shown in Fig. 4 and 5.
However, Schwerdtfeger and Prange (1984) have shown that slag-metal reactions involving iron and/or silicon under industrial process conditions are very unlikely controlled by the transfer kinetics of the reaction. Besides, whatever the means oxidation is being achieved, cutting by oxyfuel is achieved by melting of the slag. The cutting rate by oxyfuel will depend significantly on the rate of slag formation. Any reaction that retards formation of slag affects the cutting rate. FeO is a significant proportion of products of oxidation. When ferrous metals are cut by oxyfuel, the process becomes one of rapid oxidation i.e. burning (Masachuesset Institute of Technology, 2002; AWS, 1987, 1997; De Garmo et al., 1999).
The retardation to the formation of FeO affects the cutting rate. Reactions below (Eq. 6 to 8) are unfavourable to the formation of FeO.
| (6) |
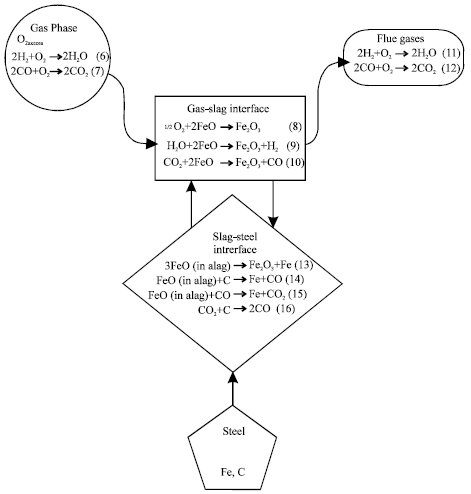 | |
| Fig. 5: | Reactions of slag forming process |
| (7) |
| (8) |
Min et al. (1999) and Kudrin (1985) confirm reaction between iron oxide (FeO) and carbon (C), while Min and Fruehan (1992), Sommerville et al. (1980), Dogan et al. (2009) and Mori, 1988 elucidated on the reactions between iron oxide and carbon monoxide (CO), carbon monoxide and carbon. Generally, high carbon steels are more difficult to flame cut because of the reactions between carbon, carbon monoxide and iron oxide (Higgins, 1993; Masachuesset Institute of Technology, 2002).
THERMODYNAMICS OF OXYFUEL GAS CUTTING OF STEELS
The general equation used to calculate ΔG (the change in Gibbs free energy) is:
| (9) |
where, ÄH is the enthalpy, i.e. the heat energy evolved by the reaction, T is the temperature in K and ΔS is the change in entropy. Equation 9 shows the temperature dependence of ΔG, ΔH is also temperature dependent, as shown in the Eq. 10.
| (10) |
where Cp is the specific heat capacity at constant pressure
During oxyfuel gas cutting, cutting flame only heats the metal to start the process; further heat is provided by the burning metal. It is generally agreed that the exothermic reaction of oxygen with the cut metal provides a considerable thermal input (Al-Mashikhi et al., 2008; Ivarson et al., 1993; Powell et al., 1992). The maximum reaction temperature of the process will consist of the sum of the heat by the flame and that of the exothermic reaction of oxygen with the cut metal. The steel flame cutting process consists of reactions as follows:
| (11) |
| (12) |
| (13) |
While the heat by the flame is expressed as:
| (14) |
| (15) |
The total heat liberated by the cutting process of steel is given as the sum of heat of Eq. 11-13 and those of Eq. 14 and 15. The maximum heat input is given as:
| (16) |
The relationship between the maximum heat input and maximum reaction temperature (Tm) is expressed thus:
| (17) |
where, Cp is the specific heat capacity of products at constant pressure, ni is the mole fractions of component i in the product. Thus;
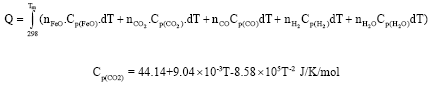 | |
| (18) |
The total value of Q depends on the ratios of nFeO : nCO2: nCO. This ratio is a function of concentration and activity of carbon and oxygen. Kudrin (1985) and Bigeyev (1977) showed that there is increased tendency for formation of carbon dioxide at lower carbon concentrations.
 | |
| Fig. 6: | A typical relationship between cut front temperature and cutting rate (Powell et al., 2009; Miyamoto and Maruo, 1991) |
| (19) |
| (20) |
| (21) |
| (22) |
Since Cp(CO2) is about twice the value of Cp(CO), higher values of nCO2: nCO will give higher heat input. Also, cutting rate has been shown to increase with temperature (Powell et al., 2009). This is shown in Fig. 4. The energy available as heat from the FeO oxidation reaction at atmospheric pressure suddenly rises in energy evolved above the boiling point of iron (Tb; 3330K). The reactions extinct at the dissociation temperature Td which is 3660K (Fig. 5).
Although, the thermal energy generated by the oxidation reaction rises by approximately 350 kJ mol-1 (from 250 to 600) at the onset of boiling (Powell et al., 2009), the latent heat of boiling iron (347 kJ mol-1) almost exactly matches this rise. Thus, if there is no increase in the proportion of iron oxidized there will be no net increase to the thermal input to the cutting process (Fig. 6). The decarburization process retards iron oxidation, thereby reducing the proportion of iron oxidized per time. Generally, high carbon steels are more difficult to flame cut because of the reactions between carbon, carbon monoxide and iron oxide (Masachuesset Institute of Technology, 2002; Kudrin, 1985).
KINETICS OF OXYFUEL GAS CUTTING OF STEELS
Kinetics of reactions is generally assumed to be controlled by collision theory. Collision theory is shown in Fig. 7. In order for any reaction to happen, those particles must first collide.
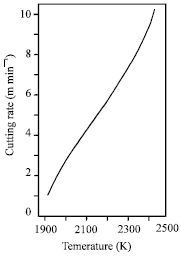
 | |
| Fig. 7: | Variation of enthalpy and Gibbs free energy with temperature for FeO oxidation reaction at atmospheric pressure (Powell et al., 2009) |
This is true whether both particles are in the gas state, or whether one is a gas and the other a solid. If the pressure is higher, the chances of collision are greater.
During oxyfuel gas cutting, the process involved five basic steps which include: Diffusion and collision of gas molecules and/or atoms with the metal, adsorption kinetics of the reactants to the surface of the metal, reaction at the surface desorption kinetics of products from the surface and diffusion of products away from the surface.
Diffusion and Collision of Gas Molecules and/or Atoms
At this stage, increased oxyfuel pressures leads to increased kinetics, this because increased pressure increases both the diffusion rate and the collision rates (Fig. 8).
Adsorption Kinetics of the Reactants to the Surface of the Metal
There are two principal modes of adsorption of gas molecules on surfaces: Physisorption and Chemisorption. At the temperature of oxyfuel cutting, chemisorption is predominant. The rate of adsorption Rads, of oxyfuel molecules onto surface of the cut metal can be expressed in the same manner as any kinetic process.
| (23) |
where, x is the kinetic order k is the rate constant and P is the oxyfuel pressure. If the rate constant is expressed in Arrehenius form, then:
| (24) |
where, Ea is the activation energy for adsorption, R the gas constant, T absolute temperature and A the pre-exponential factor.
 | |
| Fig. 8: | Collision theory |
Generally, the rate of adsorption is governed by: the rate of arrival of gas molecules/atom at the surface and the proportion of incident molecules which undergo adsorption. i.e.,
| (25) |
where, S is the sticking probability and F the incident molecular flux. The flux of incident molecules is given as:
| (26) |
P is gas pressure (Nm-2), m is mass of one molecule/atom (kg). The sticking probability is clearly a property of the adsorbate/ substrate system under consideration (Oxyfuel and steel in this case) but must lie in the range 0<S<1. It may depend upon various factors- foremost amongst these being the existing coverage of adsorbed species (θ) and the presence of any activation barrier to adsorption. In general:
| (27) |
Thus, rate adsorption is expressed as:
| (28) |
This shows that rate of adsorption is proportional to oxyfuel pressures. Thus, Rads increases with oxyfuel pressures.
Reaction at the Surface
In general, rates of chemical reactions are proportional to the concentration of the reacting substances. Increasing the pressure of a gas is exactly the same as increasing its concentration. Using the ideal gas equation:
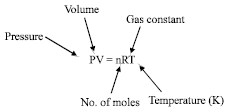 | (29) |
Rearranging this gives:
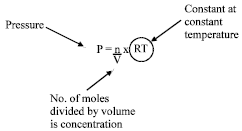 | (30) |
The RT is constant as long as the temperature is constant. During the oxyfuel cutting process, temperature is fairly constant. This shows that the pressure is directly proportional to the concentration. Increasing the pressure increased the cutting rates.
Desorption Kinetics of Products from the Surface
The rate of desorption, Rdes, of an adsorbate from a surface can be expressed in the general form:
| (31) |
where, x is kinetic order of desorption and N is the surface concentration of adsorbed species. In Arrhenius form:
| (32) |
Or alternatively,
| (33) |
However,
| (34) |
θ is the surface coverage of adsorbed molecules/atoms, b is a constant and P pressure of the gas.
If:
| (35) |
Then;
| (36) |
i.e., the desorption rate will be proportional to the pressure on the adsorbed molecules.
If:
| (37) |
Then;
| (38) |
i.e., the desorption rate will be independent of the pressure on the adsorbed molecules.
Diffusion of Products Away from the Surface
The product may diffuse into the bulk of the underlying solid. Carbon monoxide in this case and excess oxygen diffuse into the slag to the slag-steel interface through mass transport processes. Mass transport processes are known to be sensitive to concentration/pressure of the diffusing species. Also, the product of reaction at the surface may react with excess oxygen to yield a gas phase product or other surface species. The CO oxidation reaction is generally believed to be by the following mechanism:
| (39) |
| (40) |
| (41) |
where, rds is Rate Determining Step. As CO2 is comparatively weakly bond to the surface, the desorption of this product molecule is relatively fast and in many circumstances it is the surface reaction between the two adsorbed species that is the rate determining step.
If the two adsorbed molecules are assumed to be mobile on the surface and freely intermix, then the rate of reaction will be given by the following rate expression for the bimolecular surface combination step.
| (42) |
However,
| (44) |
And
| (44) |
Combining the equations into the rate expressions gives:
| (45) |
This also shows sensitivity of this reaction step on oxygen pressure.
Elucidation has been made on oxidation of steel at high temperatures. Iron in the cut zone was found to be oxidized to FeO, the proportion of iron oxidized depends on process parameters. Description of the mechanism of interactions of different phases involved in the reactions of slag forming process when steel is cut has been provided. The roles of carbon and the activity of oxyfuel gases in this process are elucidated. Generally, the cutting rates will decrease with carbon content of steel this is because of the retardation to the formation of FeO by decarburization reactions between iron oxide (FeO) and carbon (C), iron oxide and carbon monoxide (CO), carbon monoxide and carbon. Cutting rates are expected to increase with cutting pressures. Activity of the oxyfuel gases increase with pressure. Theoretical models of the thermodynamics and kinetics of cutting process pertinent to steels are also discussed. These are similar to those applicable in laser-oxygen assisted cutting.