ABSTRACT
Eleven corn samples (9.2%) out of 120 tested proved to be toxic. Aflatoxins B1 and B2 were detected in (2 samples, 9.1-13.9 μg kg-1 corn), aflatoxins B1, B2, G1 and G2 (4 samples, 7.6-16.4), T-2 toxin (3 samples, 8.4-10.2), diacetoxyscirpenol (5 samples, 6.2-9.5) and zearalenone, (5 samples, 6.8-7.9). A. flavus, A. parasiticus and A. flavo-furcatis were the most prevalent Aspergillus species on 6 aflatoxins-contaminated corn samples. Aflatoxins B1 and B2 were produced by (12/18 isolates of A. parasiticus, 88.2-126.8 μg g-1 corn), while aflatoxins B1, B2, G1 and G2 (13/29 isolates of A. flavus, 48.2-146.4) in addition to (2/18 isolates of A. flavo-furcatis, 28.6 and 36.2). This is the first report on aflatoxin production by A. flavo-furcatis. F. tricinctum, F. oysporum and F. equiseti were the dominant Fusarium species on 5 corn samples contaminated by microcyclic tricothecenes. T-2 toxin (15/20 isolates, 24.4-130.2 μg g-1 corn), diacetoxyscirpenol (18/20 isolates, 46.8-122.6) and zearalenone (17/20 isolates, 44.6-124.2) were produced by the previous Fusarium species (10, 5 and 5 isolates), respectively. Roridin E (8.2-38.6 μg g-1 corn) was produced as a macrocyclic trichothecene by four toxic isolates of Myrothecium roridum and unidentified toxic factor was produced by four toxic isolates of Acremonium strictum. Also, the effect of sodium chloride on mycotoxins synthesis was studied. This is the first report on mycotoxins and mycoflora contamination of Libyan corn.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/rjt.2009.8.22
URL: https://scialert.net/abstract/?doi=rjt.2009.8.22
INTRODUCTION
Maize (Zea mays L.) is one of three major cereal crops that dominate world agriculture. Maize kernels are processed primarily for livestock feed (78%) and to some extent for human consumption (13%) (Igawa et al., 2007). Maize grain is a good substrate for mold infection and production of dangerous mycotoxins with potentially hazardous to the health of both humans and animals (WHO, 2006).
Mycotoxins are toxic secondary metabolites produced by many filamentous fungi and contaminated various agricultural commodities in pre-harvest, harvest, post-harvest and in storage conditions (Kumar et al., 2008). Aflatoxins are one of the most dangerous food-borne mycotoxins in Africa and epidemiological studies of human populations exposed to diets naturally contaminated with aflatoxins revealed an association between the high incidence of liver cancer in Africa and dietary intake of aflatoxins (MERCK, 2006). Aflatoxins produced by toxigenic strains of Aspergillus flavus and A. parasiticus. Chemically, aflatoxins belong to the group of bifuranocoumarins, with aflatoxins B1, B2, G1 and G2 being the most toxic and the liver representing the main target organ. In addition to its hepatotoxic action, AFB1 is also highly mutagenic, carcinogenic and teratogenic to animals (Fellinger, 2006). With respect to man, the IARC classified AFB1 within class 1 of human carcinogens (IARC, 2002). Their recognition as potent carcinogens in human and some animals has made them the subject of government legislation as well as valuable tools in the study of cancer (WHO, 2006; Wagacha and Muthomi, 2008).
Trichothecenes are a related group of sesquiterpenoid metabolites characterized by tetracyclic 12, 13-epoxytrichothec-9-ene skeleton, associated predominantly with Fusarium species, but also produced by several other fungi including Myrothecium, Trichothecium, Trichoderma, Cephalosporium, Stachybotrys and Cylindrocarpon. Although more than 80 derivatives of trichothecenes characterized in the laboratory, only eleven (T-2 toxin, HT-2 toxin, AcHT-2 toxin, T-2 tetraol, diacetoxyscirpenol, triacetoxyscirpenol, nivalenol, deoxynivalenol, fusarin C, fusarenone X and neosolaniol) have been detected occurring naturally in feedstuffs, grains and seeds (Ueno, 1983; Schollenberger et al., 2006). These biologically active metabolites are strong irritants and have been associated with naturally occurring outbreaks of vomiting feed refusal and possibly gastric ulcers when consumed. Also, trichothecenes inhibit protein synthesis which followed by a secondary disruption of DNA and RNA synthesis. They affect the actively dividing cells and can decrease antibody levels, immunoglobulins and certain other humoral factors such as cytokines (Richard, 2007).
Zearalenone is a non-steroidal estrogenic mycotoxin produced by several Fusarium species, which are regular contaminants of cereal crops worldwide (Mitterbauer et al., 2003). Chemically, it is a phenolic resorcylic acid lactone, with structure [6- (10-hydroxyl-6-oxo-trans-1-undecenyl)-B-resorcylic acid lactone] and it has the empirical formula of C18H22O5 with a molecular weight of 318 (FDA, 1979). The name is derived partly from the generic name of the host plant infected by Fusarium (Zea) and partly from its chemistry (ral from recylic acid lactone, en = double bond at C-1-2 and one = ketone) as recorded by Urry et al. (1966). Zearalenol (α-Zearalenol) is also, estrogenic resorcylic acid lactone compound produced by Fusarium species and it is a hydroxylated derivative of ZEN due to ZEN reductases present in animal tissues (Weidenborner, 2001). Zearalenone has been implicated in numerous mycotoxicoses in farm animals, causing infertility and reproductive problems such as abortions, false heat, recycling, reabsorption of fetuses and mummies and vulval uterine prolapse (Chowdhury, 2005; Schollenberger et al., 2006). Doses of ZEN that are much greater than concentrations which have hormonal effect may have genotoxic and carcinogenic effects (Mitterbauer et al., 2003). In blood, zearalenone and its metabolite, zearalenol bind to human sex hormone-binding globulin to some extent (Eriksen and Alexander, 1998). It is found worldwide in a number of cereal crops such as maize, barley, oats, wheat, rice and sorghum (El-Maghraby, 1996; Kumar et al., 2008).
In Libya, no published studies exist on mycoflora and mycotoxins contamination of corn, although maize is a very basic for livestock feed and human consumption, with a 2002 production of about 1500MT and about 276,700 MT were imported (FAO, 2003). Therefore, the purpose of this research was to study mycotoxins, mycoflora and mycotoxin-producing fungi on Libyan corn.
MATERIALS AND METHODS
Collection of Corn Samples
One hundred and twenty samples of yellow corn (crops, 2006, 2007), 500 g, each were collected from eight Shabias in Libya namely; Banighazy, Messrata, Tripoli, El-Mergab, Al-Zawia, Gefara, El-Nekat El-Khams and Naloot. Each sample was put in a sterile polyethylene bag sealed and put in another one which was also sealed to minimize the loss of water-content, transferred to the mycological laboratory and kept in a cool place (3-5°C) till fungal cultivation, isolation, identification and mycotoxins assay.
Determination of Moisture Content of Corn Grains
Twenty grams of each grain sample were milled and dried in an oven at 105°C for 24 h, then cooled in a desiccator and re-weighted to a constant weight. The moisture content was calculated as percentage of the dry weight.
Sample Preparation for Mycotoxins Analysis
Extraction Procedures
Fifty grams of corn from each sample were weighted in a 500 mL Erlenmeyer flask, ethyl acetate (150 mL) was added and the contents were combined shaken on a rotatory shaker (200 r.p.m, 24 h) and filtered through Whatman No. 1 filter paper. The residue was then washed twice with ethyl acetate (50 mL, each). The ethyl acetate extracts were combined, dried over anhydrous sodium sulphate, filtered and then concentrated under vacuum to near dryness, transferred into a brown glass vial and evaporated under nitrogen stream.
Purification of Crude Extracts
For cleaning up the crude extracts; it was suspended in 1 mL chloroform and applied to 14x0.8 cm column containing 2.5 g kiesel gel 60, 70/230 silica gel (MERCK) for aflatoxins, ochratoxins, sterigmatocystin, citrinin and zearalenone. The washing and eluting solvents (8 mL, each) were carried out according to AOAC (1984). While for trichothecenes, purification was performed according to Jarvis et al. (1986).
Bioassay of Mycotoxins
Brine shrimps (Artemia salina L.) larvae were used for mycotoxins bioassay test according to Korpinen (1974).
Thin Layer Chromatography (TLC)
For qualitative detection of mycotoxins, thin layer chromatography technique was employed using precoated silica gel plates type 60 F254 TLC (E, Merck, Germany). Aflatoxins B1, B2, G1 and G2, ochratoxins A and B, sterigmatocystin, citrinin, zearalenone (ZEN), diacetoxyscirpenol (DAS), T-2 toxin, HT-2 toxin, satratoxins F, G and H, verrucarins A and J, trichodermin and roridin A and E were applied as standard references. The developing solvent system was methanol-chloroform (v/v, 3:97) and the developed plates were viewed under short and/or long wave length UV (254 and/or 366 nm) irradiation and spread with reagents for mycotoxins qualitatively detection based on fluorescence intensity and/or color intensity according to Takitani et al. (1979), AOAC (1984), Jarvis et al. (1986) and Dorner (1998).
Enzyme Linked Immuno-Sorbent Assay (ELISA)
For quantitative determination of aflatoxins B1, B2, G1 and G2 and zearalenone, ELISA technique was employed according to Rodriguez et al. (2003) as accurate method in mycotoxin detection. The quantitative determination of aflatoxins and zearalenone was carried out compared of standard mycotoxins (Sigma).
High Performance Liquid Chromatography (HPLC)
HPLC analysis was performed for quantitative determination of trichothecenes (DAS, T-2 toxin, trichodermin and roridin E) using a Cecil 1000 gradient liquid chromatography equipped with a Cecil homochrome variable wave-length detector set at 260 nm, with the following gradient system: Solvent A, 30% dichloromethane in hexane; solvent B, 50% 2-propanol in dichloromethane. A flow rate of 1.2 min with an increasing gradient A of 5 to 50% solvent B was used over a period of 20 min (Jarvis et al., 1986). A 5 μm amino spherisorb column (250x4.6 mm) was employed. Each extract was analyzed twice by injection of 10 and 20 μL of extract. The chromatograms observed were compared to those of standard toxins (Sigma).
Determination of Fungi
The dilution plate method as described by Johnson and Curl (1972) was used for isolation corn fungi. Starch-Czapek`s agar medium (g L-1; Sodium nitrate 3.0, magnesium sulphate 0.5, potassium chloride 0.5, di-potassium hydrogen phosphate 1.0, iron sulphate 0.01, starch 21.9, agar agar 15.0- 20.0, pH 7.3±0.1) was used as cultivation and isolation medium. Chloramphenicol (0.5 mg mL-1) as bacteriostatic agent and rosebengal (30 ppm) to restrict for widespreading fungi because of stimulate slow growing fungi were added to the medium (Al-Doory, 1980; Martin, 1950). Five plates were used for each sample, incubated at 28±2°C for 7-15 days and the developing fungi were identified, counted and calculated per gram dry weight of each tested sample. The colonies of slow growing fungi as well as mycelial bits were transferred into slants with special media to ensure precise counting, then to plate for identification.
Taxonomic identification of fungi (based on purely morphologically macro- and microscopic characteristics) was carried out according to Booth (1971, 1977), Raper and Fennell (1977), Pitt (1979, 1991), Moubasher (1993), Samson et al. (2002) and Summerell et al. (2003).
Fungal Isolates, Subculture and Inoculation for Mycotoxins Production
Aspergillus flavus Link (CMI 102135), aflatoxins (B1, B2, G1 and G2) producer kindly provided by Prof. Dr. I.A. El-Kady, Botany Department, Faculty of Science, Assiut University, Egypt and Fusarium tricinctum (Corda) Sacc. (Z 90-410), a diacetoxyscirpenol (DAS) and zearalenone (ZEN) producer kindly provided by Prof. Dr. O.M.O. El-Maghraby, Dean of Faculty of Science, Sohag University, Egypt were used as standard mycotoxin-producers in study the minimizing effect of NaCl on different mycotoxin-production.
A total of 78 isolates of Aspergillus flavus Link, A. parasiticus Speare, A. flavo-furcatis Batista and Maia, A. flavus var. columnaris Raper and Fennell and A. zonatus Kwon and Fennell were isolated from corn samples contaminated by aflatoxins, while a total of 38 isolates of Fusarium tricinctum (Corda) Sacc., F. oxysporum Schlecht ex Fr., F. equiseti (Corda) Sacc. ex Fr., Acremonium strictum W. Gams, A. rutilum W. Gams, A. butyri (van Beyma) W. Gams, A. furcatum (F. and V. Moreau) ex W. Gams, Myrothecium roridum Tode ex Steudel and Trichoderma viride Pers. ex S.F. Gray were isolated from corn samples contaminated by microcyclic trichothecenes. The previous fungi were maintained on potato-dextrose agar (PDA) medium in order to obtain a mass of conidia after 7-10 days of incubation period. Each isolate (106-107 spores mL-1 suspension, 3 replicates) was grown on autoclaved Libyan corn (100 g, 60% M.C.) in a 500 mL Erlenmeyer flask.
For studying the role of NaCl as mycotoxin reducing agent on the production of aflatoxins, DAS and Zen, a saturated solution of NaCl (1 g 25 mL-1) was used to elevate the concentration of NaCl. After the desired incubation period, 50 g of artificial infected corn was extracted by ethyl acetate, filtered, concentrated, purified and the mycotoxins were determined qualitatively and quantitatively as described previously.
RESULTS AND DISCUSSION
Natural Occurrence of Mycotoxins on Corn
Of the extracts of 120 corn samples tested using brine shrimp larvae, 11 samples were toxic (>50% larvae mortality) and the other 109 samples were proved to be free of detectable amounts of toxic metabolites analyzed. Based on TLC, HPLC and ELISA analyses, aflatoxins B1 and B2 (2 samples, 9.1-13.9 μg kg-1 corn), aflatoxins B1, B2, G1 and G2 (4 samples, 7.6-16.4), T-2 toxin (3 samples, 8.4-10.2), diactoxyscirpenol (5 samples, 6.2-9.5) and zearalenone (5 samples, 6.8-7.9) were detected as shown in Table 1 and Fig. 1. A. flavus and A. parasiticus are widely distributed in nature on many types of organic material including forages, cereal grains, food and feed products and decaying vegetation in cultivated soil. Also, about 50% of A. flavus isolates from corn, soil and peanut produced large sclerotia, which allow it to survive in the soil for extended periods of time as mycotoxigenic fungus (Abbas et al., 2005). The fungus was considered a problem on corn in storage and in field causing infection in favorable conditions which leads to aflatoxin production (Lewis et al., 2005).
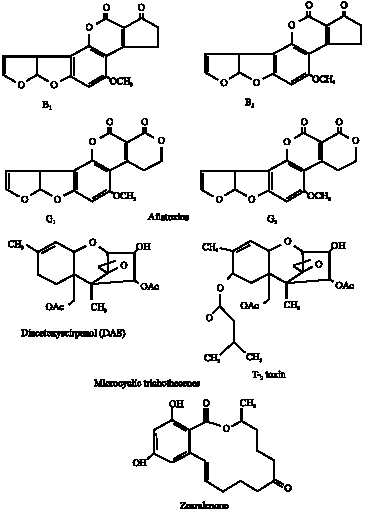 | |
| Fig. 1: | Chemical structure of different mycotoxins detected in corn |
| Table 1: | Detection of natural occurrence of mycotoxins on yellow corn samples using bioassay, TLC, HPLC and ELISA techniques |
 | |
| *Brine shrimp assay: H = High toxicity, greater than 75% mortality of brine shrimp larvae, M = Moderate toxicity, between 50-75% mortality of brine shrimp larvae **Mycotoxins detected (μg kg-1 corn): DAS = Diacetoxyscirpenol, ZEN = Zearalenone | |
Analysis of corn for natural occurrence of mycotoxins has been conducted in many areas of the world and aflatoxins are usually the dominant mycotoxins reported by Lewis et al. (2005), WHO (2006) and Kumar et al. (2008). In Africa, aflatoxins have been estimated to be widespread in major dietary staples and occur mostly in maize and groundnuts (Wagacha and Muthomi, 2008) and acute aflatoxin poisoning has severally occurred in Eastern and Central provinces of Kenya (Lewis et al., 2005). Chronic exposure to aflatoxins is associated with impaired immunity, malnutrition and liver cancer which is the third most common cause of death from cancer in Africa (Williams et al., 2004). Also, Shotwell et al. (1974) stated that in several affected crops, yellow-green masses of conidia of A. flavus and A. parasiticus may be visible at sites of kernel damage or along insect feeding paths and individual kernels of corn may contain as high as 400,000 μg kg-1 aflatoxins, as well as the higher moisture content of the grain at the time of storage increased Aspergillus infection and aflatoxin contamination. Aflatoxins are the most potent carcinogens in animal and human populations and interfere with the functioning of immune system (Hsieh, 1988).
Natural occurrence of different trichothecenes and zearalenone on corn and other cereal grains such as rice, sorghum, oats, barley and wheat have been recorded worldwide (Weidenborner, 2001; Schollenberger et al., 2006; Kumar et al., 2008). In Egypt, Abdel-Hafez et al. (1987) recorded that 9/64 samples of paddy grains were contaminated by T-2 toxin and DAS. Survey of natural occurrence of trichothecenes mycotoxins in corn grains revealed that 8/60 samples were contaminated by T-2 toxin, DAS and fusarenon C (El-Maghraby et al., 1995), while, 12/242 rice samples were contaminated by aflatoxins, T-2 toxin, DAS, zearalenone and sterigmatocystin (El-Maghraby, 1996). In India, on sorghum samples, aflatoxins, T-2, zearalenone, fumonisin B1 and B2 as well as aflatoxin B1 and T-2 toxin were recorded by Rukmini and Bhat (1978) and Sashidhar et al. (1991), respectively. In Poland, Perkowski and Basinski (2002) screened 99 oat grain samples for mycotoxin naturally contaminated and reported that HT-2 toxin was the major toxin with an incidence of 24% and its average level in positive samples was 21 μg kg-1, while the incidence of T-2 toxin and DAS was 15 and 12% with average levels 60 and 23 μg kg-1, respectively. Zearalenone is often found in corn, however, it is found also in other important crops such as wheat, barley, sorghum and rye through various countries of the world (Council for Agricultural Science and Technology, 2003). Mycotoxin regulations have been established in about 100 countries, out of which 15 are African, to protect the consumer from the harmful effects of mycotoxins (Fellinger, 2006).
Mycoflora Isolated from Aflatoxins-Contaminated Corn Samples
Twenty-six fungal species and three varieties appertaining to 13 genera were isolated on starch-Czapek`s agar medium at 28°C from 6 corn samples contaminated by aflatoxins (Table 2). The total counts of fungi fluctuated between 640-4750 colonies g-1 corn dry weight sample and the moisture content of these samples ranged between 14.2-15.8%.
Aspergillus was the most dominant genus isolated from all contaminated samples (100%), accounting 60.01% of total fungal count. It was represented by 11 species in addition to 3 varieties, of which, A. flavus Link (100% of the samples, 47.5% of Aspergillus count and 28.5% of total fungal count), A. niger van Tieghem (100, 9.5 and 5.7%), A. parasiticus Speare (80, 11.0 and 6.6%) and A. flavo-furcatis Batista and Maia (80, 4.2 and 2.5%) were the most prevalent (Table 2). A. flavus, A. parasiticus and different members of A. flavus group were the most contaminants of corn in different parts of the world and many of their isolates are aflatoxin-producers (Abbas et al., 2005; WHO, 2006; Richard, 2007; Kumar et al., 2008).
Also, Penicillium (4 species, 80% of the samples and 17.1% of total fungal count), Acremonium (one species, 80 and 10.3%), Fusarium (one species, 40 and 6.2%), Rhizopus (one species, 40 and 1.3%), Mucor (one species, 20 and 1.8%), Circinella (one species, 20 and 0.7%), Thamnidium (one species, 20 and 0.7%), Myrothecium (one species, 20 and 0.4%), Talaromyces (one species, 20 and 0.4%), Phoma (one species, 20 and 0.4%), Ulocladium (one species, 20 and 0.4%) and Setosphaeria (one species, 20 and 0.15%) were isolated from aflatoxins-contaminated samples (Table 2). Most of the previous fungi have been recovered from corn and other cereals worldwide (El-Maghraby et al., 1995; Kumar et al., 2008).
| Table 2: | Total count (TC), number of cases of isolation (NCI) of fungal genera and species isolated from 6 toxic corn samples contaminated by aflatoxins, No. of toxigenic isolates of A. flavi members (No. TI), No. of isolation sample (No. IS), brine shrimp assay and types of aflatoxins produced |
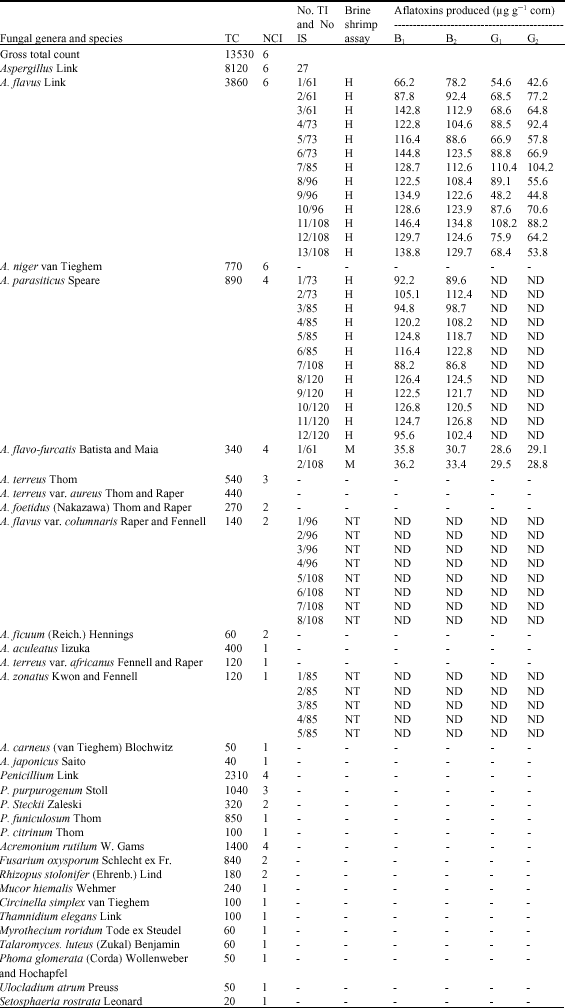 | |
| *Brine shrimp assay: H = High toxicity, greater than 75% mortality of brine shrimp larvae. M= Moderate toxicity, between 50-75% mortality of brine shrimp larvae. NT = No toxicity. ND = No detected toxin. - = Isolates not tested, other than A. flavi section | |
Aflatoxin-Producing Fungi Isolated from Contaminated Samples
Seventy-eight isolates of A. flavus (29), A. parasiticus (18), A. flavo-furcatis (18), A. flavus var. columnaris (8) and A. zonatus (5) were grown on corn (60% MC) at 20°C for 15-16 days and screened for aflatoxins production. Using brine shrimp assay and chemical analyses (TLC and ELISA), Aflatoxins B1 and B2 were produced by 12/18 isolates of A. parasiticus, 88.2-126.8 μg g-1 corn, while aflatoxins B1, B2, G1 and G2 (13/29 isolates of A. flavus, 48.2-146.4) in addition to 2/18 isolates of A. flavo-furcatis, 28.6 and 36.2. Several reports on aflatoxin production by toxigenic isolates of A. flavus and A. parasiticus worldwide (Abbas et al., 2005; Richard, 2007; Youssef, 2008). This is the first report on aflatoxin production by A. flavo-furcatis isolates. The ethyl acetate extracts of other thirteen isolates of A. flavus var. columnaris and A. zonatus proved to be non toxic to brine shrimp larvae and could not produce any detectable mycotoxin.
These results are in harmony with those obtained by El-Kady and Youssef (1993) who screened 130 isolates of three species and one variety of A. flavus group isolated from soybean seeds in Egypt and reported that 30/111 isolates of A. flavus produced aflatoxins B1, B2, G1 and G2 (143-540 μg 50 mL medium), 5/5 isolates of A. parasiticus produced aflatoxins B1 and B2 (136-297 μg 50 mL medium), while 12 isolates of A. tamarii and 2 isolates of A. flavus var. columnaris could not produce any detectable toxin.
Mycoflora Isolated from Trichothecenes-Contaminated Samples
Twenty-seven fungal species and two varieties belonging to 11 genera were isolated on starch-Czapek`s agar medium at 28°C from 5 corn samples contaminated by microcyclic trichothecenes (Table 3). The total count of fungi varied between 3620 to 4880 fungal colonies g-1 corn dry weight sample and the moisture content of these samples ranged between 14.6-17.1%.
Fusarium was the predominant genus isolated from all samples tested, contributing 48.7% of total fungi. Three species, namely; F. tricinctum (Corda) Sacc, F. oxysporum Schlecht ex Fr. and F. equiseti (Corda) Sacc. ex Fr.(single spore isolation) contributing 100% of the samples, each, 61.1, 26.4 and 12.4% of total Fusarium and 29.8, 12.9 and 6.1% of total fungi, respectively were isolated and identified. Fusarium species (F. graminearum, F. culmorum, F. equiseti, F. tricinctum, F. crookwellense, F. verticillioides, F. proliferatum, F. avenaceum, F. camptoceras. F. semitecum, F. oxysporum and F. toxicoses) are regular contaminants of different cereal crops worldwide (El-Maghraby, 1996; Schollenberger et al., 2006; Igawa et al., 2007). The presence of Fusarium in feeds is correlated with leukoencephalamalacia in horses, esophageal cancer in humans and experimentally induced hepatocarcinoma in ducks and mice (Ueno, 1983; Thiel et al., 1986).
| Table 3: | Total count (TC), number of cases of isolation (NCI) of fungal genera and species isolated from 5 toxic corn samples contaminated by microcyclic trichothecenes (T-2 and DAS) and zearalenone, No. of toxigenic isolates (No. TI), No. of isolation sample (No. IS), brine shrimp assay and mycotoxins produced |
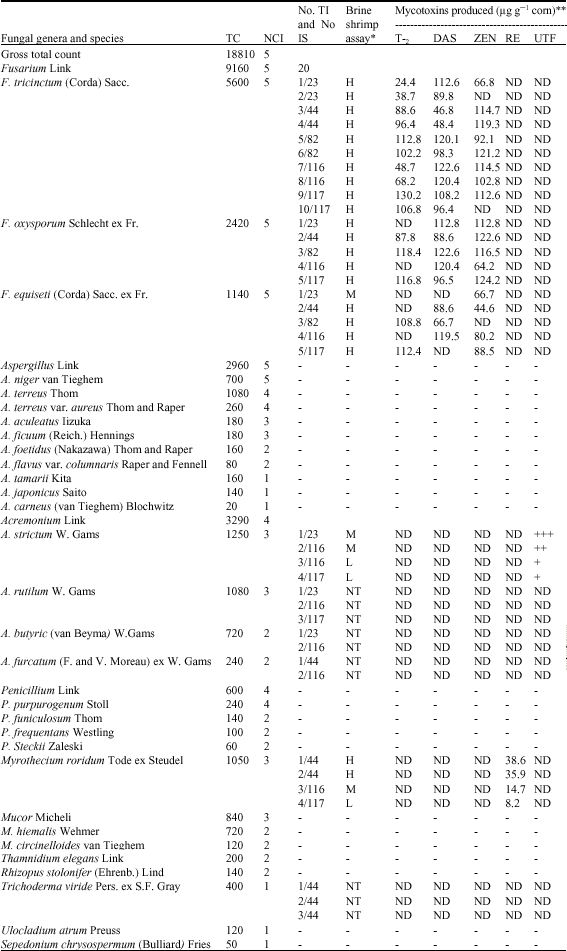 | |
| *Brine shrimp assay: H = High toxicity, greater than 75% mortality of brine shrimp larvae. M = Moderate toxicity, between 50-75% mortality of brine shrimp larvae, L = Low toxicity, between 25-50% mortality of brine shrimp larvae. NT = No toxicity. ND = No detected toxin. **Mycotoxins produced: T-2: T-2 Toxin, DAS: Diacetoxyscirpenol, ZEN: Zearalenone, RE: Roridin E, UTF: Unidentified toxic factor. +, ++ and +++: weak, moderate and high concentrations, respectively. - = Isolates not tested, other than Fusarium, Acremonium, Myrothecium and Trichoderma species | |
Aspergillus (8 species and 2 varieties, 100% of the samples and 15.7% of total fungi), Acremonium (4 species, 80 and 17.5%), Penicillium (4 species, 80 and 3.2%), Myrothecium (one species, 60 and 5.3%), Mucor (2 species, 60 and 3.9%), Thamnidium (one species, 40 and 1.1%), Rhizopus (one species, 40 and 0.7%), Trichoderma (one species, 20 and 2.7%), Ulocladium (one species, 20 and 0.6%) and Sepedonium (one species, 20 and 0.5%) were isolated also from contaminated corn samples (Table 3). Most of these genera and species have been recovered from corn and other cereals in various parts of the world (Abdel-Hafez et al., 1987; El-Maghraby et al., 1995; El-Maghraby, 1996; Kumar et al., 2008). It is noteworthy that many species isolated from trichothecenes contaminated samples are mycotoxin-producers (Ueno, 1983; Richard, 2007; Youssef, 2008).
Mycotoxigenic Fungi Isolated from Trichothecenes-Contaminated Samples
Thirty-eight isolates of Fusarium tricinctum (10), F. oxysporum (5), F. equiseti (5), Acremonium strictum (4), A. rutilum (3), A. butyri (2), A. furcatum (2), Myrothecium roridum (4) and Trichoderma viride (3) were grown on corn (60% MC, 20°C) and screened for mycotoxin production. Based on brine shrimp assay and chemical analyses (TLC, HPLC and ELISA), all isolates of Fusarium tricinctum, F. oxysporum and F. equiseti proved to be toxic and produced two trichothecenes; T-2 toxin (15/20 isolates, 24.4-130.2 μg g-1 corn) and DAS (18/20 isolates, 46.8-122.6 μg g-1) in addition to zearalenone (17/20 isolates, 44.6-124.2 μg g-1). F. tricinctum, F. oxysporum and F. equiseti were reported to produce several microcyclic trichothecenes (Ueno, 1983; El-Maghraby, 1996; Richard, 2007; Kumar et al., 2008). Trichothecenes inhibit protein synthesis, DNA and RNA in eukaryotic cells and are immunotoxic in rats and mice (Richard, 2007).
Eight isolates of Myrothecium roridum and Acremonium strictum (4 isolates, each) were proved to be toxic to brine shrimp larvae. Based on TLC and HPLC, roridin E (8.2-38.6 μg g-1 corn) could be detected in ethyl acetate extracts of different isolates of Myrothecium roridum, while an unidentified toxic factor was detected in extracts of Acremonium strictum as recorded in Table 3 and Fig. 2. Roridin E, a macrocyclic trichothecene and highly cytotoxic compound was identified previously in Myrothecium, Cylindrocarpon and Stachybotrys isolates (Ueno, 1983; Jarvis et al., 1986, El-Maghraby, 1996; Kumar et al., 2008). The extracts of other ten isolates of A. rutilum (3), A. butyri (2), A. furcatum (2) and Trichoderma viride (3) proved to be non toxic to brine shrimp larvae and could not mycotoxin detected.
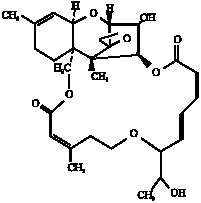 | |
| Fig. 2: | Structure of roridin E produced by Myrothecium roridum |
Control Effect of Sodium Chloride on Production of Aflatoxins, DAS and ZEN
The effect of different concentrations of sodium chloride (0.25-7.00%) added to the corn grains (60% MC) on production of aflatoxins by Aspergillus flavus (CMI 102135) and diacetoxyscirpenol (DAS) combined with zearalenone (ZEN) by Fusarium tricinctum (Z 90-410) was recorded in Table 4. At very low level of NaCl (0.25%), there was stimulatory effect and the biosynthesis of different toxins was relatively high in comparison of the control (aflatoxins, 589.2 and the control, 586.6 μg g-1 corn), (DAS, 284.6 and the control, 282.4 μg g-1 corn) and (ZEN, 263.1 and the control, 260.8 μg g-1 corn). At concentrations (0.5-1.5% of NaCl) slightly decreasing in production of each of aflatoxins (548.3-422.2 μg g-1 corn), DAS (272.1-204.8 μg g-1 corn) and ZEN (245.2-193.1 μg g-1corn) followed by sharply depression at concentrations (1.75-3% of NaCl), in case of aflatoxins (387.6-34.9 μg g-1corn) as well as in DAS (194.6-19.7 μg g-1corn) and ZEN (175.2-10.4 μg g-1 corn). At high concentration (3.5% NaCl), no DAS and ZEN detected, while aflatoxins did not detect at 3.75% NaCl, which emphasizes the effective role of sodium chloride in inhibition of mycotoxin synthesis.
| Table 4: | Production of aflatoxins by Aspergillus flavus (CMI 102135) and diacetoxyscirpenol combined with zearalenone by Fusarium tricinctum (Z 90-410) on autoclaved corn (60%MC) with adding different concentrations of sodium chloride after 14-16 days of incubation period at 20°C |
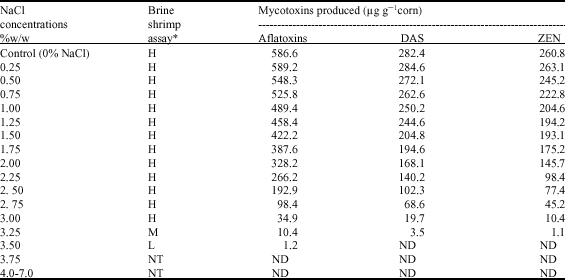 | |
| *Brine shrimp assay: H = High toxicity, greater than 75% mortality of brine shrimp larvae. M = Moderate toxicity, between 50-75% mortality of brine shrimp larvae, L = Low toxicity, between 25-50% mortality of brine shrimp larvae. NT = No toxicity, ND = No detected toxin | |
These results are in harmony with those recorded by Mashaly and El-Deep (1982) and El-Kady et al. (1991) on aflatoxin production by toxigenic aspergilli in presence of NaCl. Very rare investigations have been carried out on the production of diacetoxyscirpenol (DAS) and zearalenone (ZEN) in presence of NaCl (El-Maghraby, 1996). Generally, low concentrations of NaCl have a stimulatory effect on enzyme activity (Mashaly and El-Deep, 1982) and mycotoxin production, whereas high content inhibited synthesis of fungal toxins (El-Kady et al., 1991, El-Maghraby, 1996). In this respect, El-Kady et al. (1991) stated that Zn, Fe, Mg and Ca increased aflatoxin production at low concentrations (2-50 mg g-1), whereas Cu, Ni and Cd depressed the production.
In conclusion, it is the first report on mycotoxins and mycoflora contamination of Libyan corn and it is clearly evident that maize grain is a good substrate for mold infection and production of dangerous mycotoxins; aflatoxins, trichothecenes and zearalenone with potentially hazardous to the health of both humans and animals (WHO, 2006; Richard, 2007; Kumar et al., 2008). Also, sodium chloride played an important role in inhibition and prevention of mycotoxins synthesis. So, for human public health, grains in different stages of production chain from the field to the final consumer must be subjected to quality control and good testing protocol for molds and mycotoxins contamination to ensure a food supply free of toxic levels of mycotoxins.
ACKNOWLEDGMENTS
Thanks are due to Prof. Dr. I.A. El-Kady, Botany Department, Faculty of Science, Assiut University, Egypt and Prof. Dr. O.M.O. El-Maghraby, Dean of Faculty of Science, Sohag University, Egypt, for their valuable helps and kindly providing me standard mycotoxin-producing fungal isolates.
REFERENCES
- Abbas, H.K., M.A. Weaver, R.M. Zablotowicz, B.W. Horn and W.T. Shier, 2005. Relationships between aflatoxin production and sclerotia formation among isolates of Aspergillus section Flavi from Mississippi Delta. Eur. J. Plant Pathol., 112: 283-287.
CrossRefDirect Link - Abdel-Hafez, S.I.I., I.A. El-Kady, M.B. Mazen and O.M.O. El-Maghraby, 1987. Mycoflora and trichothecene-toxins of paddy grains from Egypt. Mycopathologia, 100: 103-112.
CrossRefDirect Link - AOAC., 1981. Official Methods of Analysis. 13th Edn., Association of Official Analytical Chemists, Rockville, Maryland, ISBN: 0-935584-14-5, Pages: 1038.
CrossRefDirect Link - El-Kady, I.A. and M.S. Youssef, 1993. Survey of mycoflora and mycotoxins in Egyptian soybean seeds. J. Basic Microbiol., 33: 371-378.
Direct Link - El-Maghraby, O.M.O., I.A. El-Kady and S.A. Soliman, 1995. Mycoflora and Fusarium toxins of three types of corn grains in Egypt with special reference to production of trichothecene-toxins. Microbiol. Res., 150: 225-232.
Direct Link - El-Maghraby, O.M.O., 1996. Mycotoxins and mycoflora of rice in Egypt with special reference to trichothecenes production and control. J. Nat. Toxins, 5: 49-59.
Direct Link - Jarvis, B.B., Y.W. Lee, S.N. Comezoglu and C.S. Yatawara, 1986. Trichothecenes produced by Stachybotrys atra from Eastern Europe. Applied Environ. Microbiol., 51: 915-918.
Direct Link - Korpinen, E.L., 1974. Studies on Stachybotrys alternans: I- Comparison of rabbit skin, mouse fibroblast culture and brine shrimp tests as detectors of Stachybotrys toxins. Acta Pathol. Microbiol. Scan. Sect. Bul., 82: 465-469.
PubMedDirect Link - Kumar, V., M.S. Basu and T.P. Rajendran, 2008. Mycotoxin research and mycoflora in some commercially important agricultural commodities. Crop Protect., 27: 891-905.
CrossRefDirect Link - Lewis, L., M. Onsongon, H. Njapau, H. Schurz-Rogers and G. Luber et al., 2005. Aflatoxin contamination of commercial maize products during an outbreak of acute aflatoxicosis in Eastern and Central Kenya. Environ. Health Perspect., 113: 1763-1767.
CrossRefDirect Link - Martin, J.P., 1950. Use of acid, rosebengal and streptomycin in plate method for estimation of soil fungi. Soil Sci., 69: 215-233.
Direct Link - Mitterbauer, R., H. Weindrofer, N. Safaie, R. Krska and M. Lemmens et al., 2003. A sensitive and inexpensive yeast bioassay for the mycotoxin zearalenone and other compounds with estrogenic activity. Applied Environ. Microbiol., 69: 805-811.
CrossRefDirect Link - Perkowski, J. and T. Basinski, 2002. Natural contamination of oat with group A trichothecene mycotoxins in Poland. J. Food Addit. Contam., 19: 478-482.
CrossRefDirect Link - Richard, J.L., 2007. Some major mycotoxins and their mycotoxicosis: An overview. Int. J. Food Microbiol., 119: 3-10.
CrossRefDirect Link - Rodriguez, V.M.L., D.M.M. Calonge and E.D. Ordonez, 2003. ELISA and HPLC determination of the occurrence of aflatoxin M1 in raw cow's milk. J. Food Addit. Contam., 20: 276-280.
Direct Link - Rukmini, C. and R.V. Bhat, 1978. Occurrence of T-2 toxin in Fusarium infested sorghum from India. J. Agric. Food Chem., 26: 647-649.
CrossRefDirect Link - Sashidhar, R.B., Y. Ramakrishna, Y., T. Ramnath and R.V. Bhat, 1991. Lack of relationship between sorghum consumption, mycotoxin contamination and pellagra in a traditionally sorghum eating population. Trop. Geogr. Med., 43: 165-170.
PubMedDirect Link - Schollenberger, M., H.M. Muller, M. Rufle, S. Suchy, S. Plank and W. Drochner, 2006. Natural occurrence of 16 Fusarium toxins in grains and feedstuffs of plant origin from Germany. Mycopathologia, 161: 43-52.
CrossRefDirect Link - Shotwell, O.L., M.L. Goulden and C.W. Hesseltine, 1974. Aflatoxin: Distribution in contaminated corn. Cereal Chem., 51: 492-498.
Direct Link - Summerell, B.A., B. Salleh and J.F. Leslie, 2003. A utilitarian approach to Fusarium identification. Am. Phytopathol. Soc. Plant Dis., 87: 117-128.
CrossRefDirect Link - Takitani, S., Y. Asaba, T. Kato, M. Suzuki and Y. Ueno, 1979. Spectrodensitometric determination of trichothecene mycotoxins with 4-(P-nitrobenzyl) pyridine on silica gel thin layer chromatograms. J. Chromatogr., 172: 335-342.
PubMedDirect Link - Thiel, P.G., W.C.A. Geldeblom, W.F.O. Marasas, P.E. Nelson and T.M. Wilson, 1986. Natural occurrence of moniliformin and fusarin C in corn screenings known to be hepatocarcinogenic in rats. J. Agric. Food. Chem., 34: 773-775.
Direct Link - Urry, W.H., H.L. Wehrmeister, E.B. Bodge and P.H. Hidy, 1966. The structure of zearalenone. Tetrahedron Lett., 27: 3109-3114.
CrossRefDirect Link - Wagacha, J.M. and J.W. Muthomi, 2008. Mycotoxin problem in Africa: Current status, implications to food safety and health and possible management strategies. Int. J. Food Microbiol., 124: 1-12.
CrossRefDirect Link - Williams, J.H., T.D. Phillips, P.E. Jolly, J.K. Stiles, C.M. Jolly and D. Aggarwal, 2004. Human aflatoxicosis in developing countries: A review of toxicology, exposure, potential health consequences and interventions. Am. J. Clin. Nutr., 80: 1106-1122.
PubMedDirect Link - Youssef, M.S., 2008. Studies on mycological status of sundried jew's mallow leaves and okra fruits in Egypt. Res. J. Microbiol., 3: 375-385.
CrossRefDirect Link








