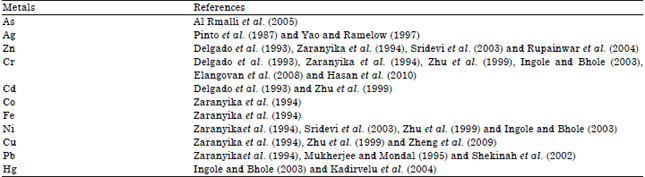
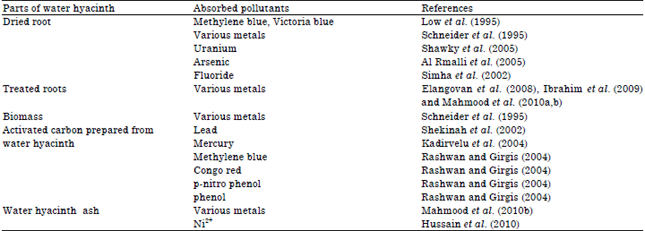
Review Article
Beyond Biocontrol: Water Hyacinth-Opportunities and Challenges
Department of Microbiology, Kurukshetra University, Kurukshetra, Haryana, 136119, India
Neeraj K. Aggarwal
Department of Microbiology, Kurukshetra University, Kurukshetra, Haryana, 136119, India
Anita Saini
Department of Microbiology, Kurukshetra University, Kurukshetra, Haryana, 136119, India
Anita Yadav
Department of Biotechnology, Kurukshetra University, Kurukshetra, Haryana, 136119, India