Research Article
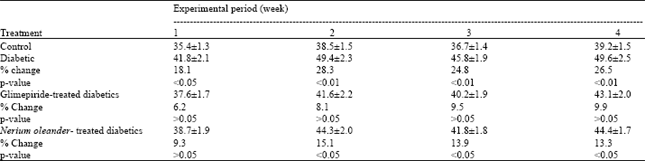
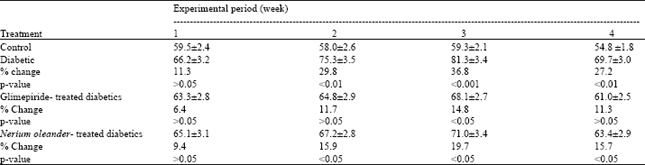
Antidiabetic Activity Evaluation of Glimepiride and Nerium oleander Extract on Insulin, Glucose Levels and Some Liver Enzymes Activities in Experimental Diabetic Rat Model
Department of Biology, Faculty of Science, Al-Azhar University of Gaza, P.O. Box 1277, Gaza Strip, Palestine
Maged M. Yassin
Faculty of Medicine, The Islamic University of Gaza, P.O. Box 108, Gaza Strip, Palestine