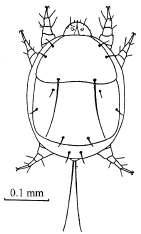
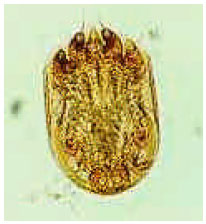
Research Article
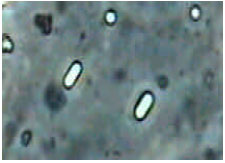
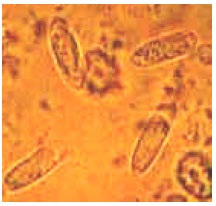
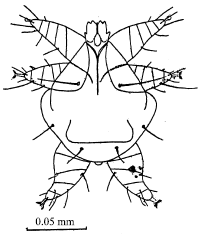
An Investigation on Some Diseases and Parasites of Bumblebee Queens (Bombus terrestris L.) in Turkey
Black Sea Agricultural Research Institute, Samsun, Turkey
Osman Kaftanoglu
Department of Animal Science, Faculty of Agriculture, cukurova University, Adana, Turkey