Research Article
Taxonomic Significance of Foliar Epidermis in Some Members of Euphorbiaceae Family in Nigeria
Department of Biological Sciences, University of Agriculture, Abeokuta, Ogun State, Nigeria
D.U. Nwoye
Department of Biological Sciences, University of Agriculture, Abeokuta, Ogun State, Nigeria
A.A. Jayeola
Department of Botany and Microbiology, University of Ibadan, Ibadan, Oyo State, Nigeria
A.O. Olagoke
Department of Forestry and Wood Technology, Federal University of Technology, Akure, Ondo State, Nigeria
A.A. Ogundele
Department of Biology, Federal College of Education (Special), Oyo, Nigeria









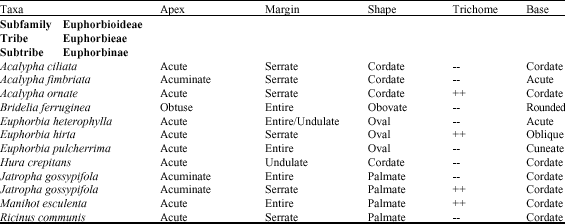
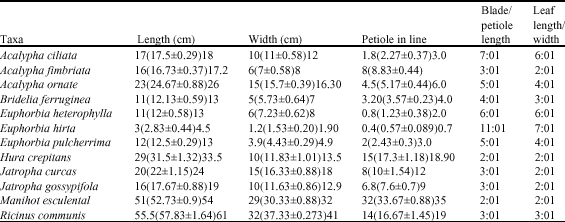
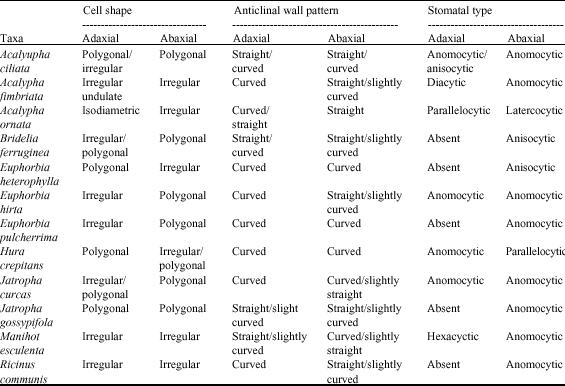
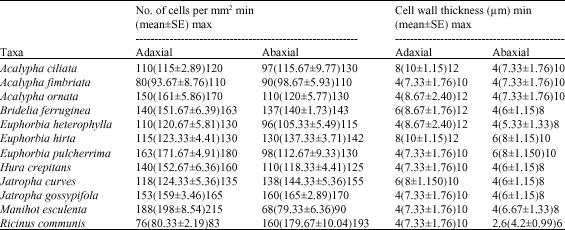

darlington nwoye Reply
well taken and explanatory...please keep it up!