Review Article
A Review on Petroleum: Source, Uses, Processing, Products and the Environment
Institute for Development Studies, Enugu Campus, University of Nigeria, Nsukka, Nigeria
The word "petroleum" means "rock oil" or "oil from the earth" (EIA, 2005).
Mankind has known petroleum or crude oil since the dawn of civilisation. It was used in ancient Persia and Burma, particularly as fuel for lamps. Burning of the natural gas (escaping from petroleum underground) gave the ‘perpetual fire’ at Baba Gurgur in Iraq (Arene and Kitwood, 1979).
USES OF PETROLEUM
Petroleum has two main uses. The first is as a fuel. The combustion of crude oil gave in-road to derivation of various energies from petroleum. Petroleum has been a source of energy for heating, lighting and locomotion and particularly the most convenient fuel for the internal combustion engine. This use has increased rapidly in importance with the coming of the motor car and a wide range of other applications of internal combustion engine (Bankole and Ogunkoya, 1978).
The second use of petroleum is the synthesis of organic compounds. By 1965, about 80% of the world organic chemicals were synthesized from petroleum. This figure rose to 98% in 1980 and 99% in the year 2000. Thus, petroleum chemicals (petrochemicals) are a wide variety of chemicals. Commercially important ones include gasoline and kerosine. Petroleum jelly is a greasy gelatinous substance sourced from petroleum and used as ointment base, lubricant and protective covering (Arene and Kitwood, 1979).
THE ORIGIN OF PETROLEUM
Nelson (1954) wrote on two plausible schools of thought on the origin and formation of petroleum. The older one, "the organic matter theory", suggests that petroleum was formed from the decomposition of dead marine organisms, like plankton. Compounds (e.g., fats) which are very similar to hydrocarbons and even traces of certain hydrocarbons themselves, are present in virtually all forms of plant and animal life. The theory is supported by the finding that very recent marine deposits (10,000 to 15,000 years old) contain hydrocarbon and asphaltic material. Besides, there is a similarity between the molecular structures of some of the minor constituents of crude oil and those of compounds found in living organisms. Again, it is hard to see where else the carbon content of petroleum could have come from other than biological material, if indeed its origin is geologically fairly recent.
Also, supporting this view, Arene and Kitwood (1979) submit that petroleum is crude oil that occurs naturally in sedimentary rocks. It had collected into small pools from seepage from underground. Aligning with this claim, EIA (2005) states that petroleum was formed from the remains of animals and plants that lived millions of years ago in a marine (water) environment before the dinosaurs. About 300-400 million years ago, tiny sea plants and animals died and were buried on the ocean floor. Over time, layers of silt and sand covered them. About 50-100 million years ago, the remains were buried deeper and deeper and covered by layers of mud. The enormous heat and pressure from these layers turned the remains into crude oil and gas. Today, drilling down through the layers of silt, sand and rock help to reach the rock formations that contain crude oil and gas deposits.
The challenge for the theory is accounting for the enormity of petroleum deposits. For such a vast amount of petroleum to be formed, the marine organisms would have to be abundant over several million years. Another challenge is accounting for the accumulation of the oil in large deposits. For this to happen, the dispersed droplets of crude oil must have migrated a very long distance. But, it is doubtful whether a mechanism within the conditions and time-span favoured this.
The more recent school of thought, "atmospheric chemical reaction theory", submits that petroleum is very much older than the existence of life on the earth. The earth is over 5,000 million years old and at the point when it was a hot molten mass, consisting mainly of Hydrogen (H) and Carbon Dioxide (CO2), chemical reactions between them formed a great range of hydrocarbons which rose to the atmosphere as vapour clouds similar to those suspected to surround the planet Venus today:
 |
As cooling of the earth proceeded, condensation led to a ‘petroleum-rain’ that lasted 2,000 million years. Many valleys and basins collected the oil with its sediments. Much latter, water-rain came, on which the oil floats.
The problem of accounting for enormous size of the earth’s crude oil deposits is common to both theories.
PETROLEUM EXPLORATION AND MAJOR PETROLEUM PRODUCING COUNTRIES
Arene and Kitwood (1979) note that crude oil is capable of flowing up the well pipe either under the pressure existing in the reservoir, or with mechanical assistance from a pump or applied pressure. Since Drake, in Pennsylvania, United States of America (USA), drilled crude oil, the petroleum industry has steadily grown and a large number of petroleum deposits have been discovered and drilled in various parts of the world.
According to EIA (2005), scientists and engineers explore a chosen area by studying rock samples from the earth. Measurements are taken and, if the site seems promising, drilling begins. Above the hole, a structure called a 'derrick' is built to house the tools and pipes going into the well. When finished, the drilled well will bring a steady flow of oil to the surface.
The world's top five crude oil-producing countries are Saudi Arabia, Russia, United States, Iran and China. Over one-fourth of the crude oil produced in the United States is produced offshore in the Gulf of Mexico. The top crude oil-producing states are Texas, Alaska, California, Louisiana and New Mexico (NRC, 2003).
The amount of crude oil produced in the United States has been getting smaller each year. On the other hand, the use of products made from crude oil has been growing, making it necessary to bring more oil from other petroleum producing countries. Thus, about 58% of the crude oil and petroleum products used in the United States come from other countries.
According to the OPEC (2011), the Organization of the Petroleum Exporting Countries (OPEC) was founded in Baghdad, Iraq, with the signing of an agreement in September 1960 by five countries, namely Islamic Republic of Iran, Iraq, Kuwait, Saudi Arabia and Venezuela. They were to become the Founder Members of the Organization.
Qatar (1961), Socialist People’s Libyan Arab Jamahiriya (1962), the United Arab Emirates (1967), Algeria (1969), Nigeria (1971), Ecuador (1973) and Angola (2007) later joined these countries.
From December 1992 until October 2007, Ecuador suspended its membership. Gabon terminated its membership in 1995. Indonesia suspended its membership effective January 2009. Thus, currently, the Organization has a total of 12 Member Countries: Islamic Republic of Iran, Iraq, Kuwait, Saudi Arabia, Venezuela, Qatar, Socialist People’s Libyan Arab Jamahiriya, the United Arab Emirates, Algeria, Nigeria, Ecuador and Angola.
The OPEC Statute distinguishes between the Founder Members and Full Members - those countries whose applications for membership have been accepted by the Conference. It stipulates that “any country with a substantial net export of crude petroleum which has fundamentally similar interests to those of Member Countries, may become a Full Member of the Organization, if accepted by a majority of three-fourths of Full Members, including the concurring votes of all Founder Members.” The Statute further provides for Associate Members who are those countries that do not qualify for full membership, but are nevertheless admitted under such special conditions as may be prescribed by the Conference.
THE NATURE AND COMPOSITION OF PETROLEUM
Crude oil is a smelly, yellow-to-black liquid and is usually found in underground areas, called reservoirs (EIA, 2005). Bankole and Ogunkoya ( 1978), Hobson (1973), Sweeney (1950), Sachanen (1945), McCain (1970) Shreve and Brink (1977) and Arene and Kitwood (1979) have observed that crude oil consists mainly of a mixture of gaseous, liquid and solid hydrocarbons. Some crude oils have sulphur-containing compounds, including the thiol (mercaptans), such as ethanethiol (C2H5SH) and cyclic sulphides, such as the thiophen derivatives (e.g., tetrahydrothiophen and benzothiophen). Others also contain organic nitrogen compounds. The nitrogen is usually present in complex ring structures, such as carbazole:
 |
Again, some crude oils contain small amounts of organic acids, whose molecules have the carbonyl group (-COOH) (e.g., benzenecarboxylic acid). Besides, some crude oils contain traces of phenol-type compounds, in which a hydroxyl group (-OH) is directly attached to a benzene ring (e.g., phenol):
 |
Shreve and Brink (1977), Nelson (1958), Rossini (1947), Sweeney (1950) and Arene and Kitwood (1979) have noted that petroleum is a highly complex mixture, the main constituents being compounds (gases, liquids and solid) of hydrocarbons (11-15% hydrogen and 83-87% carbon) which are classified as alkanes, cycloalkanes and aromatics. Crude oil is a viscous mobile liquid mixed with water and sand. Its appearance ranges from colourless to green-black. Crude oils vary in density between 0.78 and 1.00 gcm-3.
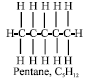
Alkanes make up 90% of most crude oils. They are compounds of hydrogen and carbon only. Some alkanes have ‘straight’ chain, e.g., pentane (Arene and Kitwood, 1979):
 |
Others have branched chains, e.g., 2,2,4-trimethylpentane (iso-octane) (Arene and Kitwood, 1979):
 |
Straight-chain alkanes are undesirable in petrol, but desirable in kerosines and diesel fuels. Also, the straight-chain alkanes are preferred for the manufacture of detergents and for the bacteriological production of protein (Eneh, 2000a).
Cycloalkanes are ring-structure hydrocarbons with single bonding only, e.g., cyclopentane, cyclohexane, etc:
 |
Aromatics have molecules containing the ring structure characteristic of benzene, e.g. benzene, ethylbenzene, 1,2-dimethylbenzene, naphthalene and biphenyl (Arene and Kitwood, 1979):
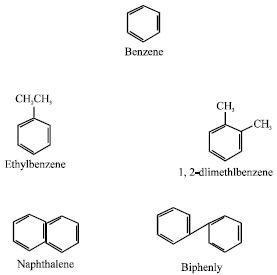 |
PETROLEUM PROCESSING AND PRODUCTS
According to Sharma (2002), Bankole and Ogunkoya (1978), Hobson (1973), McCain (1970), Kimberlin (1957), Rossini and Mair (1951), Shreve and Brink (1977), Arene and Kitwood (1979), Eneh (2007, 2000a, b), USDE (1999) and Saepudin et al. (2010), the process of getting useful products from crude oil is known as refining. After crude oil is removed from the ground, it is sent by pipeline, ship or barge to a refinery, where different parts of the crude oil are separated into useable petroleum products.
Both kerosine and petrol were obtained from petroleum by fractional distillation. With growing demand for more efficient products than the products of crude oil distillation, a large number of techniques were developed for converting the primary distillation products into useful chemicals.
Filtration separates sand and other solid matters from the crude oil which is allowed to stand in reservoir for the more dense water to form a lower layer from the oil layer above. The latter is pumped through pipelines to the refinery, where continuous distillation or fractionation separates the crude oil into a number of ‘cuts’ or fractions of different boiling ranges. Table 1 contains products from a typical fractionation.
The boiling points of products from petroleum refining range from 20 to above 400°C. The boiling range is related to the number of carbon atoms of the compound and is associated with the nature-gas or liquid-and use of the product. For example, compounds with C5-C10 have boiling range of 20-160°C and are mostly liquids.
The proportions of each fraction obtained vary both with the type of crude oil and the distillation process. For example, the yields of the most useful fractions from Nigeria’s refinery are approximately petrol (10%), kerosine (20%) and gas (30%). There is demand for Nigeria’s crude oil because of its low sulphur content (0.14%), as against others. For instance, the crude oil from Persian Golf contains about 7% sulphur. Petroleum was discovered in Nigeria in Oloibiri town in the present Bayelsa State in 1956 and commercial production started in 1957, rising steadily since then (Schatze, 1970; Arene and Kitwood, 1979).
Crude oil is measured in Barrels (bbls). A 42-U.S. gallon bbl of crude oil provides slightly more than 44 gallons of petroleum products. One barrel of crude oil, when refined, produces about 20 gallons of finished motor gasoline (19.6%), 7 gallons of diesel (10%), jet fuel (4%), heavy fuel oil (1.7%), liquefied petroleum gas, LPG (1.7%), as well as other petroleum products (7.6%). The gain from processing is about 5%. Most of the petroleum products are used to produce energy. For instance, many people across the United States use propane to heat their homes and fuel their cars. Other products made from petroleum include: ink, crayons, bubble gum, dishwashing liquids, deodorant, eyeglasses, records, tires, ammonia and heat valves (USDE, 1999).
Thus, primary distillation of crude oil yields petrol which is small relative to demand and yields far more of the higher-boiling constituents than required. Cracking is the method devised for conversion of the large hydrocarbon molecules into smaller ones. The old thermal cracking used 300-700oC temperatures and pressures up to 30 atmospheres. According to Arene and Kitwood (1979), it has been largely replaced by catalytic cracking, using catalyst (e.g. silicon IV oxide, aluminium III oxide), about 500oC temperature and about atmospheric pressure.
| Table 1: | Petroleum fractions |
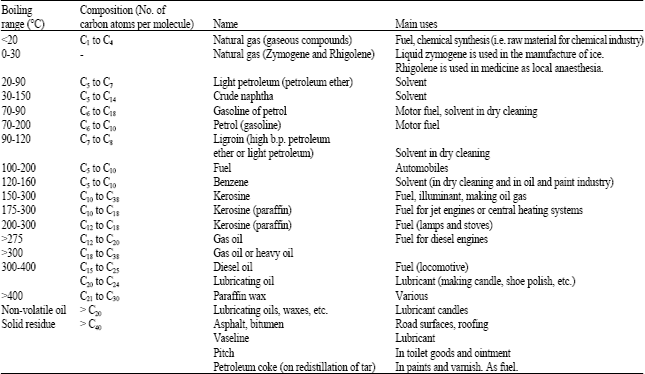 | |
| Source: Sharma (2002), Arene and Kitwood (1979) and Bankole and Ogunkoya (1978) | |
Catalytic cracking is easier to control and yields specific kinds of products. The primary cracking process breaks alkane into alkene molecule (with carbon-carbon double bond, C = C) and a small alkane molecule:
R-CH2-CH2-CH2-CH2-R1→R → CH2-CH = CH2 + CH3-R1 |
The resulting alkene is so reactive that it can undergo isomerisation (changing its structure). It can also undergo polymerisation (joining together to form larger unit). In both processes it yields not only hydrocarbons suitable for petrol, kerosine and diesel but several gases (butane and butene, 45%; propane and propene, 3%; ethane and ethene, 15%; methane 10%; and a little hydrogen). These are important as raw materials for the petrochemical industry. Typically, the total gas yield is 15-20% of the charge.
Hydrocracking is a catalytic cracking process designed to increase the yield of the petrol by supplying hydrogen to enhance the formation of lower alkanes at high pressures (150-200 atmospheres) and moderate temperatures (400-450°C). The catalysts may be aluminosilicate materials or cation of palladium and nickel. The products from Hydrocracking are largely alkanes, unlike those from simple cracking which contain alkenes also. Reforming is a process of alteration of the molecular structure of the hydrocarbons in the lower fractions obtained from primary distillation or from cracking. The yields are branched-chain hydrocarbons in the C6 to C10 range which are needed for high-grade motor fuel. The ‘feed’ generally consists of material boiling up to about 200oC. The most effective catalyst consists of platinum (about 0.5% by weight) on highly purified aluminium (III) oxide. Three kinds of reaction typical of reforming are:
Aromatisation or formation of an aromatic (benzene-type) compound from a cycloalkane, e.g.:
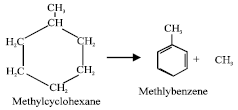 |
Dehydrocyclisation or formation of a compound with a ring structure from one with a straight chain, followed by loss of hydrogen, e.g.:
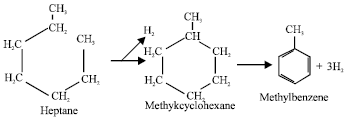 |
Isomerisation or rearrangement, without net loss of any constituent, e.g.:
 |
OCTANE NUMBER
According to Sharma (2002) and Arene and Kitwood (1979), a poor quality fuel tends to ‘knock’ or explode unevenly and prematurely, especially in a high-compression engine. 2,2,4-Trimethylpentane (wrongly called iso-octane) is a good fuel, because it burns smoothly and does not cause knocking. It is, therefore, assigned the arbitrary octane number of 100 (on a zero-to-100 scale). On the other hand, heptane is a bad fuel, because it is particularly inclined to causing knocking. It is, therefore, assigned the octane number of zero. The octane number or octane rating of petrol is, therefore, the percentage of ‘iso-octane’ blended with heptane which reflects the knocking characteristics of the fuel. It is a measure of its performance in an internal-combustion engine.
The quality of the fuel can be improved (or the octane number or octane rating of the fuel can be raised) by addition of branched-chain hydrocarbons or aromatic compounds. Ethanol and methanol are added to some fuels to improve their performance. Also, tetraethyl-lead (IV), Pb(C2H5)4, added in small proportions, greatly increases the octane number of a fuel; 0.03% by weight may raise the octane number by 15-20 units. However, this practice has been discouraged and outlawed in most countries because of environmental pollution and lead-poisoning that result from the use of such fuels (Kymisis and Hadjistavrou, 2008; Goss et al., 2004; Kim and AAPCEH, 2004).
THE PETROCHEMICAL INDUSTRY
According to Bankole and Ogunkoya (1978), Tedder et al. (1979), Waddams (1973), Shreve and Brink (1977) and Kimberlin (1957), the production of chemicals from petroleum has increased enormously since World War II.
| Table 2: | Petrochemicals and their products |
 | |
| Source: Waddams (1973), Kimberlin (1957) and Arene and Kitwood (1979) | |
Even, inorganic chemicals have been produced from petrochemical, e.g., carbon black from methane. Table 2 lists some chemicals produced from petrochemicals.
For instance, methane, ethyne and synthesis gas are petrochemicals from which other chemicals are produced. From methane, carbon black, ethyne, synthesis gas, halogenmethanes and hydrogen cyanide are produced. From ethyne, chloroethene compounds and ethanal are produced. And, from synthesis gas, methanol and ammonia are produced.
PETROLEUM AND THE ENVIRONMENT
Even though petroleum products make life easier by their use in fueling airplanes, cars, trucks, cooking stoves and other applications in combustion engines, as well as in heating our homes, using them can cause environmental problems, like air and water pollution. When petroleum products are burned as fuel, they give off carbon dioxide (CO2), a greenhouse gas that is linked with global warming. The use of petroleum products also gives off pollutants-Carbon monoxide (CO), nitrogen oxides, particulate matter and unburned hydrocarbons-that pollute the air humans breathe. Since a lot of air pollution comes from cars and trucks, many environmental laws have been aimed at changing the make-up of gasoline and diesel fuel so that they produce fewer emissions. These "reformulated fuels" are much cleaner burning than gasoline and diesel fuel was in 1990. In the next few years, the amount of sulphur contained in gasoline and diesel fuel will be reduced dramatically so that they can be used with new, less-polluting again technology (USDE, 1999).
Exploring and drilling for oil may disturb land and ocean habitats. A study on assessment of the impact of oil exploration activities on agriculture and natural resources in the Niger Delta Region of Nigeria showed that oil exploration activities adversely impacted specifically on soil/land resources, aquatic life/fisheries, water resources, crops, livestock and forests/vegetation. Oil spills have degraded most agricultural lands, reduced the availability of fish and fish products, caused the pollution of surface and ground water resources, destruction of arable and tree crops and death of farm animals in the region as a result of toxic materials in the soil and polluted water. Oil exploration activities have also resulted in the disappearance of some forest vegetation and animal species, including primates, fish, turtles and birds. The ultimate result of these impacts is a drastic reduction in farm productivity and animal farm income (Ugwu, 2009; Eneh, 2011a, b; Tabieh and Al-Horani, 2010; Al-Turki, 2010).
New technologies have greatly reduced the number and size of areas disturbed by drilling, sometimes called "footprints." Satellites, global positioning systems, remote sensing devices and 3-D and 4-D seismic technologies, make it possible to discover oil reserves while drilling fewer wells. Again, the use of horizontal and directional drilling makes it possible for a single well to produce oil from much bigger areas. Today's production footprints are only about one-fourth the size of those of 30 years ago, due to the development of movable drilling rigs and smaller "slimhole" drilling rigs. When the oil in a well is gone, the well must be plugged below ground, although its soil fertility is gone. As part of the "rig-to-reefs" programme, some old offshore rigs are toppled and left on the sea floor to become artificial reefs that attract fish and other marine life. Within six months to a year after a rig is toppled, it becomes covered with barnacles, coral, sponges, clams and other sea creatures (USDE, 1999).
If oil is spilled into rivers or oceans it can harm wildlife. Oil spills can come from natural oil seeps from the ocean floor, ships that crash, or leaks that happen when petroleum products are used on land, such as the gasoline that sometimes drips onto the ground when people are filling their gas tanks, motor oil that gets thrown away after an oil change, or fuel that escapes from a leaky storage tank. When it rains, the spilled products get washed into the gutter and eventually go to rivers and the ocean. Another way that oil sometimes gets into water is when fuel is leaked from motorboats and jet skis (USDE, 1999).
When a leak in a storage tank or pipeline occurs, petroleum products can also get into the ground. In some places where gasoline has leaked from storage tanks, one of the gasoline ingredients, called Methyl Tertiary Butyl Ether (MTBE), has made its way into local water supplies. Since MTBE makes water taste bad and many people are worried about drinking it, a number of states are banning the use of MTBE in gasoline and the refining industry is voluntarily moving away from using it when blending reformulated gasoline. To prevent leaks from underground storage tanks, all buried tanks are supposed to be replaced by tanks with a double lining (USDE, 1999).
The review has attempted to marry dated and current literature on petroleum. It has brought out clearly the meaning of petroleum from the Medieval Latin words, petra and oleum; the uses of petroleum, the two plausible theories on the origin and formation of petroleum; petroleum exploration and the major petroleum producing countries in the world, including the member states of the Organisation of the Petroleum Exporting Countries, OPEC; the nature and composition of petroleum; petroleum processing and products; octane number or rating, the petrochemical industry; and petroleum and the environment.