Research Article
Anti-bacterial Effect of Marine Algae against Oral-borne Pathogens
Department of Food and Nutrition, Kongju National University, Yesan, Chungnam 340-702, Korea
Oral cavity is invaded by many bacteria through the breathing and food intake all the time. Physiological conditions of oral cavity are suitable for bacterial growth and eventually the specific oral microbial flora exits in oral cavity. Generally, the oral microbial flora of human includes more than 30 bacteria and have the diverse species spectrum according to the age, health conditions and hygiene-status of individuals. Suppression and control against oral pathogens is important for the care of oral health.
Especially the tooth decay and halitosis are closely associated with oral pathogens. Dental caries is a highly prevalent and a costly disease throughout the world (Hu et al., 2011). Surgeon General’s in United States report showed that 45% of children aged 5-17 dental caries and the children in underserved countries are placed in severe disease risk (Anonymous, 2000).
Dental caries is a destructive disease of dental hard tissues and can progress to inflammation and to death of vital pulp tissue eventually by spread of infection to the periapical area of the tooth. Dental caries is an infectious disease by several species of cariogenic bacteria and specific dietary patterns, such as sugar intake and eating frequency (Ding et al., 2014; Nishimura et al., 2012). Streptococcus mutans, Streptococcus sobrinus and Lactobacillus casei are the well-known cavity-causing bacteria. Among these oral pathogens, S. mutans is the major contributor to tooth decay and acid production in carcinogenic process (Banas, 2013; Kohler et al., 1995; Nakajo et al., 2009). Most of tooth decaying bacteria colonize on the tooth surfaces and produce acids to dissolve tooth structures. Therefore, the anti-microbial treatments against these bacteria may help the control and the prevention of tooth decay for oral health care.
Several researchers have studied the anti-bacterial activity of plant extracts against oral pathogens. Herbal medicine extracts and its constituents, such as (-)-epigalocatechin, have anti-bacterial activities against oral pathogens: S. mutans, Fusobacterium nucleatum, Prevotella intermedia and Porphyromonas gingivalis (Lee et al., 2010; Park et al., 2010; Jeon and Han, 2004). However, as an anti-bacterial natural material against oral-borne pathogens, the marine algae extracts and its constituents have been paid little attention relatively, so far.
Marine algae are considered as sources of natural bioactive compounds, because they have a great variety of secondary metabolites characterized by a broad spectrum of biological activities. Several compounds with cytostatic, anti-viral, anthelmintic, anti-fungal and anti-bacterial activities were studied in green, brown and red algae (Lindequist and Schweder, 2001). There are numerous reports concerning anti-bacterial and anti-fungal activities of macroalgae against bacteria, fungi and yeasts related to human diseases (Wright et al., 2011; Bhatnagar and Kim, 2010). Bromophenol, dolabellane derivatives, phloroglycin and hydroquinone derivatives were isolated as anti-bacterial compounds from red algae, Rhodomelaceae, Dictyota dichotoma, Fucus versiculosus and Dictyopteris zonarioides (Choi et al., 2000; Mayer et al., 2010).
In current study, we focused to screen and investigate Korean marine algae with anti-bacterial activity against tooth decaying bacteria. It is anticipated that marine algae with anti-bacterial activity might be used as a natural anti-bacterial alternative for oral health care.
Sea weeds materials: All marine algae collected from the seashore of Jeju-island Korea, were authenticated and deposited by Jeju Biodiversity Research Institute (JBRI, Jeju, Korea) (Table 1). Marine algae were washed, dried by freeze drier and crushed. Crushed algae were extracted with 70% methanol (in water) at room temperature. Extracts were filtered through Whatman No.1 filter paper and were concentrated by evaporator under reduced pressure. Marine algal extract was redissolved in DMSO to 100 mg mL-1, stored at -20°C and used it as a stock.
Microorganisms and growth condition: Oral bacterial strains used in this study are in the following; S. mutans ATCC (American Type Culture Collection, Manassas, VA, USA) 25175, S. sobrinus ATCC 27607, Streptococcus pyogenes ATCC 21059, Streptococcus anginosus KCTC (Korean Collection for Type Culture, Daejon, Korea) 5141, Staphylococcus aureus ATCC 25923 and Staphylococcus epidermidis ATCC 12228. Streptococcus and Staphylococcus strains were cultured at 37°C in Brain Heart Infusion medium (BHI) (BactoTM, BD 237500, Sparks, MD, USA) and Nutrient Broth (NB) (DifcoTM, BD 234000, USA), respectively. Stock of bacteria were kept in 35% glycerol (in phosphate buffered saline) at -70°C. All bacterial strains were cultured by inoculating 100 μL of the thawed microbial stock suspensions into 5 mL of BHI and NB medium in a shaking incubator at 37°C for 24 h in aerobic condition.
Agar-well diffusion assay: The anti-bacterial activities of marine algal extracts were evaluated by agar-well diffusion assay. The assay was carried out according to the method with some modifications (Owen and Palombo, 2007). After the well was cut on agar plate using a sterile cork-borer (5 mm diameter), 50 μL of each bacterial suspension (OD600 = 0.2-0.3, approximately 1x107 CFU mL-1) was spread uniformly on the surface of each agar plate. Marine algal extracts (10 μg well-1) were dispensed into the wells. And agar plates were left at room temperature for 30 min to allow the liquid to diffuse into the agar before overnight incubation at 37°C.
All assays were carried out in triplicate. A clear zone of the inhibited microbial growth surrounded substances was considered as positive exhibiting anti-bacterial properties. The solvent system used for the preparation of marine algal extract samples was used for blank well. After incubation, the inhibition zones around wells were measured in mm using a caliper (Fazeli et al., 2007). Cetylpridinium chloride (CPC) and streptomycin (10 μg well-1) were used as positive control for anti-bacterial activity.
Statistical analysis: The student’s t-test was used to determine the statistical significance of data value differences. Data was expressed as Means±Standard Deviation (SD).
Using fifty-one species of marine algal extracts as target natural materials, we have screened marine algal species with anti-bacterial activity against oral-borne pathogens. All algal extracts were provided from JBRI and anti-bacterial activity was determined by agar-well diffusion assay. The DMSO, treated as a negative control, did not show any clear zone around wells of all oral pathogens (Fig. 1). In addition, streptomycin and CPC were used as positive controls of anti-bacterial activity for the comparison with marine algae extracts. Figure 1 shows the anti-bacterial activities of Laurencia okamurae Yamada and Dictyopteris undulata Holmes against S. mutans, S. sobrinus and S. pyogenes and the comparison with the anti-bacterial intensity of CPC. Extracts of Laurencia okamurae Yamada and Dictyopteris undulata Holmes had a superior anti-bacterial activity to CPC against S. mutans, S. sobrinus and S. pyogenes.
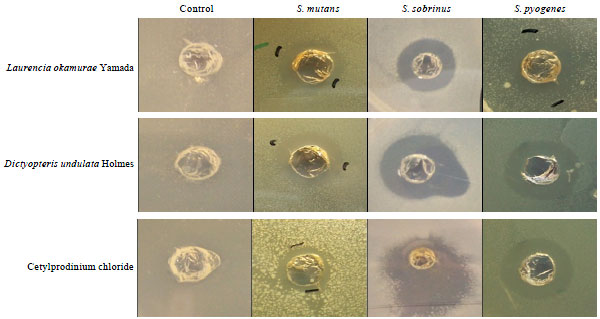 | |
| Fig. 1: | Anti-bacterial activity of Laurencia okamurae Yamada and Dictyopteris undulata Holmes against oral pathogens |
| Table 1: | Agar diffusion susceptibility of oral-pathogens to Korean domestic algal extracts |
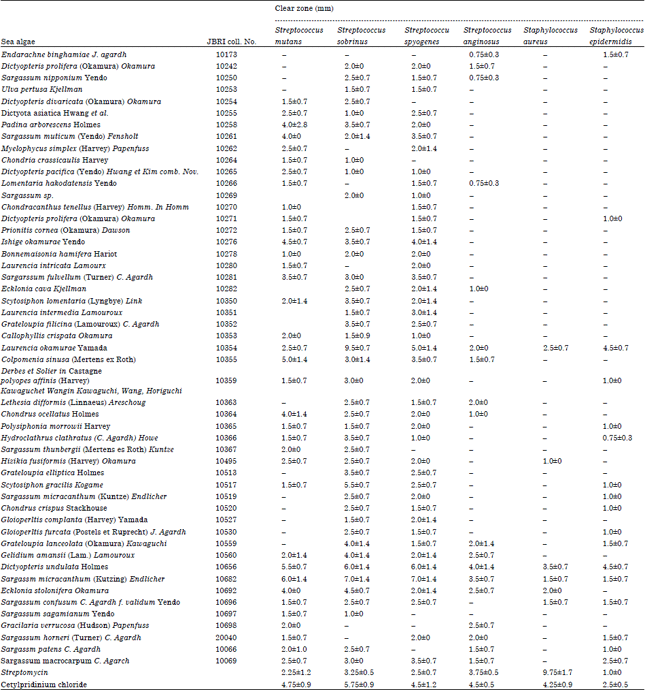 | |
Laurencia okamurae Yamada, Dictyopteris undulata Holmes and Sargassum micracanthum (Kutzing) Endlicher showed the potent anti-bacterial activities against tested all 6 oral pathogens, whereas other algal extracts had the susceptibility against only 2-5 oral pathogens (Table 1). Sargassum micracanthum (Kutzing) Endlicher showed the strongest anti-bacterial activity with 6.0±1.4 mm of clear zone against S. mutans, a major cariogenic bacterial species. Additionally, the growth of S. sobrinus and S. pyogene was inhibited the most effectively by extract of Sargassum micracanthum (Kutzing) Endlicher. Intensity of anti-bacterial activity against oral-borne pathogens showed the following order; Sargassum micracanthum (Kutzing) Endlicher> Dictyopteris undulata Holmes> Laurencia okamurae Yamada. Laurencia okamurae Yamada had the strongest susceptibility with 9.5±0.7 mm of clear zone against only S. sobrinus. And CPC, an anti-bacterial agent in commercial mouthwashes had the better anti-bacterial activity with 4.75±0.9, 5.75±0.9, 4.5±1.2, 4.5±0.5, 4.25±0.9 and 2.5±0.5 mm of clear zone against S. mutans, S. sobrinus, S. pyogene, S. anginosus, S. aureus and S. epidermidis, respectively, than streptomycin. Extracts of Laurencia okamurae Yamada, Dictyopteris undulata Holmes and Sargassm micracanthum (Kutzing) Endlicher were investigated to have the superior anti-bacterial activities to streptomycin and CPC against oral pathogens.
Interest in the personal beauty and health care has been increasing recently. Oral health is no exception to this interest and many oral care products are selling commercially. Simultaneously the public demands for the natural material without bad side-effects are surging up rapidly. Cariogenic S. mutans, S. sobrinus and S. pyogene had the great susceptibilities against most of marine algal extracts, as compared with other oral pathogens (Table 1). Extracts of nine marine algae, including Padina arborescens Holmes, Sargassum muticum (Yendo) Fensholt, Ishige okamurae Yendo, Sargarssum fulvellum (Turner) C. agardh, Colpomenia sinusa (Mertens ex Roth) Derbes et Solier in Castagne, Chondrus ocellatus Holmes, Dictyopteris undulata Holmes, Sargassm micracanthum (Kutzing) Endlicher and Ecklonia stolonifera Okamura, showed the potent anti-bacterial activity against S. mutans. And the growth of S. sobrinus was inhibited by treatment of Padina arborescens Holmes, Ishige okamurae Yendo, Scytosiphon lomentaria (Lyngbye) Link, Grateloupia filicina (Lamouroux) C. agardh, Laurencia okamurae Yamada, Scytosiphon gracilis Kogame, Grateloupia lanceolata (Okamura) Kawaguchi, Gelidium amansii (Lam.) Lamouroux, Dictyopteris undulata Holmes, Sargassm micracanthum (Kutzing) Endlicher and Ecklonia stolonifera Okamura. Also the extracts of Sargassum muticum (Yendo) Fensholt, Ishige okamurae Yendo, Sargarssum fulvellum (Turner) C. agardh, Laurencia okamurae Yamada, Colpomenia sinusa (Mertens ex Roth) Derbes et Solier in Castagne, Dictyopteris undulata Holmes and Sargassm micracanthum (Kutzing) Endlicher showed the anti-bacterial activities against S. pyogene. Anti-bacterial activities against S. anginosus, S. aureus and S. epidermis were observed by the treatment of Laurencia okamurae Yamada, Dictyopteris undulata Holmes and Sargassm micracanthum (Kutzing) Endlicher.
Among natural materials available to us, the marine algae are being recognized as potential and promising sources of bioactive compounds (Newman et al., 2003). Marine algae produce a great variety of secondary metabolites with versatile activities, such as anti-viral, anthelmintic, anti-fungal and anti-bacterial activities (Lindequist and Schweder, 2001; Bansemir et al., 2006). Red algal genus Laurencia (Rhodomelaceae) produces numerous specialized secondary metabolites with diverse structural features. Its three major classes which are sesquiterpenes, diterpenes and acetylenes have not been yet encountered in other terrestrial organisms (Blunt et al., 2008; Suzuki et al., 2009). Most of these metabolites are characterized by the presence of halogen atoms in their chemical formula. Generally Laurencia species produce bromine-containing compounds in far greater numbers than either chlorine or iodine-containing compounds. Halogenated compounds and their metabolites from Laurencia sp. were investigated to exhibit antimicrobial, insecticidal, cytotoxic activities (Suzuki et al., 2009).
Brown algae Dictyotaceae (Phaeophyceae) is a class of seaweeds that have been explored for the bioactivity potential of its metabolic products. Dictyotaceae contains a rich and diverse source of natural products which makes it the most studied with the great number of known metabolites. The most bioactive products, isolated from Dictyotaceae, contains three or four isoprenoid units in sesquiterpenes and diterpenes, respectively (Dewick, 2002). These two classes of terpenoids have many different structures and variants by addition of halogen atoms or fragments from other biogenic pathways. Due to the diverse compounds in Dictyopteris sp., it might be used for anti-viral and anti-bacterial studies (De Paula et al., 2011; Ji et al., 2009).
Dental caries and periodontal diseases are related to the oesophagus and stomach diseases, as well as oral health problems. Therefore, there is a growing interest in the personal hygiene products for maintaining oral healthy recently and the ingredients in mouthwashes have been exchanged to the biosafe natural products from chemicals, along with food and pharmaceutical materials. However, any other alternative natural product against CPC, an antibacterial agent in mouthwashes for prevention of dental caries and halitosis has not been utilized generally in present. For this reason, among Korean algae which are unknown to have the bioactivities so far, I was going to screen algae species with antibacterial activity against oral-borne pathogens as a candidate substance for alternative to chemical antibacterial agent. In this report, we elucidated that the anti-bacterial activities of Laurencia okamurae Yamada and Dictyopteris undulata Holmes against 5 oral pathogens, such as S. mutans, S. sobrinus, S. pyogenes, S. anginosus, S. aureus and S. epidermidis, were superior to antibiotics streptomycin and CPC. Based on this result, it is expected that some further researches, including safety study in human, make Laurencia okamurae Yamada and Dictyopteris undulata Holmes-extracts to be able to use as an alternative agent for CPC. Also Laurencia okamurae Yamada and Dictyopteris undulata Holmes are expected to be able to develop as a potential phytotherapeutic drug for treatment and prevention of oral caries and oral health care.