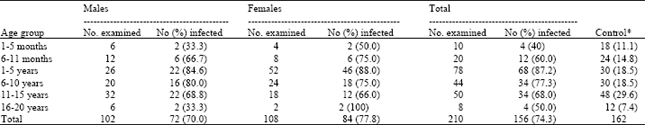
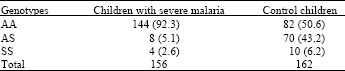
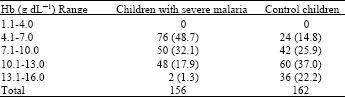Research Article
Susceptibility of Genetic Indices to Falciparum Malaria in Infants and Young Children in Southern Nigeria
Department of Zoology, University of Uyo, Uyo, Nigeria
I. A. Atting
Department of Medical Microbiology, College of Health Sciences, University of Uyo, Uyo, Nigeria
I. G. Ukpong
Department of Animal and Environmental Biology, University of Port Harcourt, Port Harcourt, Nigeria
A. A. Nwabueze
Department of Animal Science and Fisheries, Delta State University, Asaba, Nigeria
I. I. Inokon
Department of Zoology, University of Uyo, Uyo, Nigeria