Research Article
Detection of Reactive Oxygen Species Can Be Used to Distinguish ToxA-induced Cell Death from the Hypersensitive Response
USDA-ARS, Cereal Crops Research Unit, Northern Crop Science Lab, 1307 18th Street North, Fargo, ND 58105
The necrotrophic fungus Pyrenophora tritici-repentis (Died.) Drechs, a world-wide threat in wheat growing areas, causes tan spot disease in bread wheat (Triticum aestivum L.) and durum wheat (T. turgidum L. var. durum). Typical disease symptoms consist of tan-colored, diamond-shaped necrotic lesions surrounding small, dark-brown infection sites (McMullen and Francl, 1993). The necrotic symptom is due to the production of a host-selective toxin (HST), ToxA (Tomas et al., 1990; Lamari et al., 1995), which is the first proteinaceous HST cloned from fungi (Ballance et al., 1996; Ciuffetti et al., 1997; Zhang et al., 1997). A single dominant gene Tsn1, which confers sensitivity to ToxA infiltration, was mapped to wheat chromosome 5BL (Faris et al., 1996; Stock et al., 1996; Lu et al., 2006). Other HSTs have been demonstrated to be toxin-receptor interactions, such as victorin produced by Cochliobolus victorae to oats (Avena sativa L.) (Wolpert and Macko, 1989) and Hmt toxin produced by C. heterostrophus race T to corn (Zea mays L.) (Braun et al., 1990). It was proposed that the ability of ToxA to be internalized into plant cells determines the host sensitivity to this toxin (Manning and Ciuffetti, 2005; Tai et al., unpublished data). The internal target of ToxA in wheat was identified recently (Tai et al., unpublished data). Here I demonstrate that the ToxA-induced cell death is different from the programmed cell death of the Hypersensitive Response (HR).
Genetic analyses have identified a variety of corresponding gene pairs, R genes in plants and Avr genes in the associated pathogens, which mediate plant resistance to pathogens (Staskawicz et al., 1995; Dangl and Jones, 2001; Hammond-Kosack and Parker, 2003; Martin et al., 2003; Innes, 2004; Chisholm et al., 2006; McDowell and Simon, 2006). Plant defenses are often associated with the HR (Yang et al., 1997). The early events in the interaction between plants and their incompatible pathogens include the elevation of cytosolic calcium levels (Jabs et al., 1997), an oxidative burst (Lamb and Dixon, 1997) and the production of nitric oxide (NO) (Delledonne et al., 1998; Durner et al., 1998).
Nitric oxide seems to play an important role in several plant physiological responses, including abscisic acid signaling (Garcia-Mata and Lamattina, 2003), wound signaling (Huang et al., 2004), regulation of iron-ferritin homeostasis (Murgia et al., 2002), root development (Correa-Aragunde et al., 2004) and programmed cell death in barley aleurone layers (Beligni et al., 2002). NO has been proposed to activate responses involving G proteins, cGMP and calcium (Clarke et al., 2000; Klessig et al., 2000). The involvement of NO in various plant-pathogen interactions has been summarized in a review article (Romero-Puertas et al., 2004). In gene-for-gene interactions, NO has been detected in RPM1-AvrRpm1/AvrB mediated resistance in Arabidopsis (Delledonne et al., 1998; Zeier et al., 2004) and resistance to TMV infection of tobacco plants expressing the N gene (Durner et al., 1998).
In addition to the production of NO, an oxidative burst was observed in the RPS2-AvrRpt2 interaction in Arabidopsis (Alvarez et al., 1998). NO and H2O2 function complementary to each other and act as secondary messengers during the HR and Systemic Acquired Resistance (SAR) (Alvarez et al., 1998; Chamnongpol et al., 1998; Delledonne et al., 1998; Durner et al., 1998). H2O2 is also likely to play an important role in cross-tolerance, in which exposure to one stress can induce tolerance to other stresses (Bowler and Fluhr, 2000). There appears to be a positive feedback loop involving H2O2, Salicylic Acid (SA) and HR (Alvarez, 2000).
Aerobic organisms generate various Reactive Oxidative Species (ROS), such as superoxide anion and hydrogen peroxide (Finkel and Holbrook, 2000; Moller, 2001). Both enzymatic and non-enzymatic pathways are potential sources of H2O2 in plants and NADPH oxidase is one of those sources (Keller et al., 1998). Although ROS are considered to be detrimental to cells, increasing evidence indicates that H2O2 functions as a signaling molecule in plants (Lamb and Dixon, 1997) and animals (Finkel and Holbrook, 2000) mediating programmed cell death in response to pathogens, elicitors and hormones (Pei et al., 2000; Bethke and Jones, 2001). H2O2 especially can induce secondary oxidative bursts, denoted as micro-HR, in distant tissues (Alvarez et al., 1998). H2O2 is a potent activator of Mitogen-Activated Protein (MAP) kinase cascades (Kovtun et al., 2000; Romeis et al., 1999), which may mediate plant defense signaling (Bent, 2001; Innes, 2001).
Tomato Pto and Prf are required for the signaling pathway leading to resistance against Pseudomonas syringae pv. tomato (Pst) strains that express the avirulence gene AvrPto (Salmeron et al., 1994, 1996). The specific resistance to Pst is determined by Pto, which encodes a protein kinase that recognizes AvrPto (Scofield et al., 1996; Tang et al., 1996). Expression of gain-of-function mutants of Pto in transient assays results in a rapid HR (Rathjen et al., 1999). Prf encodes a nucleotide binding site-leucine rich repeat protein that is required for Pto-mediated HR (Rathjen et al., 1999) and resistance (Tobias et al., 1999). Chandra et al. (1996) observed the Pto-mediated oxidative burst in tomato cell suspension cultures after the cells were challenged by Pst carrying AvrPto. The present study was conducted to investigate and demonstrate the early events (production of NO, O2- and H2O2) in Pto-AvrPto mediated HR and their dependency upon Prf and to compare the response to those of wheat exposed to ToxA.
Tomato and Nicotiana benthamiana plants, Agrobacterium infiltration and application of dexamethasone Plants used for transient assays in this study included tomato (Lycopersicon esculentum cv Rio Grande 76R and the Pst-susceptible mutant prf-3 derived from Rio Grande 76R; Salmeron et al., 1996) and Nicotiana benthamiana (wild type and transgenic plants). The transgenic N. benthamiana plants carried Pto expressed from the CaMV 35S promoter and AvrPto fused to a dexamethasone (DEX)-inducible promoter (Chang et al., 2002). To induce expression of AvrPto, 30 μM DEX was applied to the leaves of transgenic N. benthamiana (Pto, AvrPto) immediately prior to conducting assays. For transient assays in tomato plants and wild-type N. benthamiana, Agrobacterium tumefaciens strain C58C1, carrying empty vector pBILW (derived from pBI121; Klann et al., 1996) or with the insert of PtoL205D, a construct which induces Prf-dependent HR in cv. Rio Grande 76R tomato plants independent of AvrPto (Chang et al., 2002; Rathjen et al., 1999), was used for leaf infiltration, according to the method of agroinfiltration described in Chang et al. (2002). Twenty hours after agroinfiltration of tomato plants and 36 h after agroinfiltration of wild-type N. benthamiana, stems were cut for histochemical staining.
Wheat Plants and ToxA Infiltration
The ToxA-sensitive wheat cultivar Triticum turgidum L. subsp. durum cultivar Langdon (LDN). Wheat plants were kept in a growth chamber at 23°C with 10/14-h light/dark cycle for two to three weeks until the seedlings reached the two-leaf stage. Photos of cell death induced by ToxA infiltration in wheat leaves were taken at 2, 4, 8, 12 and 24 h time post-infiltration. The procedure of ToxA infiltration was described in Meinhardt et al. (2002). Ten μg mL-1 purified ToxA were infiltrated into the second leaf of 2- to 3-week old wheat seedlings.
Histochemical Staining for Nitric Oxide, Superoxide Anion and Hydrogen Peroxide
NO was detected according to the methods described by Pedroso et al. (2000). The severed ends of plant cuttings, excised at the bases of their stems, were set in a solution of 10 μM 4,5-diaminofluorescein diacetate (DAF-2 DA; Calbiochem, La Jolla, CA, USA) dissolved in 0.2% DMSO (Kojima et al., 1998) at room temperature. After 6 to 8 h of transpiration, sections of leaf slices were hand-cut and observed using DAF bright green fluorescence and confocal laser microscopy (Bio-Rad Microscience Division, Hercules, CA, USA).
Superoxide anion (O2.¯) was detected in situ with nitroblue tetrazolium (NBT; Calbiochem, La Jolla, CA, USA) using a modification of the technique of Venisse et al. (2001). Plant cuttings were fed a solution of 0.05% NBT at room temperature (as described above for DAF-2 DA). The reaction was terminated by immersing leaves in boiling 95% ethanol for 10 min. Boiling decolorized the leaves except for the dark blue produced by the reaction of NBT with superoxide anions. Leaves were preserved at room temperature in 95% ethanol.
Hydrogen peroxide was detected in leaves by staining with 3,3-diaminobenzidine (DAB; Sigma-Aldrich, St. Louis, MO, USA) as described by Thordal-Christensen et al. (1997) and Orozco-Cardenas and Ryan (1999). The procedures used for tomato and N. benthamiana plants were similar to those utilized for NBT staining except that the solution fed to the plant cuttings contained 1 mg mL-1 DAB (pH 3.8). One hundred μM of diphenyleneiodonium chloride was used to block the NADPH oxidase activity. Due to the different morphological structures of monocot and dicot leaves, the above method did not work successfully in wheat. For the detection of hydrogen peroxide in wheat leaves, ToxA was infiltrated for one hour and then DAB was infiltrated at the same area.
For all staining described above, at lease three leaves were tested in each experiment. Two repeats were performed for NO staining, three repeats for staining of superoxide anions, at least five repeats for detection of hydrogen peroxide in tomato and N. benthamiana and three repeats for staining of hydrogen peroxide in wheat.
Production of Nitric Oxide
To investigate whether the interaction between Pto and AvrPto resulted in the production of NO, the transgenic N. benthamiana plants which constitutively expressed Pto under the 35S promoter and contained the DEX-inducible AvrPto, were used. Nitric oxide, as identified by DAF fluorescence, was observed 4 to 6 h after the application of DEX (Fig. 1). The HR was visible about 8 h after treatment with DEX. The HR was also visible by agroinfiltration with PtoL205D about 2 to 3 days after treatment in N. benthamiana or about 1 to 2 days after agroinfiltration in tomato 76R. In contrast, no HR and no DAF fluorescence were observed in tomato prf-3 mutants infiltrated with PtoL205D. The production of NO induced by the Pto-AvrPto interaction, therefore, appears to require Prf.
Production of Superoxide Anions
In N. benthamiana leaves, it is possible to observe the dark blue precipitation caused by NBT reacting with superoxide anions, even without removing chlorophyll (Fig. 2). In transgenic N. benthamiana (Pto, AvrPto) leaves, NBT staining was detected along the vascular bundles 4 to 6 h after the application of DEX, whereas local cell death was not macroscopically visible until about 8 h after DEX application. NBT was also readily detected in wild-type N. benthamiana leaves after Agroinfiltration with PtoL205D. However, dark blue staining was not evident in tomato leaves, whether the leaves subsequently exhibited the HR or not. Therefore, the prf-3 tomato mutants could not be utilized to investigate whether superoxide anion production is Prf-dependent.
Production of Hydrogen Peroxide
The production of H2O2 was detected using DAB staining. This staining was detected after 6 h in the DEX-inducible transgenic N. benthamiana (Pto, AvrPto) plants (Fig. 3a). The production of H2O2 was blocked by the treatment of diphenyleneiodonium chloride (DPI), an inhibitor of NADPH oxidase (Fig. 3b).
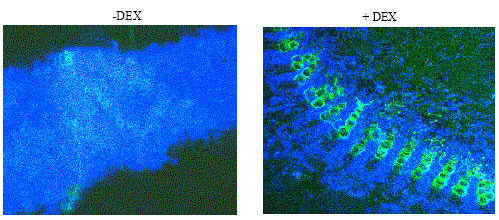 | |
| Fig. 1: | Nitric oxide detected by DAF. The Pto-AvrPto interaction induces the production of nitric oxide as detected by DAF staining in hand-cut cross sections of transgenic N. benthamiana plants after treatment with DEX to induce the Pto-AvrPto interaction. Without DEX (-DEX) the veins exhibited a slight green fluorescent background. The application of DEX (+DEX), which initiated the interaction between Pto and AvrPto, resulted in the production of No. The green fluorescence is predominantly visible in the veins |
 | |
| Fig. 2: | The production of superoxide anions as detected using NBT staining. The DEX-induced Pto-AvrPto interaction in the transgenic N. benthamiana plants, carrying 35S: Pto and DEX-inducible AvrPto, produced superoxide anion. (a) Without ethanol extraction to remove chlorophyll, the dark blue NBT staining clearly shows the production of superoxide anion precedes the HR. The time course is described in the text. (b) With the chlorophyll removed, NBT is predominantly visible in the right-hand side of the leaf which was treated with DEX |
 | |
| Fig. 3: | DAB staining of hydrogen peroxide in N. benthamiana. (a) Transgenic N. benthamiana (35S: Pto; DEX-inducible AvrPto) specifically initiates hydrogen peroxide production after the application of DEX (+DEX), as seen on the right-hand half of the leaf, compared to the left-hand half without DEX treatment. The DEX-induced production of hydrogen peroxide was inhibited by DPI (b). (c) Constitutively active PtoL205D can stimulate the production of hydrogen peroxide in transient overexpression assays with agroinfiltration, but it is inhibited by syringe-infiltration of DPI as shown in the overlapped area of infiltration having no DAB deposition |
The DAB staining was also detectable in wild-type N. benthamiana (Fig. 3c) and 76R tomato plants (Fig. 4), after agroinfiltration with PtoL205D, which mimicked the Pto-AvrPto interaction. However, infiltration with PtoL205D clearly did not cause the production of H2O2 in the tomato prf-3 mutant (Fig. 4). Therefore, Prf was required for the oxidative burst leading to HR.
 | |
| Fig. 4: | DAB staining of hydrogen peroxide in tomato. Agroinfiltration of PtoL205D leads to the production of hydrogen peroxide in 76R tomato but not in prf-3 mutant plants. Empty vector (pBILW) alone causes very little hydrogen peroxide production around the wound site of the infiltration |
 | |
| Fig. 5: | DAB staining in wheat leaves. Necrotic cell death was visible by 12 h post-infiltration of ToxA, but hydrogen peroxide was only detectable around the wound site as indicated by DAB staining. The DAB staining was clearly seen only after 24 h ToxA infiltration, when cell death was already obviously visible |
In ToxA-sensitive wheat, the ToxA-induced cell death was visible around 12 h post infiltration, which is consistent with the results of detectable electrolyte leakage in 2 h (Kwon et al., 1998). The production of hydrogen peroxide was only detectable after visible cell death was observed (Fig. 5). This infers that the source of hydrogen peroxide in the necrosis is different from the HR. The detected hydrogen peroxide after cell death might be released somehow from the necrotic cells.
The HR is usually associated with disease resistance. Localized cell death of the infected site appears to limit the spread of pathogens (Lam et al., 2001). The uninfected parts of the plants often develop Systemic Acquired Resistance (SAR), which leads to broad resistance against diverse pathogens (Ryals et al., 1996). Both the HR and SAR are regulated by a network of signal transduction, involving ROS, NO, Salicylic Acid (SA), Jasmonic Acid (JA) and ethylene (Alvarez et al., 1998; Bolwell, 1999; Alvarez, 2000; Wendehenne et al., 2001; Neill et al., 2002; Overmyer et al., 2003). Therefore, the production of ROS and NO is an indicator of disease resistance.
A plant NADPH oxidase homologous to mammalian gp91phox has been considered as a source for the apoplastic O2.- generation (Keller et al., 1998). gp91phox homologs, AtrbohD and AtrbohF in Arabidopsis (Torres et al., 2002) and NbrbohA and NbrbohB in N. benthamiana (Yoshioka et al., 2003), are required for accumulation of ROS in defense responses. NADPH oxidase was proposed to be the primary source for the accumulation of H2O2 converted from O2.¯ (Orozco-Cardenas et al., 2001; Dat et al., 2003). This suggests that H2O2 is produced by spontaneous or enzyme-catalyzed dismutation. Thus, the balance between the O2.¯ production rate and its dismutation rate determines the type of ROS that is accumulating.
NBT staining could easily be used to detect the presence of superoxide anions in N. benthamiana but not in tomato plants. Similarly, Levine et al. (1994) failed to detect superoxide anions in soybean subjected to elicitor, although accumulation of hydrogen peroxide was detected during their study. There is a correlation between H2O2 and HR in tobacco (Schraudner et al., 1998), tomato (Wohlgemuth et al., 2002) and birch (Pellinen et al., 2002), with no O2.¯ accumulation detectable. However, in Arabidopsis, O2.¯ was the ROS responsible for cell death (Jabs et al., 1996). The high enzyme activity of the superoxide dismutase in tomato plants might contribute to the undetectable levels of superoxide by NBT staining in this study.
Nitric oxide is another critical player apparently required for ROS-mediated cell death (Delledonne et al., 1998; Wendehenne et al., 2001; Wendehenne et al., 2004). Both enzymatic and non-enzymatic sources are possible for NO production. Nitrate reductase might be responsible for synthesizing NO in plants (Yamasaki and Sakihama, 2000). A hormone-activated NO-synthesizing enzyme was identified in Arabidopsis (Guo et al., 2003).
The SA, JA and ethylene are linked in a complex network of interactions (Devadas et al., 2002). It has been proposed that SA and ROS are part of a positive feedback loop responsible for amplifying signals leading to defense and cell death (Shirasu et al., 1997; Van Camp et al., 1998; Shah, 2003; Xiao et al., 2003). Vascular tissues are the major position of endogenous NO detected in response to the Pto-AvrPto interaction in this present study, which is consistent with the staining in pea plants (Corpas et al., 2004). In tomato, the accumulation pattern of NO (this study), H2O2 (Moeder et al., 2002; this study) and ethylene (Moeder et al., 2002) is preferentially localized to cells surrounding vascular bundles. ROS and micro-HR exhibit a similar spatial preference close to leaf veins, in systemic resistance in Arabidopsis (Alvarez et al., 1998).
The Pto-AvrPto interaction causes an oxidative burst and results in a HR (Chandra et al., 1996; Lamb and Dixon, 1997). The present study indicates that the oxidative burst is Prf dependent. This result is consistent with other studies in which the initiation of the HR required Prf (Chang et al., 2002; Rathjen et al., 1999). The constitutively active PtoL205D mimics the action of the Pto-AvrPto interaction. This histochemical study, combined with the use of prf-3 mutant plants, demonstrates that Prf is upstream to early events in the signaling pathway leading to the HR.
Unlike the R-gene-mediated HR as described above in the Prf-Pto model, the mechanism of ToxA-initiated cell death is unknown in the literature. Recently a nuclear-encoded chloroplast protein was identified to be the target of ToxA, which can explain the mechanism of ToxA-caused necrosis (Tai et al. unpublished data). The results presented here show that the histochemical technique is applicable to distinguish ToxA-induced necrosis from the HR.
The author thanks Dr. Richard W. Michelmore of University of California at Davis for providing the tomato and N. benthamiana plants, the members in his and my labs for technical assistance and Dr. Steven W. Meinhardt of North Dakota State University for the purified ToxA protein. I thank Belinda Martineau for helping the technical writing, Dr. Jinguo Hu, Dr. Lynn Dahleen and Dr. Michael Edwards for critical comments. I gratefully acknowledge the financial support from the NSF Cooperative Agreement BIR-8920216 to CEPRAP and from the USDA CRIS project No. 5442-21000-030-00D.