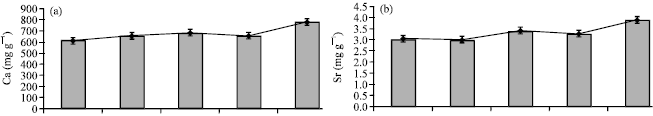
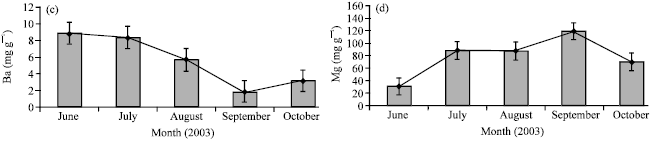
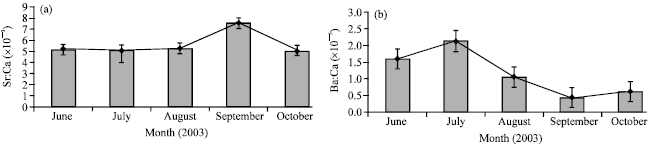
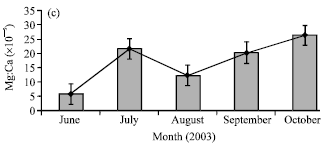
Research Article
Variation of Ca, Sr, Ba and Mg in the Otolith of Mudskipper in West Coast of Peninsular Malaysia
School of Environmental and Natural Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia
M.A. Ghaffar
School of Environmental and Natural Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia
C.A.R. Mohamed
Marine Ecosystem Research Centre, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia