Research Article
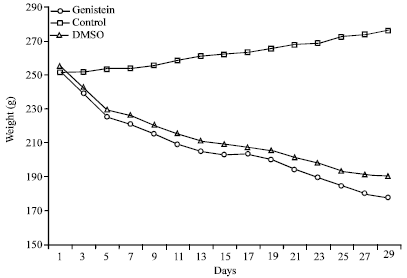
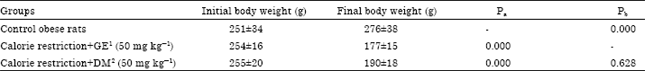
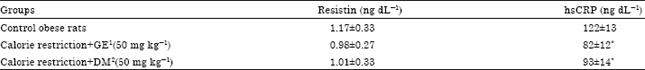
Effect of Calorie Restriction Supplemented with Genistein on Serum Levels of Glucose, Lipid Profile and Inflammatory Markers (Resistin and hsCRP) in Obese Rats
Nutrition Research Center, Jundishapur University of Medical Sciences, Ahvaz, Iran
H. Rafiei
Department of Nutrition, Faculty of Para medicine, Jundishapur University of Medical Sciences, Ahvaz, Iran
M. Karandish
Diabetes Research Center, Jundishapur University of Medical Sciences, Ahvaz, Iran
K. Omidian
Department of Nutrition, Faculty of Para medicine, Jundishapur University of Medical Sciences, Ahvaz, Iran
F. Haidari
Nutrition Research Center, Jundishapur University of Medical Sciences, Ahvaz, Iran