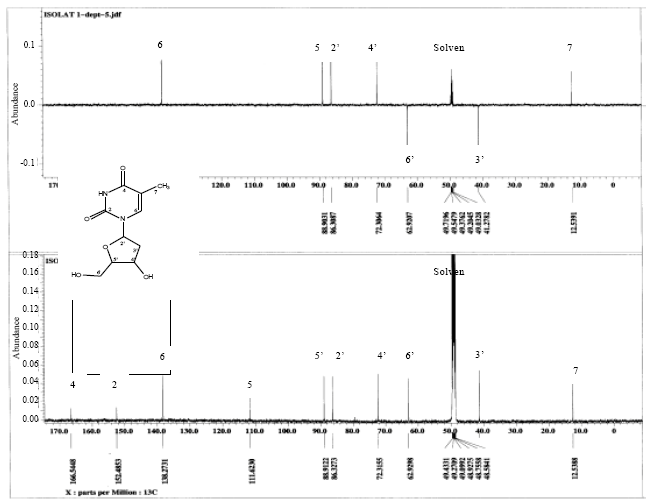
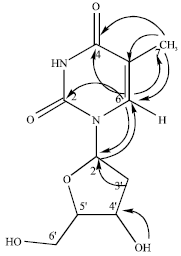
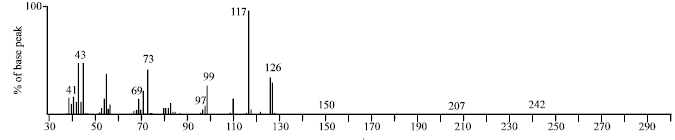
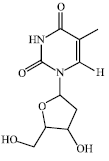
Research Article
Identification of Cytotoxic Constituent of Indonesian Sponge Kaliapsis sp. (Bowerbank)
Faculty of Pharmacy, Gadjah Mada University, Sekip Utara, Yogyakarta, Indonesia
U. A. Jenie
Faculty of Pharmacy, Gadjah Mada University, Sekip Utara, Yogyakarta, Indonesia
Sudarsono
Faculty of Pharmacy, Gadjah Mada University, Sekip Utara, Yogyakarta, Indonesia
L.B. S. Kardono
Indonesian Institute of Science (LIPI), Jl. Gatot Subroto 10, Jakarta, Indonesia
R. Rahmat
Indonesian Institute of Science (LIPI), Jl. Gatot Subroto 10, Jakarta, Indonesia