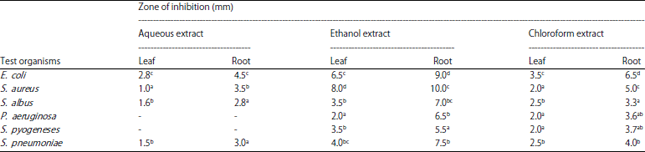
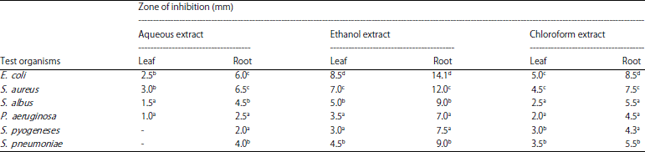
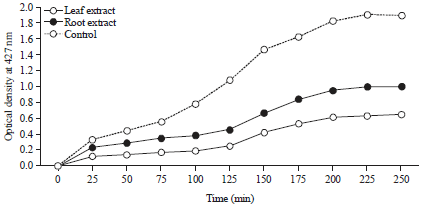
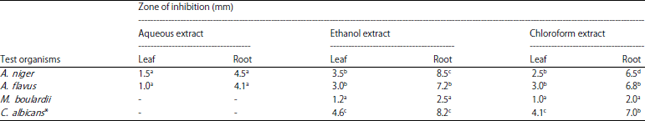
Research Article
Antimicrobial Activities of Centella asiatica Leaf and Root Extracts on Selected Pathogenic Micro-organisms
Department of Biology, Faculty of Mathematics and Natural Sciences, Medan State University (Universitas Negeri Medan), 20221, Medan, Sumatera Utara, Indonesia
Martina Restuati
Department of Biology, Faculty of Mathematics and Natural Sciences, Medan State University (Universitas Negeri Medan), 20221, Medan, Sumatera Utara, Indonesia
Ahmad Shafwan S. Pulungan
Department of Biology, Faculty of Mathematics and Natural Sciences, Medan State University (Universitas Negeri Medan), 20221, Medan, Sumatera Utara, Indonesia
Nanda Pratiwi
Department of Biology, Faculty of Mathematics and Natural Sciences, Medan State University (Universitas Negeri Medan), 20221, Medan, Sumatera Utara, Indonesia
Diky Setya Diningrat
Department of Biology, Faculty of Mathematics and Natural Sciences, Medan State University (Universitas Negeri Medan), 20221, Medan, Sumatera Utara, Indonesia
LiveDNA: 62.20093